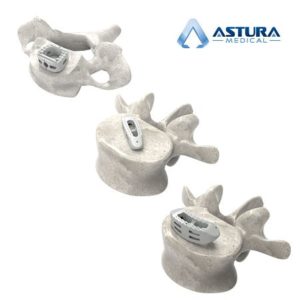
CARLSBAD, CA – June 4, 2018 – Astura Medical, a high growth, innovative spine technology company, today announced that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its ALTA ACDF Interbody Spacers and HALF DOME Lumbar Interbody Spacers in PEEK-OPTIMAä HA Enhanced from Invibio Biomaterial Solutions. Combining the clinically proven […]
NEWS
Aesculap Implant Systems Announces U.S. Military’s TRICARE Now Covers Lumbar Total Disc Replacement
CENTER VALLEY, Pa., June 4, 2018 /PRNewswire/ — Aesculap Implant Systems, LLC announced today that TRICARE now covers the company’s activL® Artificial Disc for active duty and retired military members and their families in need of Lumbar Total Disc Replacement (CPT® procedure code 22857). Aesculap is a privately-held spine and orthopaedic implant company and a […]
IMPLANET Announces Opening of a United Kingdom Sales Branch
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–Regulatory News:IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, announces the opening of a branch in the United Kingdom, accelerating expansion and direct growth in the growing adult degenerative market. The Implanet UK branch […]
Spineology Rampart One™ Standard ALIF Interbody Fusion System Gains FDA Clearance for Stand-Alone Use
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., announces FDA clearance of its Rampart One™ Standard ALIF device allowing it to be used with or without supplemental fixation. “Following the success of our beta launch, this stand-alone clearance will provide additional momentum as we prepare for the full market release of the Rampart One ALIF Interbody Fusion System,” […]
Surgeons Preferred HD LifeSciences™ Hive™ Based on Radiographic Visualization
(Ilsa Webeck)–In the last few years, companies have been launching new and improved interbody fusion devices produced using advanced manufacturing technologies, including 3D printing. Commercially available devices are marketed “to actively participate in the fusion process”, to “create a favorable environment for cell attachment and proliferation,” and be an “active participant in the fusion process.” […]
Camber Spine Announces 510(k) Approval Of ENZA™-A Titanium ALIF
WAYNE, Pa., June 4, 2018 /PRNewswire/ — Camber Spine today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its ENZA™-A Titanium Anterior Lumbar Interbody Fusion (ALIF) system, a unique, minimally invasive interbody fusion device providing integrated fixation. Daniel Pontecorvo, CEO, said, “ENZA™-A leverages two of Camber […]
SPINE Companies Catalogue From A to Z: 25 Spine Companies to know with “A”
Some years ago, we started to publish what we called SPINE Catalogue because it included most of the Spinal companies worldwide. The objective was to promote and build awareness of most of the market player even the smallest ones. Last year we accounted for almost 400 companies from many different countries. Now, it is time […]
Did an Orthopedic Surgeon accidentally shoot a Sales Rep?
(ryortho.com)–In Cayce, South Carolina a 46-year old orthopedic surgeon has been accused of fatally shooting William Holland, a sales rep employed by CrossLink Orthopaedics, one of Stryker’s largest distributors.According to the police initially, this was a tragic, horrible gun accident.In their first recounting of events, the police said that the surgeon, Adam Lazzarini, M.D., had […]
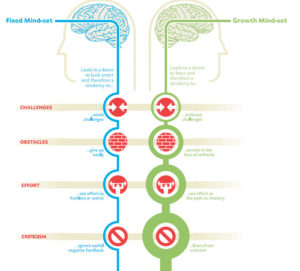
Here’s why your attitude is more important than your intelligence
When it comes to success, it’s easy to think that people blessed with brains are inevitably going to leave the rest of us in the dust. But new research from Stanford University will change your mind (and your attitude). Psychologist Carol Dweck has spent her entire career studying attitude and performance, and her latest study […]
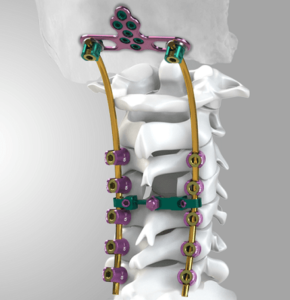
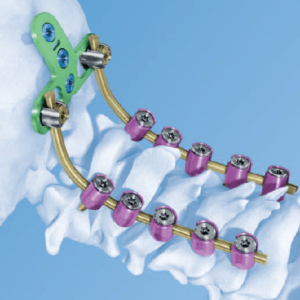
Is the Posterior Cervical Fusion Market Attractive? (Final Part) 30 Systems to Know!
Posterior Cervical fusion technique involves the fusion of more than two vertebrae to prevent unwanted motion of the discs or the compression of posterior spaces between the cervical vertebrae. Posterior Cervical Fusion is a procedure intended for the stabilization of the cervical spine through a posterior approach. The procedure is commonly performed using hooks, plates, […]
iOrthopedics Inc. (iOI), Las Vegas, Nevada, is pleased to announce patent awards and allowances for its Universally Expanding Cage (UEC)
iOrthopedics Inc. (iOI), Las Vegas, Nevada, is pleased to announce patent awards and allowances for its Universally Expanding Cage (UEC) giving exclusive rights in the spine expandable interbody arena. Validation is granted by the U.S. Patent Office (US9,861,494 and US9,999,515) with confirmed claims covering a device and method for independently altering the distance and angle […]
Safe Orthopaedics acquires a direct sales force in the United Kingdom
Eragny-sur-Oise, France, June 1st, 2018 at 7:30am CEST – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company specialising in the design and marketing of single-use implants and instruments facilitating the minimally invasive treatment of spinal fracture conditions, is today announcing a deal to acquire the sales force of UK company Qspine, which specialises in distributing medical […]
SpineFrontier Receives FDA Approval for A-CIFT SoloFuse HA Standalone System
BOSTON (PRWEB) MAY 31, 2018–SpineFrontier has received FDA approval for A-CIFT SoloFuse HA, its next wave of fusion technology, designed and developed with intention by their team of engineers. As the first company to receive FDA clearance on HA PEEK in the U.S., SpineFrontier produced the A-CIFT SoloFuse HA standalone system to leverage the familiarity […]
Medicrea Receives World’s First and Only FDA-Clearance to Market Patient-Specific Cages
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea® Group (Euronext Growth Paris: FR0004178572 – ALMED), pioneering the convergence of healthcare IT and next-generation, outcome-centered spinal device design with UNiD ASI™ (Adaptive Spine Intelligence) technology, announced today that it has obtained the first and only 510(k) clearance from the U.S. Food & Drug Administration (FDA) to market […]
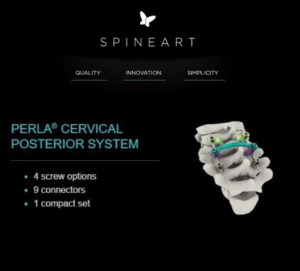
Spineart is pleased to announce it has attained the CE Mark for the PERLA® Posterior Cervical System.
Spineart is pleased to announce it has attained the CE Mark for the PERLA® Posterior Cervical System. Perla was designed according to Spineart’s philosophy: Quality, Innovation, Simplicity. The system offers a comprehensive range of implants including 4 types of screws, several versatile rod connectors and transition rods. Intuitive instruments are housed in a single, compact […]
K2M Receives FDA Clearance for BACS® Patient-Specific Module
LEESBURG, Va., May 30, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for BACS® Patient-Specific devices. With the BACS Surgical Planner, surgeons can […]
Titan Spine ‘turns it to eleven’
(BIZTIMES.COM)–With a proprietary surface technology and a runway of private equity investment, Titan Spine LLC has, to borrow a phrase from the cult classic film “This is Spinal Tap,” turned its growth up to 11.The Mequon-based medical device developer was established after surgeon Dr. Peter Ullrich came up with the idea to make interbody spinal […]
Posterior Cervical Fusion Market (Part2) 10 More Systems to Know!
The posterior cervical fusion is a type of cervical fusion surgery, that accounts the 15% to the cervical devices market share. Although it is still an small segment of the overall Spinal market, it is growing due to the increasing prevalence of osteoarthritis and rheumatoid and the rising aging population. The estimated market value of the posterior […]
In’Tech Medical enters into agreement to acquire Bradshaw Medical, industry leader in silicone overmolded solutions, ratchet and torque devices
Memphis, Tenn. May 23, 2018–In’Tech Medical SAS (http://www.intech-medical.com), the leading Contract Manufacturer of medical devices in Orthopedics, announced today that it has entered into a binding agreement to acquire world renowned instruments and silicone overmold company, Bradshaw Medical Inc. The transaction, supported by Eurazeo PME, has been approved by the Board of Directors of both organizations. Based in […]
Spinal Elements® Announces Expanded Commercial Release of its Clutch® Interspinous Process Device
CARLSBAD, Calif.–(BUSINESS WIRE)–Spinal Elements, a spine technology company, today announced the release of its Clutch interspinous process device. This new product further enhances the breadth of Spinal Elements’ thoracolumbar portfolio and offers surgeons more options for treatment of various posterior thoracolumbar pathologies. Clutch has been in use since shortly after its FDA 510(k) clearance in […]
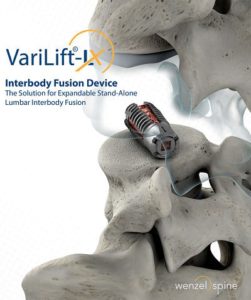
Wenzel Spine Announces Publication of Case Series Detailing Endoscopic Transforaminal Decompression and Fusion Technique with VariLift-LX
AUSTIN, Texas–(BUSINESS WIRE)–Wenzel Spine, Inc., a medical technology company focused on providing minimally invasive solutions for the treatment of spinal disorders, today announced a 24 Consecutive Patient Case Series detailing an endoscopic, stand-alone TLIF surgical technique used with the VariLift-LX System has been published in the Journal of Spine, Volume 7, Issue 2. The Case […]
Medacta International Introduces New Approach to Posterior Lumbar Midline Cortical Screw Placement with Customizable MySpine MC Surgical Guides
CASTEL SAN PIETRO, Switzerland–(BUSINESS WIRE)–Medacta International, the family-owned global leader in innovative joint replacement and spinal technologies, announced clearance from the U.S. Food and Drug Administration (FDA) and the addition of MySpine Midline Cortical Pedicle Screw Placement Guide (MySpine MC) to its MySpine portfolio. The first two stateside surgeries utilizing MySpine MC were conducted this […]
Nuvasive SpineTRACK Registry Reaches Significant Enrollment Milestone Of 10,000 Patients
NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that the Company’s SpineTRACK Registry has reached a significant enrollment milestone of 10,000 patients from more than 50 surgeons in the United States since its inception in 2011. This milestone illustrates not only the continued […]
IMPLANET Announces the Launch of JAZZ Evo® for the Promising Market of Degenerative Spine Disease in Adults
BORDEAUX, France & BOSTON–IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, announces the launch of JAZZ Evo, a new evolution of the JAZZ® implant developed to respond to the constraints of vertebral fusion indications in adults. Developed primarily […]
Zimmer Biomet Announces 510(k) Clearance for Zyston® Strut Open Titanium Interbody Spacer System
WARSAW, Ind., May 22, 2018 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Zyston® Strut Open Titanium Interbody Spacer System. This marks Zimmer Biomet’s first titanium spinal implant manufactured via […]
Life Spine Announces Line Extension of PRO-LINK® Ti Stand-Alone Cervical Spacer System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that the U.S. Food and Drug Administration (FDA) has given 510(k) marketing clearance to the PRO-LINK Ti Stand-Alone Cervical Spacer System for spinal fusions. PRO-LINK featuring Life Spines OSSEO-LOC™ surface treatment […]
NuVasive PRECICE STRYDE™ System Used In First Patient For Stature Lengthening By International Limb Lengthening Expert
SAN DIEGO, May 22, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the new PRECICE STRYDE™ system from NuVasive Specialized Orthopedics™ (NSO) was used in the first set of patients by internationally recognized limb lengthening and reconstruction expert Dr. Dror Paley. Built on […]