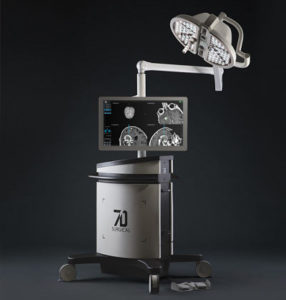
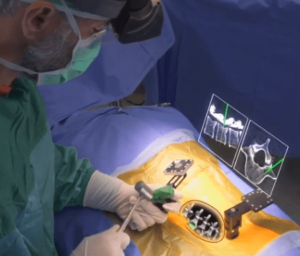
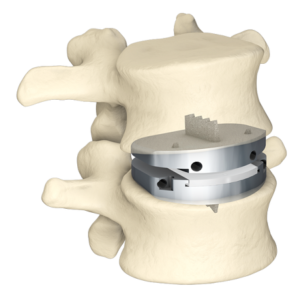
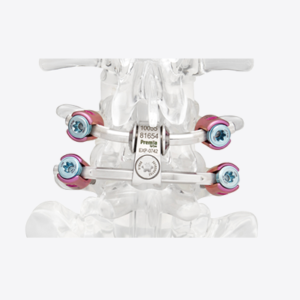
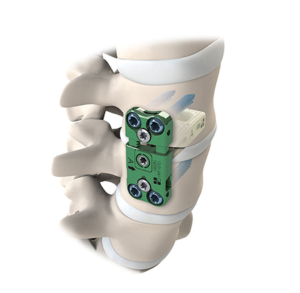
SALT LAKE CITY, Aug. 21, 2018 (GLOBE NEWSWIRE) — Amedica Corporation (NASDAQ: AMDA), an innovative biomaterial company that develops and commercializes silicon nitride for biomedical applications, is pleased to announce the successful completion of the first spine fusion surgery using the Valeo C+CSC with Lumen Interbody Fusion Device. The first surgery was performed by Dr. […]
NEWS
CoreLink, LLC Announces 510(k) Clearance for Foundation™ 3D Anterior Lumbar (ALIF)
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a vertically integrated manufacturer of spinal implant systems, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market The FoundationTM 3D Anterior Lumbar (ALIF) Interbody device. This marks the launch of the latest addition to CoreLink’s Foundation 3D Interbody Cage family. Jay Bartling, CEO, said, “The Foundation […]
NuVasive Appoints Dale Wolf To Lead Ohio Manufacturing Facility
SAN DIEGO, Aug. 16, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the appointment of Dale Wolf as vice president, Manufacturing, who will oversee the Company’s West Carrollton, Ohio manufacturing facility. Mr. Wolf will fill a newly created […]
China: Innovative Hybrid PEEK-titanium Expandable Cage for DLIF and OLIF Procedures Demonstrated
THORNTON CLEVELEYS, ENGLAND (PRWEB) AUGUST 14, 2018//The growing interest in PEEK-OPTIMA™ spinal implants in China received further impetus at the 11th Congress of the Chinese Association of Orthopaedic Surgeons (CAOS) held in partnership with the North American Spine Society (NASS). At this event, Fule Science & Technology Development, Beijing, and Invibio Biomaterial Solutions partnered to […]
Zavation Launches Facet Screw and Sacroiliac (SI) Screw Systems
FLOWOOD, Miss., Aug. 14, 2018 /PRNewswire/ — Zavation, an employee-owned medical device company that designs, develops, manufactures and distributes medical device products, announced today the launch of a fenestrated Facet Screw system and a Sacroiliac (SI) Screw system. For more information on Zavation’s complete product portfolio, visit http://zavation.com/. The Zavation Facet Screw system is a […]
7D Surgical Receives FDA Approval For Cranial Surgery
TORONTO, Aug. 13, 2018 /PRNewswire/ — 7D Surgical announced today that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Cranial Module. This achievement is a crucial step in the commercial launch of 7D Surgical’s innovative Machine-vision Image Guided Surgery (MvIGS) system for cranial surgery throughout the United States. […]
10 MORE Corpectomy Devices to Know!
Today we have included in our Corpectomy Section (CP) 10 more devices to know. We have included expandable devices as well as other differenciated corpectomy cages (Take into account that some of them may be already out of the market). As we said, there are more than 150,000 spinal fractures in North America* with an […]
NuVasive and Siemens Healthineers partner to transform spine surgery
San Diego and Erlangen, Germany, Aug. 9, 2018 / — NuVasive, Inc. (NASDAQ: NUVA) and Siemens Healthineers today announced a strategic partnership focused on technology development, marketing and commercial activities to advance clinical outcomes in minimally invasive spine surgery. NuVasive is an innovation leader in spine health technology, focused on transforming spine surgery with minimally disruptive, predictable and clinically reproducible procedurally-integrated solutions, […]
Augmedics Begins First-in-Human Clinical Trial of xvision-spine (XVS) Augmented-Reality Surgical Navigation System
YOKNEAM, Israel–(BUSINESS WIRE)–Augmedics has begun a first-in-human clinical trial of its xvision-spine (XVS) augmented-reality surgical navigation system at Sheba Tel Hashomer Medical Center and Asaf Harofeh Medical Center, in Israel. Led by Co-Principal Investigators Dr. Ran Harel and Prof. Yigal Mirovsky, the open label, prospective, single arm, multi-center study will evaluate the safety, performance, accuracy and […]
Centinel Spine Announces that UnitedHealthcare Will Now Cover prodisc L Anterior Lumbar Total Disc Replacement
New York, NY, – Centinel Spine, LLC is pleased to announce that UnitedHealthcare®, the nation’s second largest commercial insurer, has issued a positive medical policy for Lumbar Total Disc Replacement (TDR). This decision affects over 16 million patients covered through UnitedHealthcare, and means that three out of the four largest US commercial providers now have positive […]
SI-BONE, Inc. Announces 23 Commercial Health Insurance Plans Now “Exclusively” Cover Triangular iFuse Implant System® Based on Published Clinical Evidence
SAN JOSE, Calif., Aug. 7, 2018 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the minimally invasive surgical (MIS) treatment of the sacroiliac (SI) joint with the iFuse Implant System® (“iFuse”) announced that 23 commercial health plans have published exclusive positive coverage policies for the triangular iFuse Implant System. These exclusive positive coverage policies provide access to […]
Orthofix Reports Second Quarter 2018 Financial Results
LEWISVILLE, Texas–(BUSINESS WIRE)–Orthofix Medical Inc. (previously Orthofix International N.V.) (NASDAQ:OFIX) today reported its financial results for the second quarter ended June 30, 2018. Net sales were $111.5 million, diluted earnings per share from continuing operations was $0.05 and adjusted earnings per share from continuing operations was $0.42. “During the second quarter, we made excellent progress […]
10 Expandable Corpectomy Devices to Know!
Each year, there are more than 150,000 spinal fractures in North America*. There is an estimated 10,000 to 12,000 spinal cord injuries every year and approximately 39% of these injuries are cause by motor vehicle accidents. These non-elective or trauma surgeries sometimes require additional spinal structural support which is when the Expandable Corpectomy Device are […]
Fuse Medical, Inc. Completes Acquisition of Maxim Surgical
RICHARDSON, Texas–(BUSINESS WIRE)–Fuse Medical, Inc. (OTC: FZMD) (“Fuse” or the “Company”), announced the completion of the acquisition of Palm Springs Partners, LLC d/b/a Maxim Surgical (“Maxim”), a manufacturing company in the spinal fusion device market and full-service medical device and distribution company (the “Acquisition”). The effective date of Acquisition was August 1, 2018. Formed in […]
10 Spine Companies to Know with “P”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
Alphatec Reports Second Quarter 2018 Financial Results
CARLSBAD, Calif., Aug. 02, 2018 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, today reported financial results for the second quarter ended June 30, 2018. Second Quarter 2018 Financial Highlights […]
Newly Published Data Demonstrate Bone In-Growth Potential of Stryker’s 3D-Printed Tritanium® Cage
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division today announced the publication of a pre-clinical animal study comparing the performance of spinal implants made from a variety of materials, which illustrated the bone in-growth and biological fixation capabilities of its 3D-printed Tritanium cages. The study was published in the July issue of The Spine Journal. The purpose of […]
RTI Surgical® and Aziyo® Biologics Announce Exclusive Agreement to Distribute ViBone® for Spine and Other Orthopedic Procedures
ALACHUA, Fla. & SILVER SPRING, Md.–(BUSINESS WIRE)–RTI Surgical, Inc. (Nasdaq: RTIX), a global surgical implant company, and Aziyo Biologics, Inc., a fully integrated regenerative medicine company, today announced the signing of an agreement under which Aziyo will provide ViBone to RTI Surgical for exclusive distribution in the U.S. ViBone is a bone repair product designed to […]
Globus Medical Reports Second Quarter 2018 Results
AUDUBON, Pa., Aug. 01, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced its financial results for the second quarter ended June 30, 2018. Worldwide sales were $173.4 million, an increase of 13.8% as reported Second quarter net income was $45.0 million, an increase of 56.9% Diluted earnings per share […]
NuVasive Announces Second Quarter 2018 Financial Results
SAN DIEGO, July 31, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced financial results for the quarter ended June 30, 2018. Second Quarter 2018 Highlights Revenue increased 8.5% to $281.6 million, or 7.7% on a constant currency basis; GAAP operating profit margin of 10.1%; […]
SeaSpine Reports Second Quarter 2018 Financial Results
CARLSBAD, Calif., July 30, 2018 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ:SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today financial results for the quarter ended June 30, 2018 and updated its financial outlook for 2018. Summary Second Quarter 2018 Financial Results and Recent Accomplishments Revenue of […]
Cerapedics Announces First Patients Enrolled in IDE Clinical Trial of P-15L Bone Graft for Transforaminal Lumbar Interbody Fusion Surgery
WESTMINSTER, Colo., July 30, 2018 /PRNewswire/ — Cerapedics, a privately-held orthobiologics company, today announced that the first patients have been enrolled in an investigational device exemption (IDE) clinical trial evaluating the safety and efficacy of the next-generation P-15L Peptide Enhanced Bone Graft in transforaminal lumbar interbody fusion (TLIF) surgery. The prospective, single-blinded, multi-center, randomized, non-inferiority pivotal IDE […]
OrthoPediatrics, Osseus, and many others…! SPINE Catalogue From A to Z: 18 Spine Companies to know with “O”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
Ulrich Medical USA says a Texas -based hospital owes thousands of dollars for medical devices
ST. LOUIS –– Ulrich Medical USA says a Texas -based hospital owes thousands of dollars for medical devices.The Missouri-based spinal implant manufacturer filed a lawsuit on July 17 in the U.S. District Court for the Eastern District of Missouri, alleging St. Camillus Hospital purchased more than $164,000 worth of devices during 2017 and 2018. But the […]
Nuvasive, Neurostructures, and many others…! SPINE Catalogue From A to Z: 13 Spine Companies to know with “N”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
NuVasive Unveils Spine’s First Integrated Surgical Automation Platform
SAN DIEGO, July 27, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the Pulse™ surgical automation platform has received 510(k) clearance from the U.S. Food and Drug Administration (FDA). Pulse is the foundation for Surgical Intelligence™, the Company’s […]
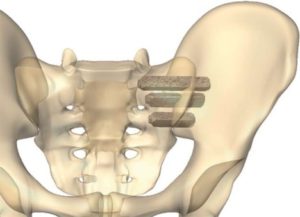
Mighty Oak Medical Receives FDA 510K Clearance for S2AI
July 25, 2018–Englewood, CO – Mighty Oak Medical announced today that its patient-specific, 3D printed FIREFLY® Pedicle Screw Navigation Guides have received an additional FDA 510K clearance for the expanded indications to include S2AI trajectory for sacral-iliac fixation in complex spinal reconstruction surgeries such as scoliosis. The precise mechanical guidance of patient-specific guides is ideal for […]