SHELTON, CT–(Marketwired – October 24, 2016) – Spine Wave, Inc. is committed to the commercialization of high-quality, innovative medical devices for the treatment of spinal disorders. Spine Wave is pleased to announce that it will be fully launching the Velocity® P Expandable Interbody Device system in mid-November with the arrival of commercial-scale inventory quantities. The […]
LAUNCHES
DeGen Medical Launches Latitude-C™ Porous Ti Cervical Interbody Spacer
FLORENCE, SC–(Marketwired – October 24, 2016) – DeGen Medical introduces Latitude-C™ Porous Ti Cervical Interbody Spacer, an extension of our popular and effective Latitude-C™ product line, is designed to consider the uncinate process of the cervical vertebra. The lateral angled shape is a shift from traditional rectangular shape and mimics patient anatomy. The variety of […]
SpineGuard® expands “PediGuard®” franchise, will launch “PediGuard Threaded” drilling device at North American Spine Society (NASS) annual meeting
PARIS & SAN FRANCISCO: SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD) announced today that it will launch its next-generation PediGuard “Threaded” device enabled by DSG™ (Dynamic Surgical Guidance) technology at next week’s 31st annual meeting of the North American Spine Society (NASS) in Boston on October 26-29. The innovative PediGuard Threaded device with DSG is a drilling instrument […]
Spineology Completes Limited Release for Elite™ Expandable Interbody Fusion System
MINNEAPOLIS & ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology is excited to announce the successful completion of the Elite™ Expandable Interbody Fusion System limited release. The Elite Expanding Interbody Implant is inserted into the disc space in a contracted minimal height to minimize dissection and neural retraction. It is then incrementally expanded to restore the anatomical height of […]
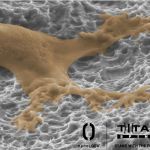
Titan Spine Initiates Full U.S. Launch of New nanoLOCK® Surface Technology
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced that it has expanded the distribution of its line of Endoskeleton®titanium implants featuring the company’s new proprietary nanoLOCK® surface technology nationwide. The full U.S. launch follows the successful alpha introduction of the nanoLOCK® technology initiated […]
Stryker’s Spine Division Announces Launch of New LITe® BIO Delivery System
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division today announced the launch of its LITe BIO Delivery System, a hand-held device used to facilitate delivery of bone graft material to spinal surgery sites, which simplifies graft delivery, accommodates a surgeon’s preferred graft materials,1 and allows for direct visualization of graft placement. The unique design of the LITe BIO […]
icotec Brands BlackArmor® as an Alternative Material to Metal in Loadbearing Musculoskeletal Surgical Procedures
ALTSTAETTEN, Switzerland, /PRNewswire/ –icotec AG, an innovative medical device company leading the industry in the development and manufacturing of non-metallic Carbon/PEEK composite medical devices, announces the brand name of BlackArmor® for its Carbon/PEEK material, which boosts a 15 year and 20,000 implantation clinical track record in spinal and fracture surgical care. BlackArmor is a combination […]
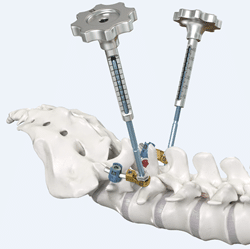
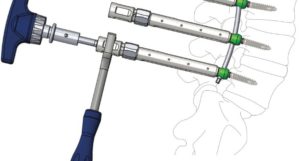
Amendia Launches New SYZYGY™ Spine Stabilization System
MARIETTA, GA (PRWEB) SEPTEMBER 27, 2016–Balanced spinal solutions provider Amendia Inc. is pleased to announce the commercial launch of its innovative Spondylolisthesis reduction system:, the Syzygy™ Stabilization System. Configured to provide immobilization and stabilization of spinal segments as an adjunct to fusion, Syzygy is ideal for use in the treatment of specific acute and chronic […]
joimax® Announces Release of First Endoscopic AMA CPT Code
IRVINE, Calif.–(BUSINESS WIRE)–The AMA (American Medical Association) released the CPT 2017 Codebook (Current Procedural Terminology), now containing the first endoscopic spinal surgery code no. 62380. This new code covers endoscopic decompression of the spinal cord, nerve root(s), including laminotomy, partial facetectomy, foraminotomy, discectomy and/or excision of herniated intervertebral disc, 1 interspace, lumbar, and will be […]
ulrich medical USA® Announces Product Line Extension, Releases uCerv® Titanium Cervical Interbody System
ST. LOUIS, Sept. 26, 2016 /PRNewswire/ — ulrich medical USA, Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States, today announced the U.S. market release of uCerv titanium spinal implants as an adjunct to its current uCerv PEEK OPTIMA® Interbody System which is an existing product used […]
G Surgical Announces FDA Clearance and Launch of the MARKSMAN MIS® Spine System
Boulder, CO (September 2, 2016)–G Surgical, a privately held medical device company offering innovative spinal implants, announced that it has received 510(k) clearance from the United States Food and Drug Administration to market their MARKSMAN MIS® Spine System. Don Grafton, President and CEO of G Surgical, comments, “We are excited to strengthen our portfolio by […]