BOCA RATON, Fla.--(BUSINESS WIRE)--SurGenTec, LLC, a leader in innovative spine and orthopedic technologies, today announced that the U.S. Food and Drug Administration (FDA) has granted 510(k) SurGenTec, LLC, a leader in innovative spine and orthopedic technologies, today announced that the U.S. Food and Drug Administration (FDA) has granted … [Read More...] about SurGenTec Secures FDA Clearance for ION-C™ – Now Navigation Compatible
Main Content
FEATURED NEWS
BREAKING NEWS

Ventris Medical Announces the Award of Additional Patent Claims for its Novel Backpack Bone Graft Containment System
NEWPORT BEACH, Calif., May 27, 2026 /PRNewswire/ -- Ventris Medical has secured additional patent coverage for its innovative Backpack bone graft containment system, reinforcing the company's commitment to advancing surgical solutions. This latest patent coverage underscores the unique design and functional advantages of the Backpack system, a novel product … [Read More...] about Ventris Medical Announces the Award of Additional Patent Claims for its Novel Backpack Bone Graft Containment System

VB Spine Signs Master Agreement with Apolo Medica
NEW YORK--(BUSINESS WIRE)--VB Spine LLC (“VB Spine”), a global, family-owned spine company, today announced that it has signed a Master Service Agreement with Apolo Medica to begin distribution of VB Spine products in Mexico. The announcement follows VB Spine’s recent expansion into the Netherlands, Luxembourg, Belgium, Australia, and New Zealand. “We are thrilled to further expand VB Spine’s … [Read More...] about VB Spine Signs Master Agreement with Apolo Medica

DePuy Synthes Announces Agreement to Acquire Miniature Radiofrequency Tracking Technology Across its Joint Reconstruction Portfolio
Agreement will bring first-to-market capabilities to eliminate line-of-sight dependency designed to improve simplicity and surgical flow RAYNHAM, Mass.--(BUSINESS WIRE)--DePuy Synthes, a global leader in orthopaedic technologies and solutions, today announced a definitive agreement to acquire selective rights to develop, manufacture and commercialize Gemtrack technology across its Joints … [Read More...] about DePuy Synthes Announces Agreement to Acquire Miniature Radiofrequency Tracking Technology Across its Joint Reconstruction Portfolio
SpineGuard obtains three new patents for full protection in the U.S. of its innovative solutions integrating DSG technology
PARIS and BOULDER (CO), May 26, 2026 – 06:00 pm CEST - SpineGuard (FR0011464452 - ALSGD), an innovative company that deploys its DSG® (Dynamic Surgical Guidance) local conductivity sensing technology to secure and streamline the placement of bone implants, today announces that three new U.S. patent applications have received notices of allowance from the USPTO (United States Patent and … [Read More...] about SpineGuard obtains three new patents for full protection in the U.S. of its innovative solutions integrating DSG technology

DePuy Synthes Enters Exclusive U.S., Canada and Australia Distribution Agreement for CGBIO’s NOVOSIS
Partnership strengthens DePuy Synthes’ portfolio with a differentiated bone graft substitute containing growth factors to address unmet need across orthopaedic specialties RAYNHAM, Mass.--(BUSINESS WIRE)--DePuy Synthes, a global leader in orthopaedic technologies and solutions, today announced an exclusive distribution agreement1 with CGBIO, a biotechnology company specializing in regenerative … [Read More...] about DePuy Synthes Enters Exclusive U.S., Canada and Australia Distribution Agreement for CGBIO’s NOVOSIS
Sanctions, Titanium and Pedicle Screws: How Russia’s Spine Market Is Rebuilding Itself
Russia was never a transparent spine market. Large enough to matter, technically capable in selected centers, quietly dependent on imported premium technology. That was the working summary most international companies operated on before 2022. The picture has shifted since. Outside visibility has dropped, domestic industrial pressure has risen, and import substitution, long discussed in policy … [Read More...] about Sanctions, Titanium and Pedicle Screws: How Russia’s Spine Market Is Rebuilding Itself

aljeel Medical Partners as Distributor for DePuy Synthes by Johnson & Johnson in Saudi Arabia
aljeel Medical has entered into a strategic distribution agreement with Johnson and Johnson Medical Saudi Arabia, becoming the orthopedics distributor for DePuy Synthes products in Saudi Arabia. The agreement is intended to improve access for healthcare providers to advanced, high-quality medical solutions, supported by an efficient and coordinated distribution network. The collaboration brings … [Read More...] about aljeel Medical Partners as Distributor for DePuy Synthes by Johnson & Johnson in Saudi Arabia

Hospital Access Is Gone. What Comes Next for Spine Field Sales?
What happens when the environment that built a generation of elite field reps no longer exists — and a new generation is building careers without ever knowing what they missed? There's a particular kind of clarity that comes from watching an industry transform from the inside. Veterans of spine sales describe an era when hospital access was assumed, relationships were built over years of OR … [Read More...] about Hospital Access Is Gone. What Comes Next for Spine Field Sales?

REALISTS Officially Launches in the United States
DELAWARE, OH, UNITED STATES, May 14, 2026 /EINPresswire.com/ -- REALISTS Training Technologies AG, a Germany-based company focused on spine surgery simulation and surgical training, today announced its official launch in the United States. The expansion includes the formation of a new U.S. subsidiary, REALISTS USA, Inc., and the appointment of Greg Rhinehart as Vice President of Strategic … [Read More...] about REALISTS Officially Launches in the United States

Amplify Surgical and Emory Healthcare Complete World’s First Continuously Navigated Endoscopic TLIF Using the dualPortal & dualX Platform
Amplify Surgical announced the successful completion of what is believed to be the world's first continuously navigated endoscopic transforaminal lumbar interbody fusion (TLIF) procedure, performed at Emory Healthcare by Dr. Daniel Refai and his surgical team. The procedure represents a significant advancement in minimally invasive spine surgery, combining real-time navigation with endoscopic … [Read More...] about Amplify Surgical and Emory Healthcare Complete World’s First Continuously Navigated Endoscopic TLIF Using the dualPortal & dualX Platform
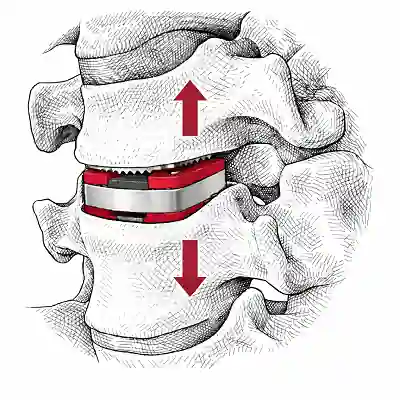
Expandable Cervical Cages: Innovation or Just a Niche Tool? | Updated May 2026
Anterior cervical discectomy and fusion (ACDF) is one of the most standardized procedures in spine surgery. The workflow is familiar: decompression, controlled distraction, sizing, and insertion of a static interbody cage. For decades, this has worked reliably and predictably. So when expandable cages appeared in the cervical spine, the reaction was immediate and reasonable: What problem are … [Read More...] about Expandable Cervical Cages: Innovation or Just a Niche Tool? | Updated May 2026