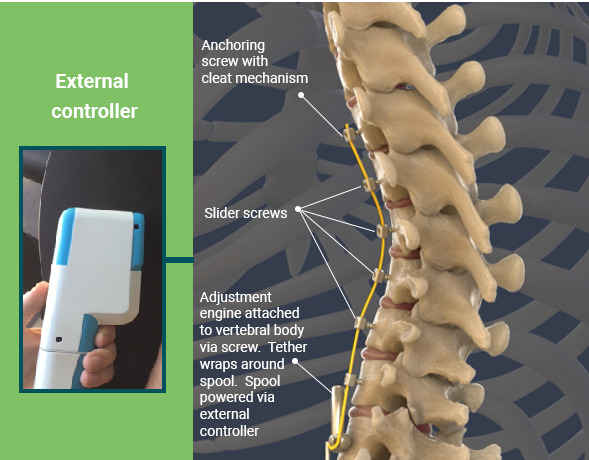
Auctus Surgical, a company devoted to non-fusion options for scoliosis patients, is developing a one-of-a-kind vertebral body tethering (VBT) system to treat pediatric scoliosis with a non-fusion, dynamic approach. The company is announcing the recent Breakthrough Device Designation by the FDA, a recognition that the Auctus dynamic vertebral body tethering system is unique and will meet a clear clinical need.
Benefits:
- Dynamic tension
- Complete non-surgical adjustment
- Most effective in making gradual non-surgical adjustments over time
- Prevents over-correction and can act as internal brace
About Auctus
Auctus is developing a dynamic vertebral body tethering system to treat pediatric scoliosis with an adjustable non-fusion system.https://auctussurgical.com
