CARLSBAD, Calif., Aug. 14, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch and completion of initial surgeries of the Mariner® MIS Posterior Fixation System. Mariner MIS is a comprehensive, minimally invasive posterior fixation […]
LAUNCHES
SeaSpine Announces the Launch of the Shoreline RT™ Cervical Interbody Implant System, featuring Reef Topography™
CARLSBAD, Calif., Aug. 01, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch and completion of the first surgery for its Shoreline RT Cervical Interbody Implant System. Shoreline RT represents the first in a […]
Nanovis Announces Commercial Launch of New Nanosurface Technology on Spinal Interbody Implants
CARMEL, IND. (PRWEB) AUGUST 01, 2019–Nanovis today announced the commercial launch of its bioceramic nanotube enhanced FortiCore interbodies following a successful alpha launch. The FortiCore interbodies are designed with a unique, proprietary, patent protected bio-ceramic enhanced titanium nanotube surface. The nanotubes are applied to a deeply porous, fully interconnected titanium scaffold intermolded with a PEEK […]
SeaSpine Announces Launch of Mariner® Outrigger® Revision System
CARLSBAD, Calif., July 31, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today the limited commercial launch of the Mariner Outrigger Revision System. Mariner Outrigger is an implant system providing surgeons a high level of versatility for multiple […]
HD LifeSciences Launches New Implant Footprints at State of Spine Surgery Meeting
BOSTON (PRWEB) JULY 30, 2019–HD Lifesciences launched 3 new implant footprints at the State of Spine Surgery Meeting in Aruba last month, including a Hyperlordotic TLIF, Hyperlordotic ALIF, and Curved TLIF. This expands the Soft Titanium® portfolio to 14 product lines and accommodates a variety of surgeon preferences and patient anatomies for PLIF, TLIF, ALIF, […]
Alphatec Announces Commercial Launch of the InVictus™ MIS and Open Spinal Fixation Systems
CARLSBAD, Calif., July 23, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of InVictus™, a comprehensive spinal fixation platform designed to address a range of pathologies through a minimally invasive (MIS), open, or […]
Alphatec Announces Commercial Launch of ALIF IdentiTi™ Porous Titanium Interbody System Implant
CARLSBAD, Calif., July 18, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of its IdentiTi Large Window Porous Titanium Interbody Implant System for anterior lumbar interbody fusion (ALIF) procedures. “The spine market’s new organic innovation […]
SeaSpine Announces Full Commercial Launch of Regatta® Lateral System
CARLSBAD, Calif., July 11, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the full commercial launch of the Regatta® Lateral System, featuring NanoMetalene®technology. Regatta is a comprehensive, minimally invasive lateral interbody fusion system designed to efficiently and reproducibly […]
Globus Medical Launches First Expandable Interspinous Fixation – AERIAL™
AUDUBON, Pa., May 30, 2019 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE: GMED), a leading musculoskeletal solutions company, today announced the launch of AERIAL™, a minimally invasive expandable interspinous fixation system with independent locking plates. The expandable central core of AERIAL™ provides continuous distraction for indirect decompression and a customized patient fit. This is Globus […]
Alphatec Announces Commercial Launch of TLIF IdentiTi™ Posterior Curved Porous Titanium Interbody Implant System
CARLSBAD, Calif., May 30, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of its IdentiTi-PC Porous Titanium Interbody Implant System for Transforaminal Lumbar Interbody Fusion Procedures (TLIF). “The commercial launch of IdentiTi-PC demonstrates […]
Silony Medical presents new 3D printed ACIF:Oyster
With OYSTER, Silony Medical presents a 3D-printed ACIF (Anterior Cervical Interbody Fusion) cage that combines the advantages of titanium and PEEK in one cage. OYSTER is available from May 2019. The 3D-printed OYSTER ACIF cage is offered sterile packed in heights from 4 to 8 mm in 1 mm increments, dome-shaped and wedge-shaped, in three […]
CoreLink Introduces Comprehensive Lateral Access and Interbody Fusion Platform
ST. LOUIS–(BUSINESS WIRE)–CoreLink Surgical, LLC, today announced the full commercial launch of its new Lateral Access System, as well as the first of several highly anticipated implant systems for lateral fusion – the F3D™ and CL5™ Lateral interbody fusion devices. Jay Bartling, CoreLink’s CEO stated, “The commercial launch of our lateral access platform, combined with […]
Globus Medical Launches FORTIFY Variable Angle Expandable Corpectomy Spacer System
AUDUBON, Pa., April 15, 2019 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the launch of FORTIFY VA™, a variable angle corpectomy solution, to add to its large and growing Expandable Technology portfolio. Globus Medical is the industry leader in expandable technology with over 20 expandable implant options backed […]
Alphatec Announces Commercial Launch of its First IdentiTi™ Porous Titanium Interbody System
CARLSBAD, Calif., March 28, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of its new IdentiTi-C Porous Titanium Interbody Implant System for Anterior Cervical Discectomy and Fusion (ACDF). The launch of the IdentiTi-C […]
Globus Medical launch the AERIAL™ Interspinous Fixation System
Globus Medical is excited to announce the launch of the AERIAL™ Interspinous Fixation System. AERIAL™’s easy insertion and expansion provides a simple, minimally invasive solution for interspinous fixation. AERIAL™ Interspinous Fixation is a minimally invasive spinous process fixation system. With its expandable core and independent locking plates, AERIAL™ offers a customized patient fit and allows for indirect […]
Atlas Spine Inc. completes alpha phase and announces the full launch of its first-to-market HiJak™ AC Expandable Cervical IBFD
JUPITER, FL, Feb. 27, 2019— Atlas Spine Inc., a spinal implant company based in Jupiter Florida, announced today the successful completion of its 50th surgical procedure and over 100 devices implanted with its new HiJak AC Expandable Cervical Inter-body Fusion Device (IBFD). The company has now moved to its full product launch. The HiJak AC […]
Alphatec Announces FDA Clearance of its Automated SafeOp Neuromonitoring System to Address Significant Unmet Needs in Spine Surgery
CARLSBAD, Calif., Feb. 25, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC) announced today that it has received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for its automated SafeOp neuromonitoring system for use in real-time intraoperative nerve location and health assessment. “I could not be more excited […]
ulrich medical USA® Announces Market Entry and First Global Implantation of New Generation Expandable Implant Technology
ST. LOUIS, Jan. 11, 2019 /PRNewswire/ — ulrich medical USA, Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States, announced today the market entry of a highly-anticipated, new generation vertebral body replacement device which is the company’s flagship technology in the U.S. spine implant market. The Solidity […]
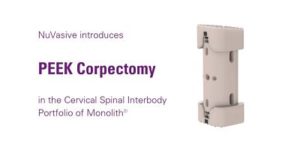
NuVasive Grows Cervical Spinal Interbody Portfolio With PEEK Corpectomy Implant
SAN DIEGO, Dec. 10, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for expanded use of its Monolith®Corpectomy System, providing surgeons with a modular PEEK interbody solution for […]
Nexxt Spine Launches SAXXONY® Posterior Cervical Thoracic Spine System
NOBLESVILLE, Ind.–(BUSINESS WIRE)–Nexxt Spine LLC, a medical device company focused on designing, manufacturing, and distributing innovative spinal solutions, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) and full commercial launch of its SAXXONY® Posterior Cervical Thoracic System—a system designed to stabilize cervical (C1 to C7) and thoracic (T1 to T3) spinal […]
Nvision Biomedical Technologies Launches Pedicle Screw System at NASS
Wednesday, September 26, 2018–LOS ANGELES – Nvision Biomedical Technologies, a San Antonio-based medical device and biologics manufacturer, debuted its focus® Pedicle Screw System today at the North American Spine Society (NASS) Annual Meeting, held at the Los Angeles Convention Center, September 26-29. The Nvision focus Pedicle Screw System, which received clearance from the U.S. Food […]
Medtronic Launches the Infinity(TM) OCT Spinal System
DUBLIN and LOS ANGELES – September 25, 2018 – Medtronic plc (NYSE:MDT) today announced the U.S. launch of the Infinity(TM) Occipitocervical-Upper Thoracic (OCT) System designed to simplify posterior cervical spine surgery. The Infinity OCT System is a complete procedural solution that integrates navigation and biologics with Medtronic’s comprehensive devices and instrumentation to create efficiency in fusion […]
Biedermann Motech to launch iMAS 360™ procedure at NASS
Miami, FL – September 24, 2018 / Business Wire/ – Biedermann Motech will launch the iMAS 360™procedure at NASS 2018 in Los Angeles, September 26-28, 2018. Biedermann Motech, the developer of the first polyaxial pedicle screw (MOSS System) worldwide and pioneer in the spinal market for more than 30 years, announced today that it will […]
Captiva Spine Adds Cervical Cages to TirboLOX™ Dual Layer Lattice Structure 3D Printed Titanium Cage Offering
JUPITER, FLA. (PRWEB) SEPTEMBER 21, 2018–Captiva Spine is a medical device organization located in Jupiter, Florida, dedicated to delivering smart, elegant and intuitive spinal fusion solutions. Today, Captiva Spine is announcing it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its TirboLOX-C 3D Printed Titanium Cervical Cages. TirboLOX-C Titanium […]
BioMedGPS Launches SmartTRAK Robotic and Computer Assisted Surgery – Spine Module
IRVINE, CALIF. (PRWEB) SEPTEMBER 18, 2018–BioMedGPS announces expanded coverage of SmartTRAK Business Intelligence in the realm of Robotic and Computer Assisted Surgery with the launch of its first module, US Computer Assisted Surgery (CAS) – Spine at the upcoming NASS conference later this month. A second SmartTRAK module, US Computer Assisted Surgery (CAS) – Ortho, will […]
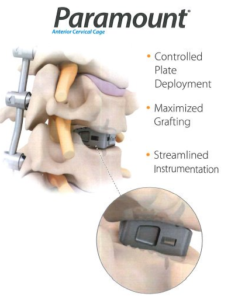
Spine Wave Announces the Commercial Launch of the Paramount® Anterior Cervical Cage
SHELTON, Conn., Sept. 17, 2018 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce the commercial launch of the Paramount® Anterior Cervical Cage. The Paramount® Anterior Cervical Cage is a titanium anterior cervical implant with integrated fixation blades. The patented system offers controlled, less invasive blade deployment and maximized bone grafting. The Paramount® Anterior Cervical […]
DePuy Synthes Launches CONCORDE LIFT Expandable Interbody Implant as Part of a New Procedural Solution for Minimally Invasive Spine Surgery (MIS) that Enhances Surgical Efficiency
RAYNHAM, Mass., July 26, 2018 /PRNewswire/ — Johnson & Johnson Medical Devices Companies* today announced that DePuy Synthes** is launching the CONCORDE LIFT Expandable Interbody Device in the U.S. The CONCORDE LIFT Implant is designed to treat patients suffering from degenerative disc disease as part of the new offering called UNLEASH MIS TLIF (Transforaminal Lumbar […]