If you’ve made the decision that it’s time for a new job and have started to reach out to friends about possible openings. It’s nearly inevitable that you’ll soon be asked, “Have you reached out to recruiters?” Ironically, although the question seems straightforward, it’s a tough one to answer. I’ll admit, before entering the career […]
NEWS
When to Hire a Headhunter? 50 Headhunters to Know
Looking for a job can be difficult. But finding the perfect and most qualified person to fill the job in your company can be even more challenging. Hiring the right people is vital to a company’s success; you’ll need a pool of experienced and trained individuals to handle the hiring process and look for people […]
Europe spinal implants market: The Top 20 European Manufacturers Of Spinal Implants (Part III)
The European spine surgery devices market is expected to reach $3.46 billion by 2021. The spinal fusion & fixation is the largest segment in the European spine surgery devices market, with a share of 66.5% in terms of value. It is projected to grow at a CAGR of 6.2% during the forecast period. While a […]
SeaSpine Reports First Quarter 2018 Financial Results
CARLSBAD, Calif., May 03, 2018 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ:SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today financial results for the quarter ended March 31, 2018. Summary First Quarter 2018 Financial Results and Recent Accomplishments Revenue of $33.2 million, an increase of 4.0% year-over-year […]
SpineGuard Reinforces the Utility of Its Dynamic Surgical Guidance (DSG®) Technology Platform by Receiving a Second US Patent for “Bone Fusion Monitoring” Application
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News: SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, announced today it has been granted a second patent by the US Patent Office for the application of its Dynamic Surgical […]
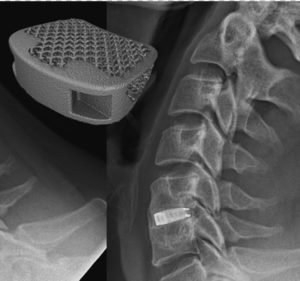
EIT Cellular Titanium® Scaffold Provides Better Primary Stability, Less Subsidence and Similar Fusion Results to PEEK in Combination with Autograft
(PRWEB) MAY 03, 2018–Emerging Implant Technologies GmbH (EIT), a German medical device manufacturer exclusively focused on creating innovative technologies for spinal application by utilizing additive manufacturing, announces that the results of the EFFECT trial have been presented at the SSA 2018 meeting in Adelaide. The EFFECT Trial is a prospective controlled trial, evaluating the clinical […]
RTI Surgical Announces First Quarter 2018 Results
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (Nasdaq:RTIX), a global surgical implant company, reported operating results for the first quarter of 2018. “2018 is off to a positive start with the acquisition of Zyga at the very beginning of the year and a solid financial performance this quarter,” said Camille Farhat, chief executive officer. “The continued strength […]
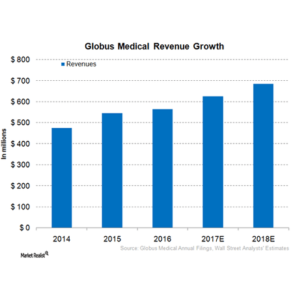
Globus Medical Reports First Quarter 2018 Results
AUDUBON, Pa., May 02, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced its financial results for the first quarter ended March 31, 2018. Worldwide sales were $174.4 million, an increase of 11.9% as reported, or 10.8% in constant currency First quarter net income was $39.5 million, or 22.7% of […]
Medtronic Announces Advanced SynergyTLIF(SM) Workflow for Minimally Invasive Lumbar Spine Surgery
DUBLIN and NEW ORLEANS – May 2, 2018 – Medtronic plc (NYSE: MDT) today announced the advanced SynergyTLIFSM workflow – a procedural solution for spine surgery that combines innovative technologies to create a completely navigated minimally invasive procedure that allows fewer intra-operative surgical steps. The announcement was made during the American Association of Neurological Surgeons (AANS) annual […]
Life Spine Announces the Launch of SIJointFixation.com with Key Focus on Surgeon and Patient Education
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today the launch of a new website, www.sijointfixation.com. This newly designed website offers quick and easy access to essential information on Life Spine’s Micro-Invasive SI Joint Fixation procedural solutions such as; SImpact™ and […]
Paradigm Spine Receives FDA Pre-Market Approval (PMA) For First Of Its Kind Disposable Instrument Kit
NEW YORK, May 2, 2018 /PRNewswire/ — Paradigm Spine, LLC, a leader in providing motion preservation solutions for the treatment of lumbar spinal stenosis, today announced that the U.S. Food and Drug Administration (FDA) has granted pre-market supplemental approval (PMA) for its coflex® Interlaminar Stabilization® disposable instrument kit. This marks the first approved disposable spinal instrument set for a […]
K2M Group Holdings, Inc. Reports First Quarter 2018 Financial Results and Updates Fiscal Year 2018 Outlook
LEESBURG, Va., May 01, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (Nasdaq:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today reported financial results for its first quarter ended March 31, 2018. First Quarter 2018 Financial Summary: Total first quarter revenue […]
Implanet Announces the First Peer-Reviewed Publication of JAZZ® Results in Complex Adult Spine Surgery
BORDEAUX, France & BOSTON–Implanet (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, is announcing the publication of a prospective study of JAZZ in complex adult deformity in the American Association of Neurological Surgeons’ Journal of Neurosurgery: Spine (JNS). These results were […]
Medtronic Announces TiONIC(TM) Technology, a Titanium 3D Printed Platform for Spine Surgery Implants
DUBLIN and NEW ORLEANS – May 1, 2018 – Medtronic plc (NYSE: MDT) today announced the launch of a titanium 3D printed platform – TiONIC(TM) Technology – that enables more complex designs and integrated surface technologies for spine surgery implants. The announcement was made during the American Association of Neurological Surgeons (AANS) annual meeting in New […]
joimax® Launches Its Latest Interlaminar Endoscopic System, iLESSYS® Pro, and New 4K Camera Light Source and Documentation Command System, Camsource® LED and Vitegra® at AANS in New Orleans, LA and GSC in Singapore
IRVINE, Calif.–(BUSINESS WIRE)–joimax® again launches new product innovations at the upcoming American Association of Neurological Surgeons (AANS) taking place in New Orleans from April 28 – May 2, and at the Global Spine Congress (GSC) in Singapore, starting May 2. The German based market leader of technologies and training methods for minimally invasive endoscopic spinal surgery […]
ChoiceSpine™ Launches Tiger Shark,TM a Next-Generation Porous Interbody System at AANS 2018
Knoxville, Tenn-May 01, 2018– ChoiceSpine LP, a privately-held spinal device manufacturer based in Knoxville, TN, announced today at the American Association of Neurological Surgeons’ Annual Scientific Meeting the commercial launch of Tiger Shark, a 3D manufactured, titanium alloy interbody device created with a proprietary, organic, porous structure called BioBond. With the aide of the BioBond […]
Orthofix Reports First Quarter 2018 Financial Results
LEWISVILLE, Texas–(BUSINESS WIRE)–Orthofix International N.V. (NASDAQ:OFIX) today reported its financial results for the first quarter ended March 31, 2018. Net sales were $108.7 million, diluted earnings per share from continuing operations was $0.27 and adjusted earnings per share from continuing operations was $0.39. The Company also closed the acquisition of Spinal Kinetics Inc., a privately […]
Paradigm Spine Highlights Data Presented At ISASS 2018 Further Validating Decompression With coflex
NEW YORK, April 30, 2018 /PRNewswire/ — Paradigm Spine, LLC, a leader in providing motion preservation solutions for the treatment of lumbar spinal stenosis, today announced results of three subanalyses from studies of coflex® that further validate the efficacy and benefit to patients with lumbar spinal stenosis. The data were presented during three scientific podium presentations at the 2018 […]
OrtoWay to publish scientific abstract on first use of OrtoWell® Distractor tool in a lateral corpectomy at ISASS18 Spinal Meeting in Toronto.
OrtoWay, the Stockholm-based medical technology company, will present a scientific abstract at ISASS18 in Toronto, Ontario, Canada on April 11, 2018, documenting the clinical use of its OrtoWell® device for the first time in a lateral, minimally invasive surgical (MIS) fixation procedure. The CE-marked device, which is now approved for most anterior-approach surgeries, was used […]
Medtronic Announces FDA Approval of Infuse(TM) Bone Graft in New Spine Surgery Indications Using PEEK Interbody Implants
DUBLIN – April 30, 2018 – Medtronic plc (NYSE: MDT) today announced U.S. Food and Drug Administration (FDA) approval of Infuse(TM) Bone Graft in new spine surgery indications. InfuseBone Graft is now approved for use with additional spine implants made of polyetheretherketone (PEEK) in oblique lateral interbody fusion (OLIF 25(TM) and OLIF 51(TM)) and anterior lumbar […]
Meditech Spine expands its lumbar plating options by adding the CURE LP iView and QMax to its growing portfolio of spinal implants
ATLANTA, April 30, 2018 /PRNewswire/ — Meditech Spine, LLC, announced today it has received additional 510(k) clearance for its CURE Lumbar Plating (LP) System from the US Food and Drug Administration (FDA). The CURE LP iView and Cure LP QMax System clearance compliments Meditech’s recent 510(k) of the CURE Lumbar Plate System in 2017 with additional plating […]
Globus Medical: Innovating Beyond The Spine Market
(SEEKINGALPHA.COM) –The orthopedic implants industry is relatively fragmented among ten large competitors who operate in the space, with some like Globus Medical (GMED), specializing in certain products such as minimally invasive spine implants. Despite Globus Medical’s recent success, it remains a relatively small player in the orthopedic implants market. Stryker (SYK), Zimmer-Biomet (ZBH), and Medtronic […]
Europe spinal implants market: The Top 20 European Manufacturers Of Spinal Implants (Part II)
The European spine surgery devices market is expected to reach $3.46 billion by 2021. The spinal fusion & fixation is the largest segment in the European spine surgery devices market, with a share of 66.5% in terms of value. It is projected to grow at a CAGR of 6.2% during the forecast period. Factors driving […]
Which are the 7 Relevant Fixation Band Systems?
Recent literature has shown clinical benefits of posteromedial translation using sublaminar bands, for treatment of AIS. Adolescent idiopathic scoliosis (AIS) is a tridimensional deformity that affects the trunk height changing both in coronal and sagittal profiles, with usually a loss of thoracic kyphosis and lumbar lordosis and a trend to cervical kyphosis. The aim of surgical […]
Stryker to Highlight Expanding Line of 3D-Printed Tritanium® Cages at AANS
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division will showcase its expanding line of Tritanium® cages, including the new Tritanium® TL Curved Posterior Lumbar Cage, a 3D-printed interbody fusion cage intended for use as an aid in lumbar fixation, at the American Association of Neurological Surgeons (AANS) Annual Scientific Meeting, April 28–May 2, 2018, in New Orleans (booth No. 413). Tritanium […]
HD LifeSciences Exhibiting New Lumbar Interbody Configurations at AANS Annual Scientific Meeting 2018
HD LifeSciences’ Soft Titanium™ lattice technology will be exhibited at the 2018 AANS Annual Meeting in New Orleans. HD LifeScience’s booth will feature their latest Hive interbodies, including the new Long TLIF, Wide ALIF and Hyperlodotic ALIF configurations. Lucas Diehl, CEO of HD LifeSciences states, “We are tremendously excited to expand our lumbar interbody portfolio, and apply our […]
Safe Orthopaedics’ full-year 2017 results and first-quarter 2018 revenues
Eragny-sur-Oise, France, April 27, 2018 at 7:30am CEST – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company specialized in the design and marketing of single-use implants and instruments improving the minimally invasive treatment of spinal fracture conditions, is today reporting its full-year 2017 results and its Q1 2018 revenues. Safe Orthopaedics’ 2017 annual financial report will be […]