Russia was never a transparent spine market. Large enough to matter, technically capable in selected centers, quietly dependent on imported premium technology. That was the working summary most international companies operated on before 2022. The picture has shifted since. Outside visibility has dropped, domestic industrial pressure has risen, and import substitution, long … [Read more...] about Sanctions, Titanium and Pedicle Screws: How Russia’s Spine Market Is Rebuilding Itself
2026
aljeel Medical Partners as Distributor for DePuy Synthes by Johnson & Johnson in Saudi Arabia
aljeel Medical has entered into a strategic distribution agreement with Johnson and Johnson Medical Saudi Arabia, becoming the orthopedics distributor for DePuy Synthes products in Saudi Arabia. The agreement is intended to improve access for healthcare providers to advanced, high-quality medical solutions, supported by an efficient and coordinated distribution network. The … [Read more...] about aljeel Medical Partners as Distributor for DePuy Synthes by Johnson & Johnson in Saudi Arabia
Hospital Access Is Gone. What Comes Next for Spine Field Sales?
What happens when the environment that built a generation of elite field reps no longer exists — and a new generation is building careers without ever knowing what they missed? There's a particular kind of clarity that comes from watching an industry transform from the inside. Veterans of spine sales describe an era when hospital access was assumed, relationships were built … [Read more...] about Hospital Access Is Gone. What Comes Next for Spine Field Sales?
REALISTS Officially Launches in the United States
DELAWARE, OH, UNITED STATES, May 14, 2026 /EINPresswire.com/ -- REALISTS Training Technologies AG, a Germany-based company focused on spine surgery simulation and surgical training, today announced its official launch in the United States. The expansion includes the formation of a new U.S. subsidiary, REALISTS USA, Inc., and the appointment of Greg Rhinehart as Vice President … [Read more...] about REALISTS Officially Launches in the United States
Amplify Surgical and Emory Healthcare Complete World’s First Continuously Navigated Endoscopic TLIF Using the dualPortal & dualX Platform
Amplify Surgical announced the successful completion of what is believed to be the world's first continuously navigated endoscopic transforaminal lumbar interbody fusion (TLIF) procedure, performed at Emory Healthcare by Dr. Daniel Refai and his surgical team. The procedure represents a significant advancement in minimally invasive spine surgery, combining real-time navigation … [Read more...] about Amplify Surgical and Emory Healthcare Complete World’s First Continuously Navigated Endoscopic TLIF Using the dualPortal & dualX Platform
Expandable Cervical Cages: Innovation or Just a Niche Tool? | Updated May 2026
Anterior cervical discectomy and fusion (ACDF) is one of the most standardized procedures in spine surgery. The workflow is familiar: decompression, controlled distraction, sizing, and insertion of a static interbody cage. For decades, this has worked reliably and predictably. So when expandable cages appeared in the cervical spine, the reaction was immediate and … [Read more...] about Expandable Cervical Cages: Innovation or Just a Niche Tool? | Updated May 2026
We are proud to announce that L&K SPINE will be a Sponsor of SPINEMarketGroup in 2026!
Thank you, L&K SPINE! On behalf of the SPINEMarketGroup team, we sincerely appreciate your support as a PLATINUM Sponsor for 2026. We are proud to welcome L&K SPINE as a sponsor for the first time and look forward to starting this journey together. About L&K Spine L&K Biomed, whose name stands for Learning & Knowledge Biomed, applies knowledge … [Read more...] about We are proud to announce that L&K SPINE will be a Sponsor of SPINEMarketGroup in 2026!
L&K Biomed Clears the FDA with a Height-Expandable Cervical Cage — Entering a Segment Where Almost Nobody Plays
On May 16, 2026, L&K Biomed received FDA 510(k) clearance for the BluEx-C, a height-expandable cervical interbody cage. The clearance lands at the right moment for a Korean spine company that has spent the better part of the past three years systematically building its U.S. regulatory footprint — and it does so in a segment that, by the company's own characterization, has … [Read more...] about L&K Biomed Clears the FDA with a Height-Expandable Cervical Cage — Entering a Segment Where Almost Nobody Plays
SpineCraft Announces U.S. FDA 510(k) Clearance of ANTERIS Thoracolumbar Plate System
Westmont, Illinois – SpineCraft, a U.S.-based spine surgery systems developer and manufacturer, announces U.S. FDA 510(k) clearance of the ANTERIS Thoracolumbar Plate System, a comprehensive anterior fixation solution designed to provide supplemental stabilization for anterior lumbar interbody fusion procedures. The ANTERIS plate system is intended for use with anterior … [Read more...] about SpineCraft Announces U.S. FDA 510(k) Clearance of ANTERIS Thoracolumbar Plate System
How a $1,400 Spine Procedure Became a $34,000 Arbitration Award Under the No Surprises Act
The No Surprises Act was designed to protect Americans from unexpected medical bills. Five years later, the patient may be better protected — but the system created to settle the remaining disputes is producing a different problem. In some cases, arbitration awards appear far removed from the prices paid elsewhere in the market, turning a law built around patient protection … [Read more...] about How a $1,400 Spine Procedure Became a $34,000 Arbitration Award Under the No Surprises Act
DeGen Medical Expands Patient-Specific Spine Platform
Florence, SC, May 18, 2026 (GLOBE NEWSWIRE) -- DeGen Medical announced continued expansion of its patient-specific implant (PSI) platform designed to provide surgeons with patient-specific solutions for complex spinal pathologies while maintaining compatibility with a streamlined off-the-shelf workflow. DeGen Medical offers patient-specific implants, off-the-shelf implant … [Read more...] about DeGen Medical Expands Patient-Specific Spine Platform
Boston Scientific Puts $1.5 Billion on MiRus: Why the Spine Market Should Look Beyond TAVR
On May 18, Boston Scientific announced a $1.5 billion investment for an approximate 34% equity stake in MiRus LLC, a privately held company based in Georgia and focused on biomaterials, implants, and procedural solutions for cardiovascular and orthopedic diseases. Most of the early coverage has naturally focused on the TAVR opportunity. That is where the clearest near-term … [Read more...] about Boston Scientific Puts $1.5 Billion on MiRus: Why the Spine Market Should Look Beyond TAVR
The ASC Opportunity in Spine Will Be Won by Commercial Redesign, Not by Product Alone
Spine surgery is moving toward ambulatory surgery centers. That part of the conversation is largely settled. What remains underexamined — especially at the executive level — is the commercial question underneath it: what does a sustainable spine business actually look like when the customer is no longer a large hospital system, but a cost-sensitive, operationally lean surgical … [Read more...] about The ASC Opportunity in Spine Will Be Won by Commercial Redesign, Not by Product Alone
PainTEQ Launches TRAQ™, A Next-Generation Posterior Approach to SI Joint Fusion
Built on the proven LINQ® posterior approach, TRAQ™ adds bi-cortical fixation to deliver enhanced sacropelvic stability TAMPA, Fla., May 15, 2026 /PRNewswire/ -- PainTEQ, the pioneer of posterior sacroiliac joint fusion, today announced the U.S. commercial launch of TRAQ™ SI Joint Fusion — the first posterior SI joint implant designed to engage both the sacral … [Read more...] about PainTEQ Launches TRAQ™, A Next-Generation Posterior Approach to SI Joint Fusion
Xtant Medical Reports First Quarter 2026 Financial Results
BELGRADE, Mont., May 13, 2026 /PRNewswire/ -- Xtant Medical Holdings, Inc. (NYSE American: XTNT), a medical technology company focused on surgical solutions for spinal and other orthopedic conditions, today reported financial and operating results for the first quarter ended March 31, 2026. First Quarter 2026 Financial Highlights First Quarter 2026 … [Read more...] about Xtant Medical Reports First Quarter 2026 Financial Results
Autonomous Pedicle Screw Placement: Is Spine Robotics Ready for the Next Step?
Robotic guidance, navigation and planning are no longer experimental ideas in spine. They are already part of the market. Different systems follow different strategies, but the underlying message is the same: robotics has become part of how major spine companies want to compete. Still, most of what we call spine robotics today remains robotic assistance. The surgeon is still … [Read more...] about Autonomous Pedicle Screw Placement: Is Spine Robotics Ready for the Next Step?
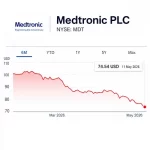
Medtronic Hits a 52-Week Low: What Is the Market Seeing?
In recent weeks, much of the spine market discussion has focused on Globus Medical and the negative market reaction that followed otherwise strong operating results. But Globus is not alone. Medtronic, one of its most direct competitors in spine technology, has also been under pressure. Its stock recently hit a reported 52-week low at $77.15, and by May 11, 2026, was quoted … [Read more...] about Medtronic Hits a 52-Week Low: What Is the Market Seeing?
SMAIO Expands Its North American Scientific Advisory Board With a New Renowned Surgeon
DALLAS & LYON, France--(BUSINESS WIRE)--Regulatory News: SMAIO (Software, Machines and Adaptative Implants in Orthopaedics – Euronext Growth Paris, ISIN: FR0014005I80 / Ticker: ALSMA), a French-American company specialized in complex spine surgery and offering an integrated pre-, intra- and postoperative solution based on 3D planning software, … [Read more...] about SMAIO Expands Its North American Scientific Advisory Board With a New Renowned Surgeon
Cervical Fusion Implant Denials: Why Spine Societies Are Pushing Back
There are reimbursement disputes that affect only the margins of a market. Others say something more structural about the direction of the industry. The recent statement from leading U.S. spine surgery societies on cervical fusion implant denials belongs to the second category. The issue is not a new device seeking early coverage, nor an experimental technology asking for … [Read more...] about Cervical Fusion Implant Denials: Why Spine Societies Are Pushing Back