DUBLIN, Dec. 16, 2020 /PRNewswire/ — Medtronic plc (NYSE: MDT) announced that the U.S. Food and Drug Administration (FDA) has cleared the use of navigated interbody and Midas Rex™ high speed drills with the Mazor™ Robotic Guidance System earlier than originally anticipated. The Mazor™ platform now provides surgeons with unprecedented procedural integration by seamlessly combining the power of Midas Rex™ drills […]
NEWS
SI-BONE, Inc. Announces that Humana Establishes Exclusive Coverage for iFuse in MIS SI Joint Fusion Procedures
SANTA CLARA, Calif., Dec. 16, 2020 (GLOBE NEWSWIRE) — SI-BONE, Inc., (Nasdaq: SIBN) a Silicon Valley-based medical device company dedicated to solving musculoskeletal disorders of the sacro-pelvic anatomy, today announced that Humana has revised its coverage policies to cover minimally invasive SI joint fusion using triangular titanium implants (i.e., iFuse Implant System). Humana is one […]
Centinel Spine Continues to Innovate in Total Disc Replacement through First Case in U.S. with Angled Implants
WEST CHESTER, Pa., Dec. 15, 2020 /PRNewswire/ — Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced the first implantation of newly available angled endplates in the U.S. for the prodisc® L Lumbar Total Disc Replacement System. These unique endplates have been designed to shift the lordotic angle of the implant to the […]
7D Surgical Surpasses its 50th Unit Sale While Expanding into European Market
TORONTO, Dec. 15, 2020 /PRNewswire/ — 7D Surgical, a Toronto based medical device company developing advanced, cutting-edge optical and machine-vision technologies for surgical navigation, announced today that it is expanding its footprint into the European Market with multiple units already placed. 7D Surgical has also completed several spinal surgical cases and tissue labs utilizing the FLASH™ Navigation System in Germany and Switzerland. Since […]
SeaSpine Announces FDA 510(k) Clearance for Amended Indication for NanoMetalene® Surface Technology
CARLSBAD, Calif., Dec. 15, 2020 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its NanoMetalene® surface technology with amended indications. NanoMetalene describes […]
First DSG Connect surgery at Trousseau Hospital in Paris: new milestone of the SpineGuard robotics program
Paris (France) and Boulder (CO, USA), December 14, 2020 – SpineGuard (FR0011464452 – ALSGD), an innovative company that deploys its DSG® (Dynamic Surgical Guidance) sensing technology to secure and streamline the placement of bone implants, announces the first surgery performed at the Trousseau Hospital in Paris with the new DSG Connect platform as part of the collaboration […]
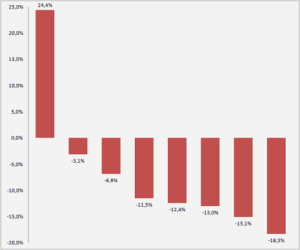
Which are the Leading Spine companies that are better withstanding the negative effects of COVID-19 in Sales?
This year 2020 is being tough for everyone. Most companies are experiencing significant sales drops. The Spine market in 2020 has fallen an average of 12% due to the COVID-19. Most of the leading companies are publishing negative results as Medtronic (-13%), Johnson & Johnson (-18,3%), Stryker (-12,4%), Nuvasive (-11,5%), and Zimmer Biomet (-15,1%). However, […]
Spinal Elements® Announces First Procedure with Sapphire X™ Anterior Cervical Fixation System
CARLSBAD, Calif.–(BUSINESS WIRE)–Spinal Elements, a spine technology company, today announced the first procedure with its Sapphire X Anterior Cervical Fixation System. FDA clearance of the Sapphire X system was announced in July of this year as the newest product in the MIS Ultra™ suite of products aimed at minimizing the unintended consequences of traditional spine […]
BIOMECH Paonan Launches NEST-C Interbody System
BIOMECH Paonan a medical device company focused on designing and manufacturing spinal solutions announced the launch of its NEST-C (anterior cervical interbody fusion) System. NEST-C Interbody System is a cervical implant consisting both solid and porous structures designed for bone in‑growth and biological fixation. Inspired by the microstructure of cancellous bone, and enabled by Paonan’s […]
Globus Medical Trauma Division Awarded Purchasing Agreement with Premier Inc.
AUDUBON, Pa., Dec. 09, 2020 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, was recently awarded a group purchasing agreement with Premier, one of the largest group purchasing organizations in the United States. The agreement covers Globus’ Trauma Platform, which includes ANTHEM® Plating, AUTOBAHN® Nailing, ARBOR® External Fixation, and CAPTIVATE® Screw Platforms. […]
Amplify Surgical®, Inc. Announces First Surgical Case with 12-Degree Hyperlordotic dualX® Dual Expanding Interbody Fusion Implant
LAGUNA HILLS, CALIF. (PRWEB) DECEMBER 08, 2020–Amplify Surgical, Inc., a medical device company focused on innovative dual-expanding technology for the lumbar spine, is excited to announce the launch of the dualX T/PLIF 12-Degree hyperlordotic interbody cages. Dr. Nima Salari, MD, FAAOS, from Scottsdale AZ, became the first spine surgeon to implant the new hyperlordotic dualX interbody […]
SeaSpine Announces Limited Commercial Launch of Meridian™ ALIF System with Reef™ A Interbody
CARLSBAD, Calif., Dec. 07, 2020 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch of its Meridian ALIF (anterior lumbar interbody fusion) System and Reef A interbody with NanoMetalene® surface technology. “SeaSpine has created one […]
CMS grants transitional pass-through (TPT) payment for Stryker’s SpineJack® System
KALAMAZOO, Michigan, USA, Dec. 4, 2020 /PRNewswire/ –The Centers for Medicare & Medicaid Services (CMS) announced the SpineJack implantable fracture reduction system qualified for the Transitional Pass-Through (TPT) payment as part of the 2021 Medicare Hospital Outpatient Prospective Payment System. The SpineJack System now becomes one of only 11 medical devices to receive TPT status since 2016. […]
Nexxt Spine Announces First Case with Newly Launched NEXXT MATRIXX® SA Cervical Turn-Lock System
December3, 2020- Noblesville, IN– Nexxt Spine, LLC, amedical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, is proud to publicize the firstimplantation of the NEXXT MATRIXX®SA Cervical Turn-Lock System. Showcasingtechnology unique to the NEXXT MATRIXX® line of products, the system is centered around anintentionally engineered 3D laser […]
Centinel Spine’s Unique 3D-Printed Porous-Titanium FLX™ Platform Experiences Rapid Market Acceptance through Over 3,500 Implantations
WEST CHESTER, Pa., Dec. 3, 2020 /PRNewswire/ — Centinel Spine, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced that it has successfully surpassed 3,500 implantations with its novel FLX™ technology platform of 3D-Printed Porous-Titanium Interbody Devices. This implant milestone was quickly achieved after full commercial availability of the FLX technology over the last year. The FLX platform represents the […]
SeaSpine Announces Limited Commercial Launch of its Admiral™ ACP System
CARLSBAD, Calif., Dec. 02, 2020 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch and subsequent first case of its Admiral ACP System. Admiral ACP represents the next generation of anterior cervical plating designed to strike […]
Camber Spine’s OLIF Retractor System Receives ‘2020 Best Spine Technology Award’
KING OF PRUSSIA, Pa.–(BUSINESS WIRE)–Camber Spine, a leading innovator in spine and medical technologies has been presented with Orthopedics This Week’s “2020 Best Spine Technology Award” for its Lateral ALIF / OLIF Retractor System, an integral part of the OLIF (oblique lateral interbody fusion) technique for anterior column reconstruction procedures. The prestigious award, typically presented […]
Safe and Efficacious Treatment of Spondylolisthesis via MIS TLIF Approach with the FlareHawk® Expandable Cage
PALM BEACH GARDENS, Fla., Dec. 01, 2020 (GLOBE NEWSWIRE) — Integrity Implants Inc., a privately held medical device company dedicated to delivering innovative solutions for spine surgery, today announced the publication of another in a series of recent clinical articles in the International Journal of Spine Surgery (www.ijssurgery.com) highlighting its FlareHawk® expandable interbody cage. In their paper, Clinical […]
First use of nView medical’s novel 3D imaging technology in pediatric spine surgery
SALT LAKE CITY (PRWEB) NOVEMBER 30, 2020–Children’s National Hospital today performed the first surgical use of breakthrough medical imaging technology designed specifically for kids. The innovation, insta-3D™ imaging from company nView medical, is designed to make 3D images available in the operating room quickly and safely. The 3D images provide surgeons with better visualization, allowing them […]
NuVasive Unveils C360 Portfolio and Launches New Anterior Cervical Plate
SAN DIEGO, Nov. 30, 2020 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced the unveiling of its new C360™ cervical spine portfolio, including the commercial launch of the Anterior Cervical Plating (ACP) system, featuring the thinnest plate on the market1 at 1.6 mm. “The unveiling […]
Aurora Spine Announces New Pain Care Division
CARLSBAD, Calif., Nov. 30, 2020 (GLOBE NEWSWIRE) — Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSXV: ASG), a designer and manufacturer of innovative medical devices that improve spinal surgery outcomes, today announced it has created a new Pain Care Division focused on the rapidly growing segment of pain care. Pain management has become more […]
Centinel Spine Recognized as Top Orthopedic Company in 2020
WEST CHESTER, Pa., Nov. 24, 2020 /PRNewswire/ — Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced recognition by Becker’s Spine as the top Orthopedic Solution Company in 2020. Becker’s Spine referenced MedTech Outlook as the source behind the recognition of innovators at the forefront of orthopedic technologies. This is the second year in a […]
Medtronic Reports Second Quarter Fiscal 2021 Financial Results
DUBLIN, Nov. 24, 2020 /PRNewswire/ — Medtronic plc (NYSE:MDT) today announced financial results for its second quarter of fiscal year 2021, which ended October 30, 2020. The company reported second quarter worldwide revenue of $7.647 billion, a decrease of 0.8 percent as reported and 1.5 percent on an organic basis, which adjusts for the $59 million benefit of foreign currency translation. Unless […]
Surgalign Holdings, Inc. Appoints Paul Lewicki, PhD to Board of Directors
DEERFIELD, Ill., Nov. 23, 2020 (GLOBE NEWSWIRE) — Surgalign Holdings, Inc. (Nasdaq: SRGA), a global pure-play spine company focused on advancing spine surgery including through the application of digital technologies to improve patient outcomes, today announced the appointment of Paul Lewicki, PhD as a member of its board of directors. With Dr. Lewicki’s appointment the […]
UPDATED: 8 Single-Use Spinal Systems to Know! Do they really have Advantages?
10 years ago, in 2010 a French company launched the first single-use spine system creating a lot of expectation and also a lot of skepticism about the future of this new concept. Since then, Safe Orthopedics has not stopped growing and several competitors have also appeared on the market. Thus, in 2011 Xenco Medical was […]
Spine Wave announces upgrades to its Leva® Interbody Device system
SHELTON, Conn., Nov. 19, 2020 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce immediate availability of the upgraded Leva® Interbody Device system. The Leva® Interbody Device is now a complete solution for surgeons preferring expandable titanium implants used together with large amounts of bone graft. The Leva® Interbody Device is Spine Wave’s all-titanium expandable […]
Centinel Spine Wins 2020 Spine Technology Award for its Two-level Indication for the prodisc® L Total Disc Replacement System
WEST CHESTER, Pa., Nov. 19, 2020 /PRNewswire/ — Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced it has won a 2020 Orthopedics this Week Spine Technology Award for its two-level indication approval by the FDA for the prodisc® L Total Disc Replacement (TDR) system. The 12th Annual Spine Technology Awards recognize exemplary and innovative spine surgery […]