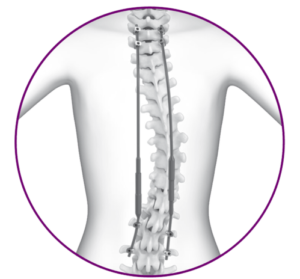
SAN DIEGO, July 19, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the U.S. launch of MAGEC X, a new procedural solution within the MAGEC product family that addresses Early Onset Scoliosis (EOS). MAGEC X features updates across all rod diameters […]
LAUNCHES
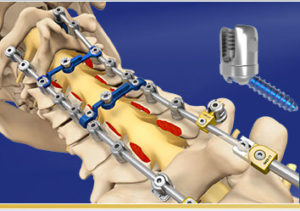
Globus Medical Launches Cement Augmented Pedicle Screw System
AUDUBON, Pa., June 13, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the launch of the CREO® Fenestrated Screw System used in the treatment of patients with advanced stage tumors of the thoracolumbar spine. CREO® fenestrated screws, when augmented with the company’s FORTRESS™ radiopaque bone cement, are designed […]
NuVasive Launches AttraX Scaffold Biologic And Reports First Clinical Use
SAN DIEGO, June 12, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the U.S. launch of AttraX® Scaffold, an absorbent ceramic-collagen bone graft with an optimized surface that has been validated in preclinical testing to drive increased bone formation and faster fusion […]
RTI Surgical® Announces Commercial Launch of Fortilink®-TS and -L IBF Systems with TETRAfuse® 3D Technology
ALACHUA, Fla.–RTI Surgical, Inc. (Nasdaq: RTIX), a global surgical implant company, announced the full commercial launch of its Fortilink®-TS and -L IBF Systems, adding to a growing series of interbody fusion devices featuring proprietary TETRAfuse® 3D Technology. The Fortilink-TS and -L Systems are intended for use in lumbar interbody fusion procedures at one or two […]
CoreLink Announces Full Commercial Launch of Foundation™ 3D Printed Titanium Interbody Cage Systems
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a fast growing, vertically integrated designer and manufacturer of spinal implant systems, announced the full commercial release of its Foundation 3D Interbody Cage Systems for cervical and lumbar fusions. The titanium devices utilize additive manufacturing to create Mimetic Metal™ technology that mimics key characteristics of natural bone, featuring 100% open-pore architecture […]
Globus Medical Announces Launch of ELSA® Expandable Lateral Spacer System
AUDUBON, Pa., April 05, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the launch of two innovative lateral expandable interbody spacers with integrated screw fixation, ELSA® AL and ELSA®-ATP, the latest additions to Globus’ unmatched expandable solutions. These next generation expandable implants provide an optimized fit for the […]
K2M Launches Next-Generation YUKON™ OCT Spinal System at AANS/CNS 2018
LEESBURG, Va., March 14, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced the commercial launch of the YUKON™ OCT Spinal System at the 34th Annual Meeting of the American Association of Neurological Surgeons/Congress of Neurological Surgeons Section […]
Astura Medical Announces Initial Cases and Full Commercial Release For BRIDALVEIL OCT Stabilization System
CARLSBAD, CA – February 21, 2018 – Astura Medical, a high growth, innovative spine technology company, announced today the completion of the initial surgeries and full commercial release for its BRIDALVEIL Occipital-Cervico-Thoracic (OCT) System. The first cases were successfully completed at multiple hospitals across the country, including at the University of Colorado Hospital by Dr. […]
Spine Wave Launches Proficient® Posterior Cervical Spine System Implants for Complex Posterior Cervical-Thoracic Procedures
SHELTON, Conn., Feb. 21, 2018 (GLOBE NEWSWIRE) — Spine Wave is increasing the Proficient® Posterior Cervical Spine System’s product offering to further serve the posterior cervical spine market. The system is gaining rapid adoption by spine surgeons, primarily due to its patented tri-lobe polyaxial screw design which offers a best in class 120 degrees of […]
SeaSpine Expands Ventura™ NanoMetalene® Implant Offering to Accommodate Larger Range of Posterior Procedures
CARLSBAD, Calif., Jan. 31, 2018 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ:SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the portfolio expansion of the Ventura™ NanoMetalene® posterior interbody device. Ventura NanoMetalene, which has been commercially available since 2015, has expanded its size offerings to accommodate […]
Spine Wave Announces the Commercial Launch of the GraftMag® Graft Delivery System
SHELTON, Conn., Jan. 23, 2018 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce its commercial launch of the GraftMag® Graft Delivery System. The GraftMag® Graft Delivery System is designed to safely and rapidly deliver large amounts of bone graft. The system can transform the tedious and sometimes frustrating graft delivery process into a more efficient […]
SpineGuard and XingRong Medical Launch PediGuard® in China
SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices to make spine surgery safer, announced today that XinRong Medical Group will officially launch the Classic PediGuard product range in China during the Chinese Orthopedic Association (COA) annual meeting being held in Zhuhai, China from November 15-18. The COA meeting […]
AxioMed Announces Release of Lateral Viscoelastic Total Disc Replacement at NASS 2017
BOSTON, MASS. (PRWEB) OCTOBER 25, 2017–AxioMed announces the release of its lateral total disc replacement at the North American Spine Society (NASS) 2017 Annual Meeting. The company is expecting the first surgery to be done in Jamaica by Dr. Kingsley R. Chin before the end of this year. AxioMed has hired two top executives – Chief […]
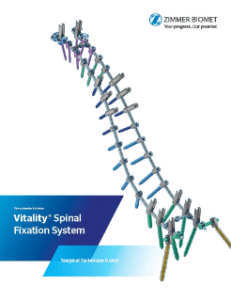
Zimmer Biomet Announces U.S. Launch of Vitality®+ and Vital™ Spinal Fixation Systems
WARSAW, Ind., Oct. 25, 2017 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced the official launch of its Vitality®+ and Vital™ Spinal Fixation Systems in the United States at the 2017 North American Spine Society (NASS) Annual Meeting. The comprehensive Vitality+ Spinal Fixation System consists […]
RTI Surgical® Launches Fortilink®-C IBF System, the First 3D Printed Polymer-based Interbody Device
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (RTI) (Nasdaq: RTIX), a global surgical implant company, today announced the commercial launch of Fortilink-C IBF System with TETRAfuse® 3D Technology. The Fortilink-C system is the first in a family of devices to incorporate RTI Surgical’s TETRAfuse 3D Technology. “TETRAfuse 3D Technology combines the best characteristics of titanium, allograft bone […]
SpineGuard Announces First Ever DSG™ Sensor-Guided Pedicle Screw System Launch in the USA by Zavation
PARIS & SAN FRANCISCO–Regulatory News:SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices to make spine surgery safer, announced today during the North American Spine Society (NASS) annual meeting being held in Orlando, Florida from October 25-27, the commercial release in the USA of the DSG enabled Z-Direct Pedicle Screw […]
VGI Medical Enters Lateral Spine Market With 1st Of Its Kind Technology
LARGO, FLA. (PRWEB) OCTOBER 24, 2017–VGI Medical, LLC, a privately held medical device company specializing in unique spinal implants, today announced the full the commercial launch of the VerteLP® Lateral Interbody Fusion System. VerteLP was designed to address many of clinical challenges that surgeons face such as insufficient fixation of the implant or interference with pedicle […]
RTI Surgical® Introduces TETRAfuse® 3D Technology Becoming First to Offer 3D Printed Interbody Polymer Optimized to Participate in Fusion
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (RTI) (Nasdaq: RTIX), a global surgical implant company, today announced the introduction of TETRAfuse® 3D Technology, providing surgeons a solution they have been seeking. From the beginning, surgeons have had to choose between bone in-growth, radiolucency or mechanical strength when selecting a spinal interbody implant. TETRAfuse 3D Technology is the […]
Spineology Initiates Post-Market Lateral Interbody Fusion Study Using Novel Implant Design
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., the innovator in anatomy-conserving spine surgery, has announced the initiation of a prospective, post-market lateral interbody fusion study designed to evaluate patient outcomes using its FDA-cleared Duo™ Lumbar Interbody Fusion System. The Duo System offers a unique implant design that is the first to combine PEEK, titanium, and graft containment mesh […]
NuVasive Launches New 3D-Printed Porous Titanium Implant In Expanding Advanced Materials Science Portfolio
SAN DIEGO, Oct. 18, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the launch of Modulus® XLIF® titanium implants. The new 3D-printed, fully porous device provides a differentiated offering for the Company’s flagship XLIF procedure, highlighting a continued commitment […]
4WEB Medical Announces Launch of Next Generation Anterior Spine Truss System
DALLAS, Oct. 4, 2017 /PRNewswire/ — 4WEB Medical, the industry leader in 3D printed implant technology, has announced the launch of its next generation interbody fusion product line for anterior lumbar spine procedures. The new release of the Anterior Spine Truss System recently received FDA clearance for several impactful line extensions along with some key new indications for […]
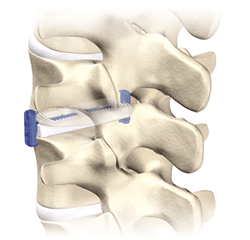
Zimmer Biomet Announces U.S. Launch of Avenue® T TLIF Cage with Integrated VerteBRIDGE® Plating
WARSAW, Ind., Oct. 2, 2017 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced it is officially launching in the United States the Avenue® T TLIF Cage. Avenue T advances posterior lumbar cage technology by incorporating VerteBRIDGE® plating, which facilitates simplified cage insertion and zero-profile, intradiscal fixation through a direct, minimally invasive surgical […]
SeaSpine Announces Launch of Shoreline® Anterior Cervical Standalone System
CARLSBAD, Calif., Sept. 26, 2017 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ:SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the full commercial launch of the Shoreline Anterior Cervical Standalone (ACS) System, featuring TruProfile™ technology. The Shoreline ACS System is designed to deliver maximum flexibility and modularity, offering zero- and […]
K2M Launches EVEREST® Minimally Invasive XTower™ Instrumentation & Showcases 3D Balance ACS™ Platform at SMISS 2017
LEESBURG, Va., Sept. 14, 2017 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced the global launch of the EVEREST® Minimally Invasive (MI) XTower™ instrumentation—an enhancement to the EVEREST MI XT Spinal System—at […]
NuVasive Announces Commercial Launch Of LessRay System Designed To Significantly Reduce Radiation Exposure For Surgeons, Operating Room Staff And Patients
SAN DIEGO, Sept. 13, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the commercial launch of its LessRay® software technology system. The LessRay system, comprised of a propriety software algorithm and hardware components, is designed to help address over exposure to […]
Spine Wave Announces the Commercial Launch of the Proficient® Posterior Cervical Spine System
SHELTON, Conn., Aug. 15, 2017 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce its entry into the posterior cervical fixation market with the successful completion of a limited market release of the Proficient® Posterior Cervical Spine System. The Proficient® Posterior Cervical Spine System substantially strengthens Spine Wave’s position in the cervical spine fixation market […]
Vertera Spine Announces FDA Clearance of COALESCE™ porous PEEK Lumbar Interbody Fusion Device
ATLANTA, July 25, 2017 /PRNewswire/ — Vertera Spine, a developer of medical devices using advanced biomaterial technologies, today announced the COALESCE™ Lumbar Interbody Fusion Device has received U.S. Food and Drug Administration (FDA) 510(k) clearance for use in anterior, transforaminal, posterior, and lateral lumbar interbody fusion procedures. COALESCE features Vertera Spine’s novel proprietary porous PEEK biomaterial and is […]