FDA authorization marks critical milestone enabling advancement of next-generation bone regeneration therapy for patients with degenerative spinal diseases FREDERICK, Md., April 30, 2026 /PRNewswire/ -- Theradaptive, Inc., a clinical-stage biotechnology company pioneering targeted protein therapeutics for tissue regeneration, … [Read More...] about Theradaptive Secures FDA Approval to Advance OASIS Trial to Pivotal Study Evaluating OsteoAdapt® SP in Spinal Fusion
Main Content
FEATURED NEWS
BREAKING NEWS

Demetra acquires Maxxspine Innovative GmbH, a company that has been a leader in advanced spinal solutions for over twenty years, further strengthening its position as a benchmark in spine surgery
MILAN, April 30, 2026 /PRNewswire/ -- Demetra, an innovative and independent international medical company leader providing bone cements, spacers and biomaterials for orthopedic, spine and neuro applications and orthopedic infection control, announces the successful completion of the acquisition of Maxxspine Innovative GmbH, a German-based company specialized in advanced spinal … [Read More...] about Demetra acquires Maxxspine Innovative GmbH, a company that has been a leader in advanced spinal solutions for over twenty years, further strengthening its position as a benchmark in spine surgery

VB Spine Announces VB Spine Solutions, a Partnership-Driven Program to Help Hospitals Acquire Technology
New partnership-driven approach expands access to spine technology NEW YORK--(BUSINESS WIRE)--VB Spine LLC (“VB Spine”), today launched VB Spine Solutions, a program designed to bring critical spine technology to hospitals faster, with structures built around patient need. While focused on spine, VB Spine Solutions aligns with hospitals to simplify how capital equipment is placed, so … [Read More...] about VB Spine Announces VB Spine Solutions, a Partnership-Driven Program to Help Hospitals Acquire Technology
Taiwan’s Spine Industry: Why a Small Market Could Become a Serious Export Hub
The Taiwanese spine market is relatively small, it has a sophisticated healthcare system, broad access to care and strong medical infrastructure. However, reimbursement discipline and price pressure remain important realities. For spine companies, this creates a demanding environment: surgeons may be open to advanced technologies, but hospitals remain highly sensitive to cost, procurement value … [Read More...] about Taiwan’s Spine Industry: Why a Small Market Could Become a Serious Export Hub

Amnovis expands to the United States and acquires Westconn Precision Technologies’ additive manufacturing activities
Aarschot, Belgium - April 29, 2026--Amnovis, a Belgian additive manufacturing contract manufacturing organization serving regulated industries including medical devices, announces the establishment of a new operational entity in the United States and the acquisition of the additive manufacturing activities of Westconn Precision Technologies. Together, these steps represent a key milestone in … [Read More...] about Amnovis expands to the United States and acquires Westconn Precision Technologies’ additive manufacturing activities

Life Spine Announces Expanded Indications for ARx® SAI Spinal Fixation System
HUNTLEY, Ill.--(BUSINESS WIRE)--Life Spine, Inc., a medical device company that designs, develops, manufactures, and markets products for the surgical treatment of spinal disorders, announced today their 510k for ARx SAI (Sacral Alar Iliac) Spinal Fixation System was approved. ARx SAI has expanded its compatibility clearance and is now approved for use with compatible pedicle screws with 5.5mm or … [Read More...] about Life Spine Announces Expanded Indications for ARx® SAI Spinal Fixation System

Official launch of three New PediGuard® Models during the Annual Scoliosis Meeting in China
PARIS and BOULDER (CO), April 28, 2026 – 06:00pm CEST - SpineGuard (FR0011464452 - ALSGD), an innovative company that deploys its DSG® (Dynamic Surgical Guidance) local conductivity sensing technology to secure and streamline the placement of bone implants, today announced the official launch of the newly cleared Curved and XS PediGuard models, in Nanjing (China), during … [Read More...] about Official launch of three New PediGuard® Models during the Annual Scoliosis Meeting in China
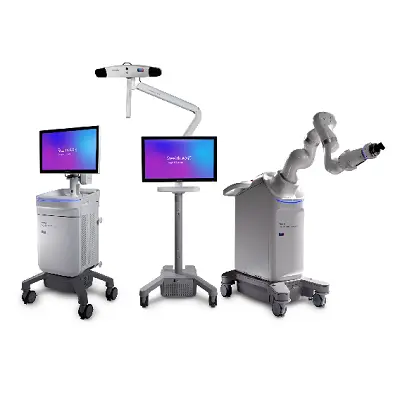
Medtronic announces CE Mark for Stealth AXiS™ surgical system
Following recent FDA clearances, Medtronic is accelerating access to integrated planning, navigation, and robotics platform across Europe GALWAY, Ireland, April 28, 2026 /PRNewswire/ -- Medtronic, a global leader in healthcare technology, has received CE mark for the Stealth AXiS™ surgical system, a next-generation platform that brings planning, navigation, and robotics together … [Read More...] about Medtronic announces CE Mark for Stealth AXiS™ surgical system
Do Interspinous Spacers Still Have a Place in Spine Surgery? | Updated 2026
Last week, we published the news that Amplio Spine, LLC had signed a Letter of Intent to acquire the FDA-cleared KeyLift™ Expandable Interlaminar Stabilization System from FloSpine, LLC. This announcement brings interspinous and interlaminar implants back into focus. For that reason, we have updated our overview of this product category and its market, looking not only at KeyLift™ itself, but also … [Read More...] about Do Interspinous Spacers Still Have a Place in Spine Surgery? | Updated 2026

Cerapedics Announces Supplier Agreement with Vizient for PearlMatrix® P-15 Peptide Enhanced Bone Graft
Agreement expands access to the Cerapedics portfolio of peptide-powered drug-device products for spinal fusion procedures WESTMINSTER, Colo., April 24, 2026 /PRNewswire/ -- Cerapedics Inc., a global, commercial-stage orthopedics company dedicated to redefining the path to bone repair, today announced it has entered into a supplier agreement with Vizient, the nation's largest … [Read More...] about Cerapedics Announces Supplier Agreement with Vizient for PearlMatrix® P-15 Peptide Enhanced Bone Graft

ulrich medical increases revenue by 10 percent in 2025 and repeatedly grows above the industry average
The ulrich medical Group's strategy of increasingly positioning itself internationally has also paid off in 2025: With sales growth of 10 percent, ulrich medical significantly exceeds the average of the German medical technology industry. According to the BVMed Autumn Survey 2025*, the weighted sales growth of the branch was only 3.1 percent domestically and 5.5 percent worldwide.Several growth … [Read More...] about ulrich medical increases revenue by 10 percent in 2025 and repeatedly grows above the industry average

FDA-cleared. Quietly adopted. Nationally ready. KeyLift™ is about to get loud
Amplio Spine, LLC signs Letter of Intent to acquire the FDA–cleared KeyLift™ Expandable Interlaminar Stabilization System (K232484) — comprising KeyLiftXS™ and KeyLiftXL™, the only minimally invasive platform in the United States offering both XS and XL sizing across two platforms and eight sizes, indicated for lumbar spinal stenosis, degenerative disc disease, and other non–cervical … [Read More...] about FDA-cleared. Quietly adopted. Nationally ready. KeyLift™ is about to get loud