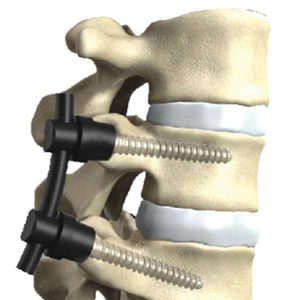
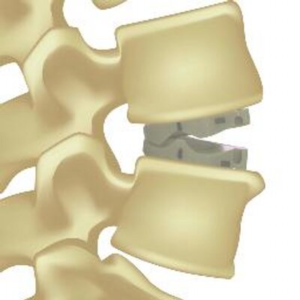
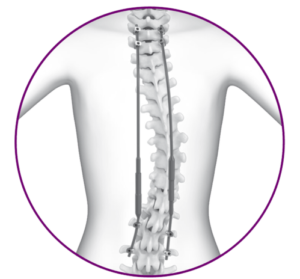
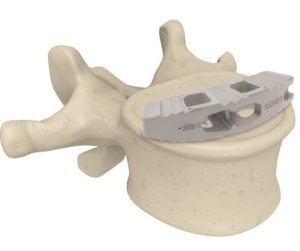
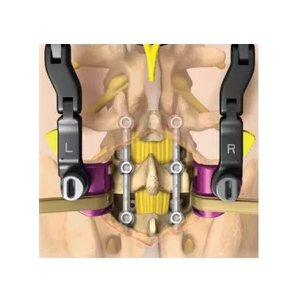
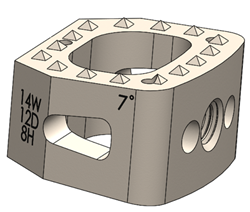
RAYNHAM, Mass., July 26, 2018 /PRNewswire/ — Johnson & Johnson Medical Devices Companies* today announced that DePuy Synthes** is launching the CONCORDE LIFT Expandable Interbody Device in the U.S. The CONCORDE LIFT Implant is designed to treat patients suffering from degenerative disc disease as part of the new offering called UNLEASH MIS TLIF (Transforaminal Lumbar […]
NEWS
CarboFix’s CarboClear®, Carbon Fiber Pedicle Screw System Has Been Cleared to Be Used in the United States by the FDA
HERZELIYA, Israel, July 26, 2018 /PRNewswire/ — CarboFix Orthopedics LTD., is pleased to announce that the U.S. Food and Drug Administration (FDA) has given 510(k) clearance for the CarboClear® Carbon Fiber Pedicle Screw System, a novel device to surgically treat oncological patients in the United States. The CarboClear Pedicle Screw System is intended to restore […]
Implanet Opens German Subsidiary
BORDEAUX, France & BOSTON–Regulatory News:Implanet (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, today announces its establishment in Germany. Following the opening of the Implanet UK sales branch in June 2018, Implanet continues direct operational expansion in the European […]
NeuroStructures Announces Distribution Agreement Awarded to Centinel Spine
NeuroStructures, Inc. announces that they have concluded a manufacturing and distribution agreement with Centinel Spine® for the Trifore™ Anterior Cervical Plating System. NeuroStructures, Inc. remains the manufacturer of record and Centinel Spine® owns the Trifore™ IP and will add it to their product portfolio under the name Fortos C™. The Trifore™ Anterior Cervical Plating System […]
Mahe Medical USA launches Spine Emergency Screw Removal system and makes it available via Amazon
Mahe Medical USA announced the introduction and availability of its universal spine emergency screw removal system. “Anything that can be done in the OR to help the patient through a procedure in a shorter period of time without impacting the outcome should be celebrated and that’s exactly what the Mahe spine emergency screw removal system […]
The Benefits of Contract Manufacturing | 46 Contract Manufacturers in Spine
Companies often choose contract manufacturing for a few primary reasons, all of which make it easier, less expensive and more efficient to bring new products to the market and distribute them broadly. Cost savings: Companies can achieve savings by contracting with a manufacturer that already has knowledge of the manufacturing process, has invested in the appropriate […]
FBC Device ApS Receives First FDA Clearance For Spinal Interbody Implant Providing Lordosis Adjustment from 9 to 21 Degrees to Aid Surgeons & Patients
AARHUS, Denmark–(BUSINESS WIRE)–FBC Device ApS, a medical device company focused on restoring natural alignment of the spine through innovative interbody implants, has received its first US FDA clearance for its technology, enabling access to the US market. Professor Finn Christensen, MD, PhD and founder and CEO of FBC Device says: “There is overwhelming evidence that […]
Titan Spine Appoints Peter Wehrly to Board of Directors
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, Inc., a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced the appointment of Peter Wehrly to its Board of Directors. Mr. Wehrly’s appointment, effective July 9, 2018, provides the Company with extensive medtech market knowledge and experience as it continues to drive improved patient […]
Medtronic, Medacta, Medicon, Medicrea and many others…! SPINE Catalogue From A to Z: 20 Spine Companies to know with “M”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
cultivate(MD) Funds Announce Investments Into Happe Spine
HOLLAND, Mich., July 19, 2018 /PRNewswire/ — Genesis Innovation Group, LLC, announces the formation of HAPPE Spine LLC as well as investments by cultivate(MD) Capital Fund I, LLC and cultivate(MD) Capital Fund II, LP into HAPPE Spine. Based on the work of Ryan K. Roeder, Ph.D. and researchers from the University of Notre Dame, HAPPE Spine has developed and will commercialize a […]
Nuvasive Launches MAGEC X For Early Onset Scoliosis Treatment
SAN DIEGO, July 19, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the U.S. launch of MAGEC X, a new procedural solution within the MAGEC product family that addresses Early Onset Scoliosis (EOS). MAGEC X features updates across all rod diameters […]
ChoiceSpine™ Expands Its US Western Region Sales Team
KNOXVILLE, TENN. (PRWEB) JULY 18, 2018–ChoiceSpine LP, a privately-held spinal device manufacturer based in Knoxville, TN, announced the hiring of a Western Regional Sales Director and Clinical Sales Manager expanding its sales team and emphasizing its commitment to growth. The two new additions to the sales team are: Rajeev Shrikhande as Western Regional Sales Director; and […]
Zavation Announces 510(k) Clearance of the Normandy VBR System (Cervical and Thoracolumbar Expandable Corpectomy Cage)
FLOWOOD, Miss., July 17, 2018 /PRNewswire/ — Zavation, an employee-owned medical device company that designs, develops, manufactures and distributes medical device products, announced today that it has received 510(k) clearance from the FDA to market Normandy VBR System (Cervical and Thoracolumbar Expandable Corpectomy Cage). For More Information on Zavation’s Complete Product Portfolio, Visit: http://zavation.com/. The Normandy VBR System is an […]
K2M Enhances CASCADIA™ Lateral 3D Interbody System Featuring Lamellar 3D Titanium Technology™ Following Clearances for CAYMAN® United Plate System
LEESBURG, Va., July 17, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced that it has received 510(k) clearance from the U.S. Food & Drug Administration (FDA) and a CE Mark for its CAYMAN® United […]
SPINE Companies Catalogue From A to Z: 7 Spine Companies to know with “L”
We are already building (week by week) the new SPINE Catalogue that will include most of the Spinal companies worldwide. We want to promote and build awareness of most of the market players, even the smallest ones. Today we present all the Spinal companies which name start with L including LDR, Life Spine and many […]
FDA Clears First Nanotechnology Peek Devices for Spinal Intervertebral Fusion
NASHUA, N.H.–(BUSINESS WIRE)–Vallum Corporation, a medical device company, today announced that it has received clearance from the U.S. Food and Drug Administration (FDA) to market a polyetheretherketone (PEEK) spinal interbody fusion device with a PEEKplus® nanotextured1 surface created by Vallum’s proprietary and patented Accelerated Neutral Atom Beam (ANAB) technology. PEEKplus® is the first and only FDA-cleared […]
Acron Medical Announces FDA 510(k) Clearance of its Signature PEEK TLIF Interbody System, the ACRON Interbody
ORLANDO (PRWEB) JULY 16, 2018–Acron Medical, LLC (http://www.Acron-Medical.com), part of the spineMED Group, is a spine technology organization, dedicated to developing and commercializing globally innovative spinal implants. The company is proud to announce that it has received 510(k) clearance from the FDA for its signature new technology, the ACRON TLIF Interbody system. “The ACRON Interbody […]
NuVasive Portfolio Introduces Low-Profile Modular Implants For Posterior Fixation
SAN DIEGO, July 16, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the launch of Reline MAS Midline (RMM), the next evolution of the Reline system that provides procedural versatility in a compact midline construct. RMM incorporates extremely […]
Global spine market growth 2018-2023. Updated: 150 Companies from “A to K”
Over the recent years, global spinal fusion market has been witnessing growth, on account of several driving factors including rising healthcare awareness amongst consumers, surging prevalence of spinal deformities due to accidents along with increasing health expenditure in developing countries. Moreover, ongoing demographic shift towards geriatric population with significant population suffering from inveterate spinal ailments, […]
Aziyo Partners with SurGenTec for Minimally Invasive Bone Graft Delivery
SILVER SPRING, Md.–(BUSINESS WIRE)–Aziyo Biologics, Inc., a fully integrated regenerative medicine company, announced today it has entered into a co-marketing agreement with SurGenTec, a privately-owned medical device company based in Boca Raton, FL. The Company will supply ViBone, its proprietary bone repair product, to SurGenTec for marketing and distribution with its patented GraftGun universal graft delivery […]
SPINE Companies Catalogue From A to Z: 6 Spine Companies to know with “J”
This week, we present the TENTH chapter (J) that includes all the Spinal companies which name start with J including Joimax among others.In that list you can find 6 Companies. Please visit the following page: https://thespinemarketgroup.com/category/spine-companies/page/12/ For any question please contact us: info@thespinemarketgroup.com or Spinemarketgroup@gmail.com
NuVasive Unveils Newest Addition To Lateral Procedural Solutions At IMAST 2018
SAN DIEGO, July 10, 2018 — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the Company will officially unveil its XLIF® Lordotic Expandable (XLX) Interbody System, which just received 510(k) clearance from the U.S. Food and Drug Administration (FDA), at the 25th International Meeting on Advanced Spine Techniques […]
SPINEWAY : Revenue for the first half of 2018
Ecully, 11 July 2018 Revenue for the first half of 2018 In thousands of euros 2017 2018* 1st quarter 3 091 1 834 2nd quarter 2 925 1 517 HY1 revenue 6 016 3 351 * Not audited Spineway, specialist in surgical implants and instruments for treating disorders of the spinal column (spine), closed the […]
Life Spine Announces the Limited Commercial Release and First Surgery of the CENTRIC®-T Pedicle-Based Retractor System.
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine Inc., a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today the Limited Commercial Release and first clinical usage of the CENTRIC-T Pedicle-Based Retractor System. As the newest addition to Life Spine’s CENTRIC Access Portfolio, the CENTRIC-T Pedicle-Based Retractor delivers micro-invasive access and foraminal […]
Globus Medical Features Latest Innovative Technologies at Scoliosis Research Society’s International Meeting on Advanced Spine Technologies
AUDUBON, Pa., July 11, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, will showcase its advanced technologies at the Scoliosis Research Society’s (SRS) 25th Annual International Meeting on Advanced Spine Techniques (IMAST) being held July 11-14, 2018, in Los Angeles, California. The company will also highlight its robotics platform technology through […]
SpineGuard posts H1 2018 revenue
PARIS & SAN FRANCISCO–Regulatory News:SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, announced today that its first half 2018 revenue was 3.6ME, a decrease of -5% at constant currency, and that its preliminary non audited […]
Cutting Edge Spine Announces FDA 510(K) Clearance of its New HA Enhanced PEEK Interbody for the Cervical Spine, the EVOL ha-C
WAXHAW, N.C. (PRWEB) JULY 11, 2018//Cutting Edge Spine (http://www.CuttingEdgeSpine.com), a US leader in the development and commercialization of bio-active technologies for the spine, today announced the 510(k) clearance and commercial launch of it’s latest innovation, the EVOL ha-C cervical inter-body system. The EVOL ha-C system is made of PEEK-OPTIMA HA Enhanced material supplied by Invibio […]