BORDEAUX, France & BOSTON–Regulatory News:Implanet (Paris:ALIMP) (OTCQX:IMPZY) is holding a conference call in French at 6:30pm this evening. To join the conference call, dial in on +33(0)1.70.71.01.59 then enter PIN code 56375595#. Implanet (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, is today […]
NEWS
SPINEART Reports 1000th Juliet TI-Life US Procedure
SPINEART today announced that it has completed over 1,000 JULIET Ti-LIFE procedures in the US. Ti-LIFETECHNOLOGY is an enhanced algorithm for additive manufacturing resulting in a porous structure that mimics trabecular bone. Ti-LIFE structure has an average pore diameter and overall porosity similar to trabecular bone, which may enable cell colonization and promote bone in-growth. […]
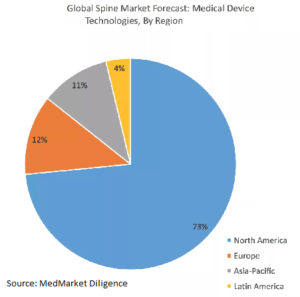
Europe spinal implants market to reach $3.8 B by 2024.The Top 20 European Manufacturers Of Spinal Implants (Part I)
How Big is The Europe Spinal Implants Market? The Europe Spinal Implants Market is expected to exceed more than US$ 3.8 Billion by 2024 by growing at a compound annual growth rate of 5.7 percent according to a Data Bridge Market Research analysis. The German market is the greatest followed by France, United Kingdom, Italy and Spain. The major […]
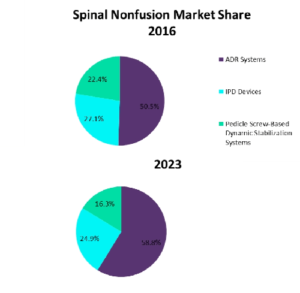
Global spinal nonfusion market is expected to grow to $1.3bn in 2023. Are Motion-retaining spinal implants set back by insufficient clinical efficacy? 45 Pedicle Screw Dynamic Systems to know
According to GlobalData the global spinal nonfusion market valued $696m in 2016 and is expected to grow to $1.3bn in 2023 propelled by the adoption of cervical ADR systems.These nonfusion devices include the pedicle screw-based dynamic stabilisation systems among others (ADR systems, interspinous process decompression (IPD) devices). Only cervical artificial disc replacement systems have collected a body […]
Medacta International Underscores Global Education Commitment With 1st M.O.R.E. Americas Symposium
MONTREAL–(BUSINESS WIRE)–Medacta International, the family-owned global leader in innovative joint replacement and spine surgery products, this morning kicks off the 1st M.O.R.E. Americas Symposium, held April 20-21, 2018 in Montreal. An expansion of Medacta’s M.O.R.E. International Symposium, which is held bi-yearly in Europe, this event will gather upwards of 150 surgeons from throughout the Americas and […]
SAGICO Announces Market Release of Expandable Stand-Alone Cervical Technology
TAMPA, Fla., /PRNewswire-iReach/ — Previously, the US FDA issued to SAGICO a 510(k) clearance for the SAGICO IBF (Inter-Body Fusion) System. Contained within the 510(k) was the clearance for the cervical appliance branded as the “ARION”. Per the Indications for Use, “…the Arion Cervical implant is an interbody fusion device intended for use in skeletally mature […]
This remarkable spinal implant was created by an algorithm
This coral-like form is a spinal implant. Created by Californian medical company NuVasive, it is made from titanium and fits precisely between two vertebrae. By mimicking the porousness and stiffness of human bone, it can accelerate bone growth following back surgery. Spinal surgeons typically use implants made from high-performance plastic, because the material is less […]
SpineGuard® Announces First Spinal Deformity Surgery in China with PediGuard® by Prof. Yong Qiu
PARIS & SAN FRANCISCO–Regulatory News: SpineGuard (FR0011464452 – ALSGD), an innovative company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, announced today that Professor Yong QIU performed the first spinal deformity surgery in China with the PediGuard® device. Pr. Yong QIU is the […]
Medtech companies raised more than $839 million in Q1
Venture capital firms invested more than $839 million in medical devices and equipment companies during the first quarter of 2018, an increase of more than $338 million from the last quarter of 2017, according to the MoneyTree Report from PricewaterhouseCoopers (PwC) and CB Insights. There was a total of 48 deals during Q1 this year, […]
Two Data Presentations Highlight the Unique Potential of Spineology’s Duo™ Implant
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., an innovator in anatomy-conserving spine surgery, is excited to announce that two laboratory studies relating to the Spineology Duo™ Lumbar Interbody Fusion System were presented at the recent annual meeting of the International Society for the Advancement of Spine Surgery (ISASS) in Toronto, Canada. The Duo implant is the first […]
Safe Orthopaedics announces the 10,000th surgical kit sold
ERAGNY-SUR-OISE, France–SAFE ORTHOPAEDICS (Paris:SAFOR) (FR0012452746 – SAFOR), a company specialized in the design and marketing of single-use implants and instruments improving the minimally invasive treatment of spinal fracture conditions, is today announcing that it has now sold more than 10,000 surgical kits worldwide since they were first launched. Safe Orthopaedics’ sterile kits all include single-use instruments […]
Will Spinal Deformities Drive the Global Spinal market in the next 10 years? Did you know that there are already more than 30 systems in the market?
According to Persistence Market Research, the global deformity spinal system market is driven by the increasing prevalence of spinal deformities and instabilities and spine related diseases.A rising number of spine deformity cases and increasing technological advancements will drive the global spinal implants and devices market. Deformity spinal implant keep on creating as innovation advances the […]
Lumbar Artificial Disc Replacement with the activL® Artificial Disc Found to be More Effective at Delaying the Progression of Adjacent-Level Disease, a Costly Outcome which Spinal Fusion has been Found to Accelerate
CENTER VALLEY, Pa., April 12, 2018 /PRNewswire/ — Aesculap Implant Systems, LLC today announced the publication of “Progression of Adjacent-level Degeneration after Lumbar Total Disc Replacement: Results of a Post-hoc Analysis of Patients with Available Radiographs from a Prospective Study with 5-year Follow-up” in Spine.1 This is another significant milestone in Aesculap’s commitment to enhancing patient access to lumbar total […]
Spineology U.S. Clinical Trial for Interbody Fusion
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., an innovator in anatomy-conserving spine surgery, is excited to announce that John Chi, M.D., M.P.H, Associate Professor of Neurosurgery at Harvard Medical School and the Director of Neurosurgical Spinal Oncology at Brigham and Women’s Hospital in Boston MA, has presented the first 12-month outcomes data from Spineology’s SCOUT clinical trial. […]
SpinalCyte Applies with FDA to Expand Human Clinical Trial of CybroCell™ Dermal Fibroblasts as Investigational New Drug (IND)
HOUSTON–(BUSINESS WIRE)–SpinalCyte, LLC, a Texas-based regenerative medicine company focused on regrowth of the spinal disc nucleus using Human Dermal Fibroblasts, has filed an Investigational New Drug (IND) Application with the U.S. Food and Drug Administration (FDA) to expand its study of CybroCell Human Dermal Fibroblasts to treat degenerative disc disease. “The filing of this IND […]
Life Spine® Achieves 93% Sales Growth of ProLift® Micro-Invasive Expandable TLIF/PLIF Spacer System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today continued accelerated growth of their Micro-Invasive Expandable TLIF/PLIF Spacer System, ProLift. Sales in the first quarter grew 93% as compared to sales of Q1 2017. Mariusz Knap, Vice President of Marketing […]
CoreLink Announces Full Commercial Launch of Foundation™ 3D Printed Titanium Interbody Cage Systems
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a fast growing, vertically integrated designer and manufacturer of spinal implant systems, announced the full commercial release of its Foundation 3D Interbody Cage Systems for cervical and lumbar fusions. The titanium devices utilize additive manufacturing to create Mimetic Metal™ technology that mimics key characteristics of natural bone, featuring 100% open-pore architecture […]
4WEB Medical Surpasses 30,000 Implant Milestone
TORONTO, April 11, 2018 /PRNewswire/ — 4WEB Medical, the industry leader in 3D printed implant technology, announced today at the annual meeting of the International Society for the Advancement of Spine Surgery (ISASS) that the company has exceeded 30,000 implants of its proprietary Truss Implant Technology worldwide. Jessee Hunt, 4WEB President and CEO, said, “We […]
RTI Surgical® Showcases SImmetry® System Data and TETRAfuse® 3D Technology at ISASS 2018
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (Nasdaq: RTIX), a global surgical implant company, presented data on the SImmetry Sacroiliac Joint Fusion System at the International Society for the Advancement of Spine Surgery (ISASS) 2018 Annual Meeting taking place April 11-13, 2018 in Toronto, Canada. Data show the SImmetry System, the only minimally-invasive surgical procedure that promotes […]
Titan Spine® Announces Review of Research Supporting Its nanoLOCK® Titanium Spinal Implants Published in SPINE
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine®, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced that a review of research supporting its nanoLOCK® surface technology has been published in the journal SPINE. nanoLOCK® is the company’s next-generation surface technology featuring enhanced micro and nano-scaled architecture, proven to significantly improve the […]
Implanet Partners with the SRS
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–Regulatory News:Implanet (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, is announcing its participation at the next Scoliosis Research Society (SRS) course in Bordeaux. JAZZ will be presented in a workshop on restoring sagittal alignment. […]
SI-BONE, Inc. Announces Publication of 3-Year Long-term Prospective Outcomes Including Randomized Controlled Trial Data on 103 Patients Post SI Joint Fusion with Triangular iFuse Implant System®
SAN JOSE, Calif., April 9, 2018 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the minimally invasive surgical (MIS) treatment of the sacroiliac (SI) joint with the iFuse Implant System® (“iFuse”) announced the publication of results from a long-term outcomes study called LOIS (Long Term Outcomes from INSITE and SIFI). The publication, titled Long-term prospective outcomes after minimally invasive trans-iliac sacroiliac […]
45 Expandable Lumbar cages: Which are their Advantages vs Static Cages?
Spinal surgery has evolved in the last years toward MIS approaches but also trying to maximize Lumbar Lordosis and to control Sagittal and Coronal Alignment. The usage of Interbody fusion devices has become critical for that purpose as well as a challenge when facing minimally invasive surgeries. Although, now most of the companies -with the […]
CTL Wins FDA Approval for Seurat Universal Pedicle Screw
DALLAS, TX / ACCESSWIRE / March 29, 2018 / CTL Medical Corporation, a Dallas-based medical device manufacturing and service company, has recently secured clearance and approval from the Food and Drug Administration to market its new SEURAT Universal Pedicle Screw System for the practice of spine fusion surgery. This is the second FDA clearance for the […]
Medicrea Publishes 2017 Full Year Results and First Quarter 2018 Sales
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea® Group (Euronext Growth Paris: FR0004178572 – ALMED), pioneering the convergence of healthcare IT and next-generation, outcome-centered spinal device design and manufacturing with UNiD ASI™ (Adaptive Spine Intelligence) technology, publishes its 2017 IFRS annual results, as audited and approved by the Board of Directors on April 4, 2018, as well […]
Globus Medical Announces Launch of ELSA® Expandable Lateral Spacer System
AUDUBON, Pa., April 05, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the launch of two innovative lateral expandable interbody spacers with integrated screw fixation, ELSA® AL and ELSA®-ATP, the latest additions to Globus’ unmatched expandable solutions. These next generation expandable implants provide an optimized fit for the […]
SpineGuard Reports Q1 2018 Revenue
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News: SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, reported today its first-quarter 2018 revenue of €1.95m decreasing 7.9% in constant currency (-2.9% in the USA) and of €1.8m at […]