Globus Medical’s latest results created an apparent contradiction. The company reported a very strong first quarter, raised its earnings guidance, continued to show the benefits of the NuVasive integration, and reinforced its position as one of the leading players in spine. Yet the stock did not move as many would have expected. In fact, the immediate market reaction was … [Read more...] about Globus Medical: Strong Q1, Weak Stock Reaction — Is the Market Asking for More?
2026
Globus Medical: Stronger in Spine, but Could DePuy Synthes’ Trauma Business Be the Next Big Move?
Globus Medical’s first quarter 2026 results confirm something important: the company is no longer just digesting major acquisitions. It is beginning to show the operating logic behind them. The headline figures are strong, but the more relevant point is the quality of the performance. Growth came not only from Nevro, but also from the underlying business, particularly U.S. … [Read more...] about Globus Medical: Stronger in Spine, but Could DePuy Synthes’ Trauma Business Be the Next Big Move?
Globus Medical Reports First Quarter 2026 Results
AUDUBON, Pa., May 07, 2026 (GLOBE NEWSWIRE) -- Globus Medical, Inc. (NYSE: GMED), a leading musculoskeletal technology company, today announced its financial results for the first quarter ended March 31, 2026. First Quarter 2026: “We’re off to a strong start in 2026 with 27% overall revenue growth and record first quarter earnings. Organic … [Read more...] about Globus Medical Reports First Quarter 2026 Results
Aurora Spine Announces Issuance of Fourth U.S. Patent Covering DEXA Technology® Bone Density-Matched Implants
CARLSBAD, Calif., May 07, 2026 (GLOBE NEWSWIRE) -- Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSXV: ASG) (OTCQB: ASAPF), a leader in the development of groundbreaking medical devices for spinal surgery, today announced the issuance of its fourth U.S. patent related to Aurora’s proprietary DEXA Technology® platform. The newly issued patent, U.S. Patent No. … [Read more...] about Aurora Spine Announces Issuance of Fourth U.S. Patent Covering DEXA Technology® Bone Density-Matched Implants
Carlsmed® Reports First Quarter 2026 Financial
First quarter 2026 revenue of $16.1 million, representing 58% growth year-over-year Full year 2026 revenue guidance raised to $72 million to $77 million CARLSBAD, Calif., May 05, 2026 (GLOBE NEWSWIRE) -- Carlsmed, Inc. (Nasdaq: CARL) (“Carlsmed” or the “Company”), a medical technology company pioneering AI-enabled personalized spine surgery solutions, today reported … [Read more...] about Carlsmed® Reports First Quarter 2026 Financial
ATEC Reports First Quarter Financial Results
CARLSBAD, Calif--(BUSINESS WIRE)-- Alphatec Holdings, Inc. (Nasdaq: ATEC), a spine-focused provider of innovative solutions dedicated to revolutionizing the approach to spine surgery, today announced financial results for the quarter ended March 31, 2026, and business highlights. First Quarter 2026 Financial Results First Quarter … [Read more...] about ATEC Reports First Quarter Financial Results
SMAIO Announces that Portzamparc (BNP Paribas Group) has Initiated Coverage of its Stock with a Buy Rating
DALLAS & LYON, France--(BUSINESS WIRE)--Regulatory News: SMAIO (Software, Machines and Adaptive Implants in Orthopaedics – Euronext Growth Paris, ISIN Code: FR0014005I80 / Ticker: ALSMA), a French-American company specialized in complex spine surgery and offering an integrated pre-, intra- and postoperative solution based on 3D planning software, adaptive implants and … [Read more...] about SMAIO Announces that Portzamparc (BNP Paribas Group) has Initiated Coverage of its Stock with a Buy Rating
Orthofix Reports First Quarter 2026 Results Highlighting Steady Execution Following Spine Commercial Channel Actions; Reaffirms Full-Year 2026 Financial Guidance
LEWISVILLE, Texas--(BUSINESS WIRE)--Orthofix Medical Inc. (NASDAQ:OFIX), a leading global medical technology company, today reported its financial results for the first quarter ended March 31, 2026, reflecting steady execution following recent spine commercial channel actions. Based on first-quarter performance, the Company reaffirmed its full-year 2026 financial guidance. All … [Read more...] about Orthofix Reports First Quarter 2026 Results Highlighting Steady Execution Following Spine Commercial Channel Actions; Reaffirms Full-Year 2026 Financial Guidance
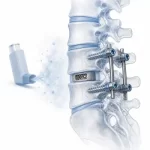
Inhaled Corticosteroids and Lumbar Fusion: A New Revision Risk Signal?
A new study published in Global Spine Journal, carried out by researchers from Johns Hopkins together with collaborators from the All India Institute of Medical Sciences (AIIMS) in New Delhi, has put an unusual detail on the radar of spine surgeons: the long-term use of inhaled corticosteroids in asthma patients. At first sight, this may sound like a minor point. Inhaled … [Read more...] about Inhaled Corticosteroids and Lumbar Fusion: A New Revision Risk Signal?
Spineway : First-quarter 2026 revenue
First-quarter 2026 revenue Spineway, a specialist in innovative implants for the treatment of severe spine disorders, recorded revenue of €2.2 million in the first quarter of 2026. Following strong growth in the fourth quarter of 2025 (+38%), the slowdown observed in the first quarter of 2026 will not undermine the Group’s momentum. It is mainly attributable to delays … [Read more...] about Spineway : First-quarter 2026 revenue
Theradaptive Secures FDA Approval to Advance OASIS Trial to Pivotal Study Evaluating OsteoAdapt® SP in Spinal Fusion
FDA authorization marks critical milestone enabling advancement of next-generation bone regeneration therapy for patients with degenerative spinal diseases FREDERICK, Md., April 30, 2026 /PRNewswire/ -- Theradaptive, Inc., a clinical-stage biotechnology company pioneering targeted protein therapeutics for tissue regeneration, today announced that the U.S. Food … [Read more...] about Theradaptive Secures FDA Approval to Advance OASIS Trial to Pivotal Study Evaluating OsteoAdapt® SP in Spinal Fusion
Demetra acquires Maxxspine Innovative GmbH, a company that has been a leader in advanced spinal solutions for over twenty years, further strengthening its position as a benchmark in spine surgery
MILAN, April 30, 2026 /PRNewswire/ -- Demetra, an innovative and independent international medical company leader providing bone cements, spacers and biomaterials for orthopedic, spine and neuro applications and orthopedic infection control, announces the successful completion of the acquisition of Maxxspine Innovative GmbH, a German-based company specialized in … [Read more...] about Demetra acquires Maxxspine Innovative GmbH, a company that has been a leader in advanced spinal solutions for over twenty years, further strengthening its position as a benchmark in spine surgery
VB Spine Announces VB Spine Solutions, a Partnership-Driven Program to Help Hospitals Acquire Technology
New partnership-driven approach expands access to spine technology NEW YORK--(BUSINESS WIRE)--VB Spine LLC (“VB Spine”), today launched VB Spine Solutions, a program designed to bring critical spine technology to hospitals faster, with structures built around patient need. While focused on spine, VB Spine Solutions aligns with hospitals to simplify how capital equipment … [Read more...] about VB Spine Announces VB Spine Solutions, a Partnership-Driven Program to Help Hospitals Acquire Technology
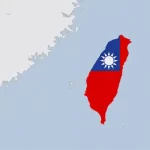
Taiwan’s Spine Industry: Why a Small Market Could Become a Serious Export Hub
The Taiwanese spine market is relatively small, it has a sophisticated healthcare system, broad access to care and strong medical infrastructure. However, reimbursement discipline and price pressure remain important realities. For spine companies, this creates a demanding environment: surgeons may be open to advanced technologies, but hospitals remain highly sensitive to cost, … [Read more...] about Taiwan’s Spine Industry: Why a Small Market Could Become a Serious Export Hub
Amnovis expands to the United States and acquires Westconn Precision Technologies’ additive manufacturing activities
Aarschot, Belgium - April 29, 2026--Amnovis, a Belgian additive manufacturing contract manufacturing organization serving regulated industries including medical devices, announces the establishment of a new operational entity in the United States and the acquisition of the additive manufacturing activities of Westconn Precision Technologies. Together, these steps represent a … [Read more...] about Amnovis expands to the United States and acquires Westconn Precision Technologies’ additive manufacturing activities
Life Spine Announces Expanded Indications for ARx® SAI Spinal Fixation System
HUNTLEY, Ill.--(BUSINESS WIRE)--Life Spine, Inc., a medical device company that designs, develops, manufactures, and markets products for the surgical treatment of spinal disorders, announced today their 510k for ARx SAI (Sacral Alar Iliac) Spinal Fixation System was approved. ARx SAI has expanded its compatibility clearance and is now approved for use with compatible pedicle … [Read more...] about Life Spine Announces Expanded Indications for ARx® SAI Spinal Fixation System
Official launch of three New PediGuard® Models during the Annual Scoliosis Meeting in China
PARIS and BOULDER (CO), April 28, 2026 – 06:00pm CEST - SpineGuard (FR0011464452 - ALSGD), an innovative company that deploys its DSG® (Dynamic Surgical Guidance) local conductivity sensing technology to secure and streamline the placement of bone implants, today announced the official launch of the newly cleared Curved and XS PediGuard models, in … [Read more...] about Official launch of three New PediGuard® Models during the Annual Scoliosis Meeting in China
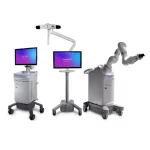
Medtronic announces CE Mark for Stealth AXiS™ surgical system
Following recent FDA clearances, Medtronic is accelerating access to integrated planning, navigation, and robotics platform across Europe GALWAY, Ireland, April 28, 2026 /PRNewswire/ -- Medtronic, a global leader in healthcare technology, has received CE mark for the Stealth AXiS™ surgical system, a next-generation platform that brings planning, navigation, … [Read more...] about Medtronic announces CE Mark for Stealth AXiS™ surgical system
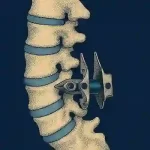
Do Interspinous Spacers Still Have a Place in Spine Surgery? | Updated 2026
Last week, we published the news that Amplio Spine, LLC had signed a Letter of Intent to acquire the FDA-cleared KeyLift™ Expandable Interlaminar Stabilization System from FloSpine, LLC. This announcement brings interspinous and interlaminar implants back into focus. For that reason, we have updated our overview of this product category and its market, looking not only at … [Read more...] about Do Interspinous Spacers Still Have a Place in Spine Surgery? | Updated 2026