SpinePoint LLC announces U.S. FDA 510(k) clearance of the Flex-Z™ Cervical Cage (K252432) on March 25, 2026 — the first cleared product in an eight-device ultra low stiffness interbody platform protected by IP through 2043. Built from 3D-printed porous titanium, the Flex-Z™ is engineered to match cancellous bone stiffness in anterior cervical fusion RENO, Nev., March … [Read more...] about SpinePoint Receives U.S. FDA 510(k) Clearance for Flex-Z™ Cervical Cage/ First Product in the Ultra-Low Stiffness Cushioning Interbody Platform
2026
Medtronic and Merit Medical Systems enter into agreement to offer new, FDA-cleared ViaVerte™ system, a basivertebral nerve ablation treatment for chronic vertebrogenic lower-back pain
Exclusive agreement expands Medtronic's comprehensive pain interventions portfolio, increases treatment options for patients living with chronic pain GALWAY, Ireland, March 24, 2026 /PRNewswire/ -- Medtronic plc, a global leader in healthcare technology, today announced that it has entered into a distribution agreement with Merit Medical Systems, Inc. (NASDAQ: MMSI), to … [Read more...] about Medtronic and Merit Medical Systems enter into agreement to offer new, FDA-cleared ViaVerte™ system, a basivertebral nerve ablation treatment for chronic vertebrogenic lower-back pain
ATEC’s Valence: Robotics in Service of a Procedural Strategy
ATEC is no longer just a fast-growing spine company adding another technology box to its portfolio. With $764 million in revenue in 2025, up 25% year over year, it is increasingly behaving like a company trying to build procedural control around the areas where it already believes it has competitive leverage. That is the real significance of Valence, ATEC’s robotic solution … [Read more...] about ATEC’s Valence: Robotics in Service of a Procedural Strategy
VySpine Reaches 20th FDA Clearance with DiversiVy Facet Screw System
VySpine has received FDA 510(k) clearance for its new DiversiVy Facet Screw System, a device intended for use in the lumbosacral spine to provide stabilization and support fusion procedures. The clearance is another notable milestone for the company, which says this is its 20th FDA clearance in less than five years. For a relatively small player in the spine market, that … [Read more...] about VySpine Reaches 20th FDA Clearance with DiversiVy Facet Screw System
The Regulatory Trap: FDA and MDR Are Milestones. They Are Not a Business Model
In medtech, including the spine business, few goals generate as much obsession as obtaining FDA clearance in the United States or CE marking under MDR in Europe. For many startups, that milestone eventually becomes the company’s central focus. Everything revolves around approval: the product, trials, documentation, investors, timelines, and resources. The problem is that … [Read more...] about The Regulatory Trap: FDA and MDR Are Milestones. They Are Not a Business Model
What Really Worries Surgeons About Subsidence? A practical industry perspective on what Cage Subsidence means in daily spine surgery
In spine surgery, cage subsidence is often framed as a radiographic complication or an implant-related event. Yet its real significance lies elsewhere. What makes subsidence important is not the image itself, but what it may reveal and what it may compromise. As the cage settles, disc height can decrease, indirect decompression may be reduced, segmental correction can be … [Read more...] about What Really Worries Surgeons About Subsidence? A practical industry perspective on what Cage Subsidence means in daily spine surgery
BioRestorative Therapies to Present Positive Phase 2 Blinded Data for BRTX-100 Demonstrating Meaningful Improvements in Pain and Function in Chronic Lumbar Disc Disease
New blinded data from ~45 patients to date in key pain and function scales and safety and tolerability of the Company’s proprietary hypoxic-cultured mesenchymal stem cells to be unveiled on March 28th at the 2026 Orthopaedic Research Society Annual Meeting MELVILLE, N.Y., March 19, 2026 (GLOBE NEWSWIRE) -- BioRestorative Therapies, Inc. (“BioRestorative,” “BRTX,” or the … [Read more...] about BioRestorative Therapies to Present Positive Phase 2 Blinded Data for BRTX-100 Demonstrating Meaningful Improvements in Pain and Function in Chronic Lumbar Disc Disease
First Subjects Randomized in Clinical Trial Evaluating the Benefits of Total Segmental Fusion in Treating Degenerative Conditions in the Lumbar Spine
PLEASANTON, Calif., March 19, 2026 /PRNewswire/ -- Providence Medical Technology, Inc., a pioneer in less invasive solutions for spine surgery, today announced enrollment of the first patients in its prospective, randomized controlled trial (RCT) evaluating the CORUS-LX System for the treatment of degenerative lumbosacral disease. This multi-center RCT will … [Read more...] about First Subjects Randomized in Clinical Trial Evaluating the Benefits of Total Segmental Fusion in Treating Degenerative Conditions in the Lumbar Spine
Spinal Elements Expands Ventana® Platform with FDA 510(k) Clearance and First Cases of Ventana® A ALIF System
CARLSBAD, Calif., March 18, 2026 /PRNewswire/ -- Spinal Elements®, a spine technology company known for developing innovative surgical solutions that prioritize clinical performance and surgical efficiency, today announced 510(k) clearance from the FDA and the first cases using its Ventana A Anterior Lumbar Interbody Fusion (ALIF) System. Ventana A strengthens Spinal Elements' … [Read more...] about Spinal Elements Expands Ventana® Platform with FDA 510(k) Clearance and First Cases of Ventana® A ALIF System
Kuros Biosciences delivers 72% year-over-year sales growth, reaching USD 146.1 million for 2025
Kuros Biosciences delivers 72% year-over-year sales growth, reaching USD 146.1 million for 2025 Financial Highlights Schlieren (Zurich), Switzerland, March 10, 2026 – Kuros Biosciences (“Kuros” or the “Company”) a leader in next generation bone healing technologies, today announced its financial and operational results for the full year 2025, marking another year … [Read more...] about Kuros Biosciences delivers 72% year-over-year sales growth, reaching USD 146.1 million for 2025
Sanara MedTech Inc. Announces Publication of Peer-Reviewed Study Evaluating the Economic and Clinical Value of CellerateRX® Surgical Powder in the Management of Spine Surgery Wounds
Study demonstrates cost savings and improved health outcomes associated with the use of CellerateRX® as an adjunct to the standard of care for high-risk spinal surgery patients, compared to the standard of care alone FORT WORTH, TX, March 11, 2026 (GLOBE NEWSWIRE) -- Sanara MedTech Inc. (“Sanara,” the “Company,” “we,” “our” or “us”) (Nasdaq: SMTI), a medical … [Read more...] about Sanara MedTech Inc. Announces Publication of Peer-Reviewed Study Evaluating the Economic and Clinical Value of CellerateRX® Surgical Powder in the Management of Spine Surgery Wounds
+10 Anterior Buttress Plates to Know…! | Updated 2026
Buttress plates play a simple but important role in spine surgery. Rather than providing general fixation alone, they are mainly used to help keep a graft, cage, or reconstructed vertebral segment in place and to reduce the risk of migration after surgery. Their value is especially clear in procedures such as corpectomy, trauma cases, and other anterior reconstructions, where … [Read more...] about +10 Anterior Buttress Plates to Know…! | Updated 2026
Beyond Imports: The Argentine Spine Manufacturers Shaping a Growing Market | 2026 Update
The Argentine spine implants market is already big enough to support its own industrial base, a specialized commercial network, and a local competitive landscape worth watching. In 2024, the Argentine spinal implants market is estimated at USD 146.5 million, with a projection of USD 285.3 million by 2033 (7.8% compound annual growth rate). Within the market, the largest … [Read more...] about Beyond Imports: The Argentine Spine Manufacturers Shaping a Growing Market | 2026 Update
From Early Innovations to Modern Practice: Where Are Dynamic and Semi-Rigid Spine Systems Today?
In recent days, we published “Russian Researchers Advance Flexible Spine Implants for Motion Preservation.” Today, I would like to republish this article that first appeared in October of last year, offering readers a broader view of the dynamic and flexible spine systems currently on the market, as well as those that have since disappeared or are no longer in use. This was … [Read more...] about From Early Innovations to Modern Practice: Where Are Dynamic and Semi-Rigid Spine Systems Today?
Russian Researchers Advance Flexible Spine Implants for Motion Preservation
The Moscow Aviation Institute (MAI) has announced progress in the development of a new generation of flexible spinal implants designed to preserve natural spinal mobility after surgery while improving reliability, service life, and biocompatibility. The project addresses a familiar challenge in spine surgery: how to stabilize the spine without fully eliminating physiological … [Read more...] about Russian Researchers Advance Flexible Spine Implants for Motion Preservation
Fziomed Announces Clinical Results from Level I Study Evaluating Oxiplex Absorbable Gel for Spine Surgery
SAN LUIS OBISPO, Calif., March 10, 2026 /PRNewswire/ -- Fziomed, Inc. ("Fziomed" or the "Company"), a recognized global leader in postsurgical adhesion prevention with the best-in-class synthetic, absorbable gel technology platform, today announced results from a prospective, randomized, double-blinded, multi-center clinical study demonstrating that Oxiplex® … [Read more...] about Fziomed Announces Clinical Results from Level I Study Evaluating Oxiplex Absorbable Gel for Spine Surgery
VB Spine Announces Successful First Clinical Cases of Vulcan™ Spinal System
NEW YORK--(BUSINESS WIRE)--VB Spine LLC (“VB Spine”), today announced the successful completion of the first clinical cases utilizing the Vulcan™ Spinal System for thoracolumbar fixation. The milestone marks the system’s entry into Limited Market Release and represents a significant step forward in the company’s strategy to expand its next-generation implant portfolio. The … [Read more...] about VB Spine Announces Successful First Clinical Cases of Vulcan™ Spinal System
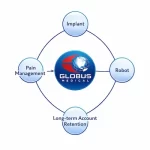
Globus Medical’s Strategy: Building a Closed-Loop Spine Ecosystem
Globus Medical is not simply entering 2026 with an “aggressive” posture. It is attempting something structurally more ambitious: building a self-reinforcing spine ecosystem that compresses competitive space around it. This is not a growth story. It is a control story. Spine as the Economic Engine Roughly 95% of revenue still comes from spine. Over 80% of that from the … [Read more...] about Globus Medical’s Strategy: Building a Closed-Loop Spine Ecosystem
SMAIO Announces a €3.6 Million Capital Increase to Support Its Long-Term Growth Momentum
DALLAS & LYON, France--(BUSINESS WIRE)--Regulatory News: This press release may not be published, distributed or disseminated, directly or indirectly, in the United States of America or Canada, Australia, Japan or South Africa. SMAIO (Software, Machines and Adaptive Implants in Orthopaedics – Euronext Growth Paris, ISIN: FR0014005I80 / Ticker: … [Read more...] about SMAIO Announces a €3.6 Million Capital Increase to Support Its Long-Term Growth Momentum