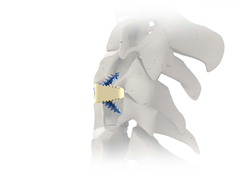
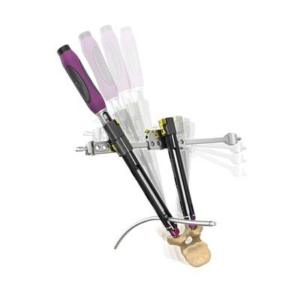
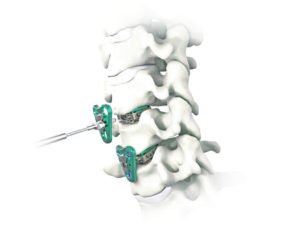
CARLSBAD, Calif., Aug. 11, 2020 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch of the Explorer™ TO System. Explorer TO represents SeaSpine’s first new product offering since 2017 in the estimated $400 million expandable interbody […]
LAUNCHES
LES Spine Innovations announces launch of the new A-CIFT SoloFuse HYPER-LORDOTIC Implant offerings
BOSTON (PRWEB) AUGUST 08, 2020 LES Spine Innovations and Sagitechnology are proud to announce the launch of the new A-CIFT SoloFuse Hyperlordotic Cervical Standalone Interbody System, the first ever HA impregnated standalone cervical cage for greater sagittal correction. The A-CIFT SoloFuse Hyperlordotic Interbody is a Less Exposure Surgery (LES®) technology. LES technologies are designed with outpatient surgery in […]
ulrich medical USA® Brings Strong Momentum® to U.S. Product Launch
ST. LOUIS, July 13, 2020 /PRNewswire/ — ulrich medical USA, Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States announced the nationwide commercial release of the Momentum Posterior Spinal Fixation System. “After completing hundreds of procedures during the alpha launch, Momentum has already made its mark with an enthusiastic reception and positive reviews from our alpha surgeon […]
SeaSpine®Announces Full Commercial Launch of Mariner® MIS and Mariner Outrigger™ Spinal Fixation Systems
CARLSBAD, CA (June 30, 2020) – SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the full commercial launches of Mariner MIS and Mariner Outrigger. “The commercial launches of Mariner® MIS and Mariner Outrigger™ result from meticulous product refinement and creative application of differentiated […]
SeaSpine® Announces Limited Commercial Launches of NorthStar™ OCT and Cervical Facet Fusion™ Systems
CARLSBAD, CA (June 22, 2020) – SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launches and completion of initial surgeries of both its NorthStar OCT and Cervical Facet Fusion systems, which significantly expand its procedural offerings for posterior […]
NuVasive Expands Complex Spine Portfolio with Global Launch of Reline 3D for Pediatric Deformity
SAN DIEGO, June 18, 2020 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced the expansion of its complex spine portfolio with the global commercial availability of Reline 3D™, a posterior fixation system for patients suffering from pediatric spinal deformities. The Reline […]
Spinal Elements® Introduces its MIS Ultra™ Platform of Products and Procedures Designed to Minimize the Unintended Consequences of Spine Surgery
Carlsbad, CA – June 17, 2020 – Spinal Elements, a Carlsbad, CA-based medical device company focused on spine surgery procedures, today introduced its MIS Ultra™ platform of products and procedures. MIS Ultra is a suite of minimally invasive integrated instrument and implantable device solutions designed to optimize spinal fixation and restoration while minimizing the unintended […]
Zavation Medical Products, LLC, Launches Cortical Screw
JACKSON, Miss., June 16, 2020 /PRNewswire/ — Zavation Medical Products (“Zavation” or the “Company”), an innovative designer and manufacturer of high-quality spinal implants, instruments, MIS procedural kits, and biologics headquartered in Flowood, MS, announced the launch of the Cortical Screw, a screw designed for use in the cortical bone as an extension of the Zavation Spinal System. The Cortical […]
OrthoPediatrics Corp. Commences Initial U.S. Launch of ApiFix’s FDA-Approved Spinal Deformity Correction System
WARSAW, Ind., June 10, 2020 (GLOBE NEWSWIRE) — OrthoPediatrics Corp. (“OrthoPediatrics” or the “Company”) (Nasdaq:KIDS), a company focused exclusively on advancing the field of pediatric orthopedics, is pleased to announce the initial U.S. launch of the ApiFix Minimally Invasive Deformity Correction (“ApiFix”) system. Additionally, the Company anticipates approximately 20 leading clinical centers in the United States to […]
Zavation Medical Products, LLC. Launches eZspand™ Expandable Cage
JACKSON, Miss., May 26, 2020 /PRNewswire/ — Zavation Medical Products (“Zavation” or the “Company”), an innovative designer and manufacturer of high-quality spinal implants, instruments, MIS procedural kits, and biologics headquartered in Flowood, MS, announced the launch of eZspand™, an expandable lumbar interbody fusion device. eZspand™, the latest addition to Zavation’s portfolio, features unmatched expandable precision paired with continual expansion […]
Camber Spine Technologies Announces Nationwide Launch of SPIRA®-C Integrated and FORTICO™ Anterior Cervical Plate
KING OF PRUSSIA, Pa., May 7, 2020 /PRNewswire/ — Camber Spine, a leading innovator in spine and medical technologies, today announced the FDA clearance and nationwide launch for two novel anterior cervical products: The SPIRA®– C Integrated Interbody system, a stand-alone integrated fixation system, and the FORTICO™ Anterior Cervical Plating System, a two screw plating system intended for […]
Genesys Spine is pleased to announce the launch of a Sacroiliac Joint Fusion System.
Thursday, February 20, 2020–Austin, TX: The Genesys Spine Sacroiliac Joint Fusion System consists of partially threaded and fully threaded implants designed to secure the sacroiliac joint and minimize micro-motion enabling bony fusion. Advantages of the Genesys Spine Sacroiliac Joint Fusion System include: Dual thread designs incorporate a differential pitch for controlled compression across the joint. […]
Atlas Spine Launches its V3 Guided Segmental Cervical Plating System, Expands Solutions for Cervical Deformity
Palm Beach County, FL, January 28, 2020 — Atlas Spine Inc., a spinal implant company based in Jupiter, Florida, announced today the launch of its V3 Guided Segmental Plating System, expanding the company’s disruptive technology solutions for treating complex deformity and degenerative conditions of the cervical spine. Cervical spine pathologies afflict hundreds of thousands of people each year. The general growth of an aging population […]
SpineCraft announces the launch of the ASTRA Occipito-Cervico-Thoracic (OCT) Spine System
January 24, 2020 –The ASTRA-OCT System is a comprehensive set of implants and instruments to stabilize the spine in patients undergoing posterior cervical fusion. The system was developed to address unmet needs in complex posterior spine fixation procedures, revisions and extensions. ASTRA-OCT includes a wide range of screw, connector and rod options for both the […]
SeaSpine Announces Full Commercial Launch of Mariner® Midline Posterior Fixation System
CARLSBAD, Calif., Dec. 30, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the full commercial launch of the Mariner® Midline Posterior Fixation System. Mariner Midline is a comprehensive, less invasive posterior fixation system built upon the Mariner […]
EOS imaging Announces the Upcoming Launch of EOSedge™, Its New Generation Imaging System
PARIS–(BUSINESS WIRE)–EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI – Eligible PEA – PME), a leader in 2D/3D orthopedic medical imaging and software solutions for 3D anatomical modeling and surgical planning, today announced the launch of its new generation imaging system, EOSedge, in Europe, Canada and Australia. The Company will unveil its new system at the […]
New Cervical Spine System from DePuy Synthes Advances Treatment Options for Patients with Complex Cervical Spine Disorders
NEW YORK, Nov. 21, 2019 /PRNewswire/ — The Johnson & Johnson Medical Devices Companies* today announced that DePuy Synthes has launched the SYMPHONY Occipito-Cervico-Thoracic (OCT) System, expanding its offering for the surgical treatment of conditions in the neck and upper back. The SYMPHONY System includes a differentiated offering of instruments and implants designed for stabilization […]
4WEB Medical Announces Launch of Stand Alone Cervical Spine Truss System
NEW YORK, Nov. 20, 2019 /PRNewswire/ — 4WEB Medical, an orthopedic device company focused on developing innovative implants utilizing its proprietary Truss Implant Technology™, announced the initial launch and completion of the first procedures with the Cervical Spine Truss System-Stand Alone™ (CSTS-SA). The CSTS-SA implants incorporate an Advanced Structural Design comprised of the company’s proprietary […]
Stryker receives FDA clearance for SAHARA® Lateral 3D Expandable Interbody System
LEESBURG, Virginia, Oct. 31, 2019 /PRNewswire/ — Stryker announced today the U.S. Food and Drug Administration (FDA) 510(k) clearance of its SAHARA Lateral 3D Expandable Interbody System featuring Lamellar 3D Titanium Technology, which will be on display at the Society for Minimally Invasive Spine Surgery (SMISS) Annual Forum this week in Las Vegas, NV. SAHARA Lateral is the first ever […]
CarboFix Extends Its CarboClear® Carbon Fiber Spinal Product Line With Its FDA Cleared Vertebral Body Replacement (VBR) System
DOVER, Delaware, Oct. 17, 2019 /PRNewswire/ — CarboFix has announced today that the U.S. Food and Drug Administration (FDA) has cleared its CarboClear® Carbon Fiber Vertebral Body Replacement (VBR) System to replace a collapsed, damaged, or unstable vertebral body due to tumor or trauma. The CarboClear® VBR System joins the FDA cleared CarboClear® Pedicle Screw and CarboClear® Fenestrated Pedicle Screw Systems. […]
CTL Amedica to unveil MONET™ Integrated ACIF Cage System during NASS Sept. 25 – 28
DALLAS (PRWEB) SEPTEMBER 25, 2019–CTL Amedica Corporation will officially launch its MONET™ ACIF Integrated Cage System with Supplementary Fixation during the North American Spine Society (NASS) annual meeting and exhibition Sept. 25-28, 2019. NASS will be held at McCormick Place, West Building, located at 2301 S. King Drive in Chicago, IL. CTL Amedica representatives will […]
DePuy Synthes Expands Comprehensive Offering to Treat Degenerative Spine Disease with Launch of 3D Printed Implant Portfolio for Spine Surgery
CHICAGO, Sept. 25, 2019 /PRNewswire/ — The Johnson & Johnson Medical Devices Companies* today announced that DePuy Synthes has launched the CONDUIT Interbody Platform with EIT Cellular Titanium Technology, further expanding its comprehensive offering to treat degenerative spine disease. The innovative portfolio, which includes 3D printed titanium interbody implants for spinal fusion surgery is designed […]
Alphatec Announces Commercial Launch of IdentiTi™ TLIF Posterior Oblique Porous Titanium Interbody Implant System
CARLSBAD, Calif., Sept. 25, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of its IdentiTi TLIF Posterior Oblique Titanium Interbody Implant System (“IdentiTi PO”) for transforaminal lumbar interbody fusion procedures. The launch of […]
RTI Surgical® Announces Commercial Launch of the CervAlign® Anterior Cervical Plate System
DEERFIELD, Ill., Sept. 24, 2019 (GLOBE NEWSWIRE) — RTI Surgical Holdings, Inc. (Nasdaq: RTIX), a global surgical implant company, today announced the full commercial launch of the CervAlign® Anterior Cervical Plate (ACP) System to add to the growth of its Established Therapies spine portfolio. The CervAlign ACP System is designed to promote cervical fusion by providing […]
CoreLink Introduces Oro Lateral Plate System Designed for Optimum Versatility to Treat Patients Suffering From Spinal Conditions
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a leading designer and manufacturer of spinal implant systems, today announced the introduction of the Oro™ Lateral Plate System designed for optimum versatility to treat patients suffering from degenerative disk disease, spondylolisthesis, trauma, or spinal deformities. The CoreLink Oro Lateral Plate System has received first of its kind FDA clearance as […]
Biedermann Motech to Introduce MOSS VRS® – The Next Generation Pedicle Screw Technology™ to the US Market
MIAMI & DONAUESCHINGEN, Germany–(BUSINESS WIRE)–Biedermann Motech, the company that developed the world’s first polyaxial pedicle screw (MOSS® System) and pioneer in the spinal market for more than 30 years, today announced the further expansion of its product portfolio of advanced solutions for spinal surgery with the introduction of the MOSS VRS® Pedicle Screw System at […]
Camber Spine Announces Natiowide Launch of SPIRA®-O Open Matrix OLIF at NASS 2019
KING OF PRUSSIA, PA., September 12, 2019 – Camber Spine, a leading innovator in spine and medical device technologies, announced the launch of its first spinal implant designed and manufactured specifically for the oblique lumbar interbody fusion (OLIF) spine surgical procedure. This implant represents the fifth product in its distinctive, rapidly growing SPIRA® device family. […]