WARSAW, Ind., Jan. 04, 2019 (GLOBE NEWSWIRE) — OrthoPediatrics Corp. (“OrthoPediatrics”) (NASDAQ: KIDS), a company exclusively focused on advancing the field of pediatric orthopedics, announced today their recently updated and signed agreement with Hospithera to act as OrthoPediatrics’ exclusive sales agency in Belgium and the Netherlands. Since 2015, Hospithera has been OrthoPediatrics’ stocking distributor, developing sales in […]
NEWS
Spinal implant developer SpineEx withdraws $17 million IPO
SpineEx, which is developing spinal implants to improve spinal fusion surgeries, withdrew its plans for an initial public offering on Friday. It originally filed in September 2018 with a proposed deal size of $17 million. The company did not disclose a reason for the withdrawal. Since its initial filing, the company announced in a press […]
NuVasive CEO J. Christopher Barry Announces New Organizational Structure And Leadership Team
SAN DIEGO, Jan. 3, 2019 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced a new organizational structure and associated executive team that chief executive officer (CEO) J. Christopher (Chris) Barry has selected to lead the Company’s next phase of growth and innovation. Barry […]
SINTX Announces 30-year Spine Fusion Data with Silicon Nitride Implants
SALT LAKE CITY, Jan. 03, 2019 (GLOBE NEWSWIRE) — SINTX Technologies (NASDAQ: SINT) today reported the independent publication of 30-year clinical outcomes of lumbar fusion surgeries, performed with silicon nitride implants. The paper titled “Anterior Lumbar Interbody Fusion Using Reaction Bonded Silicon Nitride Implants” appeared in the December 2018 edition of the World Neurosurgery journal, […]
Prosecutors investigate spinal implant
(BBC.com)–The total disc replacement implant made by Cambridge-based Ranier Technology left patients across Europe in agony and needing further surgery after breaking inside their backs.Last month, BBC Panorama revealed how the Cadisc L implant was approved for use in humans despite evidence from studies in baboons that it could fail. Ranier withdrew it from market […]
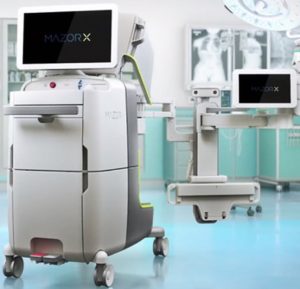
Medtronic Completes Acquisition of Mazor Robotics
DUBLIN – December 19, 2018 – Medtronic plc (NYSE:MDT) today announced it has completed the acquisition of Israel-based Mazor Robotics (NASDAQ:MZOR, TASE:MZOR.TZ). Under the terms of the acquisition agreement announced on September 20, 2018, Mazor shareholders will receive $58.50 per American Depository Share, or $29.25 per ordinary share, in cash. The total value of the […]
Which are the Key Competitors in the Deformity Market? Learn about 45 Scoliosis Devices
Today’s gold standard for correcting spinal deformities involves fusing an average of 10 motion levels using a large number of screws in a surgical procedure that lasts an average of six hours and costs upward of $100,000. Worldwide, there are approximately 126,000 scoliosis procedures per year, of which 40,000 are AIS, representing a $2.4 billion […]
Medtronic to pay $31M in DoJ settlement of medical device probes
Two companies now owned by Medtronic agree to pay $31M to resolve a U.S. Department of Justice probe into the marketing of a medical device meant to treat a vascular defect in the brain for unapproved purposes and paying kickbacks to hospitals to get them to use a second product. The U.S. Food and Drug Administration in […]
How many MIS systems are now in the market? Only 90 Minimally Invasive Pedicle Screw Systems!
According to iData Research, the U.S. market, valued at $7.7 billion in 2017. Of this total market share, 78% can be attributed to the traditional spine market, and the remaining, to the minimally invasive (MIS) segment. Growth in the former has slowed as newer, minimally invasive innovations gradually cannibalize the traditional spine market. Growth, at […]
Carmel spine surgeon wins $112M verdict in royalty battle with Medtronic
(www.ibj.com)–Dr. Rick C. Sasso, an Indiana spine surgeon and inventor, has won a sweeping, five-year legal battle against medical-device giant Medtronic, with a jury this week awarding him $112 million in damages.Sasso, 58, president of Carmel-based Indiana Spine Group, had claimed that Medtronic had violated a contract by not paying royalties he was due for […]
WishBone Medical, Inc. acquires Response Ortho
WARSAW, Ind., Nov. 28, 2018 (GLOBE NEWSWIRE) — WishBone Medical, Inc. announces the acquisition of Response Ortho. Response Ortho has a complete pediatric line of external and internal fixation and deformity correction products. Response Ortho has been in business for fourteen years and has built a modern, high-tech manufacturing facility with access to an experienced […]
Swiss implant makers come under the microscope
(swissinfo.ch)–The Implant Files revelations of faulty implants and weak safety standards are raising alarm bells for Switzerland’s rapidly growing medical device industry. Manufacturers and industry bodies say that safety standards have improved in the last decade, but do they go far enough? The market for implanted medical devices is booming globally. Tens of thousands of […]
Massive investigation of medical device industry highlights Memphis-based Medtronic Spine, kickback allegations
(commercialappeal.com)–An international journalism investigation of the medical device industry describes dangerous devices, kickbacks to doctors and weak regulations – and shines a spotlight on a medical company with a big presence in Memphis. That company is Medtronic, which has its main operations in Minneapolis and annual sales of about $30 billion. One of its units, […]
UK firm sold spinal implants that disintegrated
(theguardian.com)–A UK company’s spinal implants that allegedly moved and eroded in patients, and which are at the centre of legal action, have highlighted potential weaknesses in the way in which some medical devices enter the market, an investigation has revealed.Documents seen by the Guardian show the plastic discs were approved for sale by the British […]
Medtronic to Acquire Nutrino Health
DUBLIN and TEL AVIV, Israel – November 21, 2018 – Medtronic plc (NYSE:MDT), a global leader in medical technology, and Nutrino Health Ltd., a leading provider of nutrition-related data services, analytics, and technologies, today announced the companies have entered into a definitive agreement under which Medtronic will acquire Nutrino. Given that food and nutrition are […]
Altus Capital Partners, Inc. Acquires ChoiceSpine, LP
WILTON, Conn.–(BUSINESS WIRE)–Altus Capital Partners, Inc. (“Altus”), an investment firm focused on the North American manufacturing sector, today announced it has acquired ChoiceSpine, LP, a leading designer, manufacturer, and marketer of specialized spinal implants, instrumentation and biologics for the surgical treatment of complex spine disorders. Financial terms of the transaction were not disclosed. Based in […]
Mazor Robotics Shareholders Approve Merger Agreement With Medtronic
CAESAREA, Israel, Nov. 19, 2018 /PRNewswire/ — Mazor Robotics Ltd. (TASE: MZOR; NASDAQGM: MZOR), a pioneer and a leader in the field of robotic guidance systems, today announced that at a Special General Meeting of Shareholders held earlier today, Mazor Robotics shareholders approved the previously announced definitive merger agreement with wholly-owned subsidiaries of Medtronic plc (NYSE: MDT). Approximately […]
Statera Spine Announces Company Formation
AUSTIN, Texas–(BUSINESS WIRE)–Statera Spine, Inc. (Statera), a privately held software-enabled healthcare diagnostics company focused on spine imaging analytics, announced today the formation of the company as a subsidiary of Ortho Kinematics, Inc. Statera will leverage the operational backbone and technology platform developed by Ortho Kinematics, but will focus on the commercialization of Profile-ESP™. Profile-ESP™ is […]
Innovasis Enters Partnership with Stability Biologics
SALT LAKE CITY, November 19, 2018 – Innovasis, Inc., a fully integrated research, development and manufacturing spinal device company, today announced their expansion into the biologics market by signing a private-label agreement with Stability Biologics, LLC. Stability Biologics®, headquartered in Nashville, Tennessee with a state-of-the-art AATB accredited and FDA registered tissue processing and distribution center […]
Stryker completes acquisition of K2M
Kalamazoo, Michigan, Nov. 09, 2018 (GLOBE NEWSWIRE) — Stryker (NYSE:SYK) has completed the previously announced acquisition of K2M Group Holdings, Inc., a global leader of complex spine and minimally invasive solutions. “K2M’s broad product portfolio and robust pipeline complement Stryker’s leadership in the spine and neurotechnology markets,” said Spencer S. Stiles, Group President, Neurotechnology, Instruments […]
Why RTI Surgical has Acquired Paradigm Spine and for which Price?
RTI Surgical (RTIX) announced last week that they have agreed to acquire Paradigm Spine for up to $300 million.Paradigm Spine is a New-York based Company founded in 2005 to develop a full non-fusion product portfolio of motion preserving, tissue sparing technologies, as well as fusion products. Management is headed by Co-Founder, Chairman, and CEO Marc Viscogliosi, who was previously Principal […]
RTI Surgical® to Acquire Paradigm Spine
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (Nasdaq:RTIX), a global surgical implant company, and Paradigm Spine, LLC, a leader in motion preservation and non-fusion spinal implant technology, today announced that they have entered into a definitive agreement whereby RTI will acquire all outstanding equity interest of Paradigm Spine in a cash and stock transaction valued at up […]
Key Findings of the Spine Biologics Market
According to Allied Market Research, the Global spine biologics market generated revenue of $1,644 million in 2015, and is projected to reach $2,214 million by 2022, growing at a CAGR of 4.3% during the forecast period. Spine biologics are used during spine fusion surgery for the treatment of spinal deformities such as trauma, tumors, degenerative […]
13 Spine Companies to Know with “W, X and Z”
For many weeks, we have been working on this Spine Catalogue 2018-19 with the main objective to index all the Companies that manufacture spinal implants. Today, we have almost finished this project with more than 300 included. We have revised the list many times and we have seen that there are still some companies that […]
14 Spine Companies to Know with “U”and “V”
According to the new market research report published by MarketsandMarkets™, the Pedicle Screw Systems Market is expected to reach USD 724.23 million by 2023 from USD 538.64 million in 2018, at a CAGR of 6.1%. The restraining factors of Global Spinal Implants Market are as follows: Absence of proper diet and exercise and declining bone density Scarcity on the emerging […]
Here’s Intuitive Surgical’s Evidence That Robotic-Surgery Boom Continues
Late Thursday, Intuitive Surgical (ISRG) outdid itself — posting 20% growth in procedures using its robotic-surgery system in the third quarter.That was the best growth rate for Intuitive Surgical’s da Vinci Surgical Systems procedures in at least three years. The robotic-surgery outlet credited growth in U.S. general surgery and worldwide urologic procedures for the boom in robotic surgery. Da Vinci […]
21 Spine Companies to Know with “T”
According to the new market research report published by MarketsandMarkets™, the Pedicle Screw Systems Market is expected to reach USD 724.23 million by 2023 from USD 538.64 million in 2018, at a CAGR of 6.1%. Although this market is dominated by the big Corporations such as Medtronic, Johnson&Johnson, Stryker, Nuvasive or Globus Medical, there are in addition more than 300 […]