First-quarter 2026 revenue
- High order volume
- Slower-than-expected normalization of supply chains
- 2026: growth trajectory confirmed
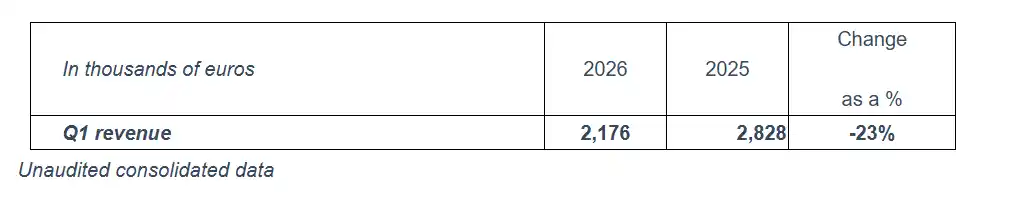
Spineway, a specialist in innovative implants for the treatment of severe spine disorders, recorded revenue of €2.2 million in the first quarter of 2026.
Following strong growth in the fourth quarter of 2025 (+38%), the slowdown observed in the first quarter of 2026 will not undermine the Group’s momentum. It is mainly attributable to delays in regulatory approvals in certain regions and supply delays from subcontractors facing increased demand. These challenges have been identified, contained, and are being resolved.
- International export sales (excluding Europe) were mixed:
- In Asia, business remains solid, with revenue up 3.7% and with growth prospects in Vietnam and Australia.
- In the Middle East, business is at a temporary standstill against adverse geopolitical backdrop.
- In Latin America, revenue came in €0.4 million below the previous year, impacted by a slower-than-anticipated ramp-up in deliveries. The Group continues to expand its distribution network in this high-potential region (particularly in Mexico and Brazil), with a view to achieving critical mass.
- In France, the domestic market remains resilient, with stable revenue supported by prioritized inventory management while in Europe revenue was slightly down.
In a structurally favorable healthcare environment, Spineway continues to expand, notably through the intensification of its multi-year regulatory approval program. With a robust order backlog, the Group expects the lag between orders and revenue to gradually normalize from the second half of 2026 onwards. This will help offset the quarterly volatility associated with the current growth phase.
Next event:
July 30, 2026 – H1 2026 revenue and results
About Spineway
Spineway designs, manufactures and markets innovative implants and surgical instruments for treating severe disorders of the spinal column.
Spineway has an international network of over 50 independent distributors and more than 70% of its revenue comes from exports.
Spineway is eligible for the PEA-PME (equity savings plans for SMEs)
Listing venue: Euronext Growth Paris
ISIN: FR001400N2P2
Ticker: ALSPW
Find out all about Spineway at www.spineway.com
Contacts:
Spineway
Shareholder-services line
Available Tuesday through Thursday (10 a.m.-12 midday)
+33 (0)806 706 060
Aelium
Investor relations
Isabelle Desmottes
spineway@aelium.fr
This press release has been prepared in both English and French. In case of discrepancies, the French version shall prevail.
