AUSTIN, TEXAS (PRWEB) SEPTEMBER 18, 2018–2018 has all the makings of a banner year for ImplantBase, the creator of the industry-leading, cloud-based software platform that drives digital transformation for orthopedic implant manufacturers. In addition to record growth in new implementations of its platform, the company has unveiled a refresh of its brand and digital presence. Amid […]
2018
MiRus™ Wins Two 2018 Spine Technology Awards
ATLANTA, Sept. 17, 2018 /PRNewswire/ — MiRus has won the Annual Spine Technology Awards for both of its initial products in the implant and navigation categories. The Spine Technology Awards are intended to bring increased recognition to exemplary and innovative spine surgery products and the engineering teams and inventors who create them. The awards will be presented to […]
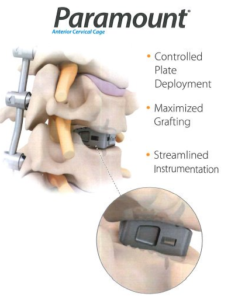
Spine Wave Announces the Commercial Launch of the Paramount® Anterior Cervical Cage
SHELTON, Conn., Sept. 17, 2018 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce the commercial launch of the Paramount® Anterior Cervical Cage. The Paramount® Anterior Cervical Cage is a titanium anterior cervical implant with integrated fixation blades. The patented system offers controlled, less invasive blade deployment and maximized bone grafting. The Paramount® Anterior Cervical […]
Simplify® Medical Technology Selected for Innovative Technology Paper at NASS 2018 Annual Meeting
SUNNYVALE, Calif., Sept. 17, 2018 (GLOBE NEWSWIRE) — Simplify Medical Pty Ltd, maker of the Simplify® cervical artificial disc, announced today that an abstract for preliminary data from the Company’s 1-level Investigational Device Exemption (IDE) clinical trial has been selected as an Innovative Technology Presentation during the 2018 North American Spine Society (NASS) Annual Meeting, […]
SpineVision® Receives FDA Clearance and CE Mark for Next-Gen Titanium 3D-Printed HEXANIUM TLIF Cage
ANTONY, France–(BUSINESS WIRE)–SpineVision announced today that it has received FDA clearance and CE mark for its next-generation titanium 3D laser-printed HEXANIUM TLIF (Transforaminal Lumbar Interbody Fusion) cage for back surgery. SpineVision will introduce its new product to spine surgeons at “EuroSpine 2018” (Booth #24C) in Barcelona, Spain (Sept. 19-21), and “NASS 2018” (Booth #1311) in […]
Globus Medical Acquires Surgimap®
AUDUBON, Pa., Sept. 13, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the acquisition of Nemaris Inc., a privately held company that markets and develops Surgimap®, a leading surgical planning software platform. Surgimap® allows healthcare professionals to simulate potential surgical outcomes and share medical imaging globally to improve […]
SpineGuard Secures 9.0 M€ Financing
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News:SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets instruments designed to secure the placement of surgical implants by bringing real-time digital technology into the operating room, announced today that it secured 9.0 million euro of financing. The two financings consist of: The issuance of a 6.0 million […]
K2M Group Holdings, Inc. Announces Definitive Agreement To Be Acquired by Stryker Corporation
LEESBURG, Va., Aug. 30, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (Nasdaq: KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced a definitive merger agreement with Stryker Corporation (“Stryker”, NYSE: SYK) pursuant to which Stryker has agreed to acquire […]
FDA Grants Eden Spine 510(K) Clearance for Its Thoraco Lumbar Spine Locking Plate – SPHYNX™
GENEVA, SWITZERLAND (PRWEB) AUGUST 30, 2018–The SPHYNX™ is the ideal complement to Eden Spine proprietary, expandable titanium vertebral body replacement implant with rotatable endplates, the GIZA™ which provides multiple angulation options by simple endplates rotation. The GIZA™ is intended to replace and fuse a collapsed, damaged, or unstable vertebral body due to a tumor or […]
Globus Medical to Exhibit Latest Advancements in MIS Spine Surgery at SMISS Annual Forum
AUDUBON, Pa., Aug. 29, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, will feature its ExcelsiusGPS® revolutionary robotic guidance and navigation system along with its latest advancements in expandable interbody technology at the Society for Minimally Invasive Spine Surgery (SMISS) Annual Forum being held September 6-8, 2018, in Las Vegas, […]
Nanovis Announces Successful Completion of $5.5 Million Investment Round To Fund Technology-Driven Growth
CARMEL, IND. (PRWEB) AUGUST 28, 2018–Nanovis, an innovative and fast-growing technology company selling nano-technology enhanced spinal implants, announced today the successful completion of a $5.5 million funding round managed by Commenda Securities. Key investors include Elevate Ventures, 1st Source Capital Corporation, Purdue’s Foundry Investment Fund, Commenda Capital, and Ellipsis Ventures. “Our investment in Nanovis is consistent […]
SI-BONE, Inc. Announces France’s National Healthcare System Establishes Exclusive Positive Coverage for the Triangular iFuse Implant System® for MIS SI Joint Fusion
SANTA CLARA, Calif., Aug. 27, 2018 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the minimally invasive surgical (“MIS”) treatment of the sacroiliac (“SI”) joint with the iFuse Implant System® (“iFuse”) announced that iFuse will be added to the List of Refundable Products and Services in France (Liste des Produits et Prestations Remboursables – LPPR), meaning that the French National […]
7D Surgical Announces New Distribution Agreement With LifeHealthcare Group Limited for Australia and New Zealand
TORONTO, Aug. 27, 2018 /PRNewswire/ — 7D Surgical Inc announced today that it has entered into a new supply agreement for the distribution of 7D Surgical’s innovative image guidance technologies with LifeHealthcare Group Limited. LifeHealthcare is a leading independent provider of medical devices and healthcare solutions in Australia and New Zealand. 7D Surgical develops advanced optical technologies and machine vision-based […]
SPINE Companies Catalogue From A to Z: 10 Spine Companies to know with “R”
Over the recent years, global spinal fusion market has been witnessing growth, on account of several driving factors including rising healthcare awareness amongst consumers, surging prevalence of spinal deformities due to accidents along with increasing health expenditure in developing countries. Moreover, ongoing demographic shift towards geriatric population with significant population suffering from inveterate spinal ailments, […]
Medicrea to Begin Trading on the OTCQX Market in the U.S.
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris: FR0004178572 – ALMED), pioneering the transformation of spinal surgeries through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD ASI™ technologies, announced today that the Company will begin trading on the OTCQX Best Market under the tickers “MRNTY” and “MRNTF” on Tuesday, […]
Caldwell Cassady & Curry Helps Acantha Win $8.2 Million Patent Infringement Verdict Against Medical Device Giant DePuy Synthes
GREEN BAY, Wisc., Aug. 23, 2018 /PRNewswire/ — The Dallas-based intellectual property and business litigation law firm Caldwell Cassady & Curry has won an $8.2 million patent infringement verdict for Acantha LLC against medical device manufacturer DePuy Synthes, a subsidiary of health care giant Johnson & Johnson (NYSE : JNJ ). A jury of five […]
Centinel Spine Announces Initial Cases with FLX™ Platform of 3D Printed All-Titanium Interbodies
NEW YORK, Aug. 23, 2018 /PRNewswire/ — Centinel Spine, LLC announced today that it has completed initial cases with three different implant portfolios within their FLX™ Platform of Integrated Interbody™ and non-integrated interbody fusion devices. Centinel Spine is the largest privately-held spine company focused on anterior column reconstruction. FLX devices are 3D-printed, all-titanium implants which […]
First Silony VERTICALE Open Procedures Performed in United States
MIAMI, Aug. 22, 2018 /PRNewswire/ — Silony Medical is pleased to announce that it has performed its first lumbar cases in the US. Silony offers tailor-made sets of sterile pedicle screws ranging from 4.5 to 10.2mm width in 25 to 100mm lengths. One of the initial users, Dr. Mehta at St. Mary Medical Center in […]
Life Spine Announces Another Period of Staggering Growth for PROLIFT Expandable Spacer System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that the state of the art PROLIFT® Expandable Spacer System has grown 70% in revenues from the first half of 2018 over the same period the prior year. “There are many […]
EOS imaging Achieves Its 100th Installation in the United States
PARIS–EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI – Eligible PEA – PME), the pioneer of 2D/3D imaging and data solutions for orthopedics, today announced that the Methodist Dallas Medical Center installed an EOS® system for low-dose, 2D/3D imaging of adult and pediatric patients. The installation marks the 100th system installed in the United States and […]
Life Spine to Showcase Latest Innovations and Micro-Invasive Expandable Technology at NASS 2018
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that it will be participating in the 33rd North American Spine Society (NASS) Annual Meeting, taking place in Los Angeles, September 26th – 29th, 2018. Life Spine is excited to showcase […]
Amedica Announces Successful Completion of First Valeo® C+CSC with Lumen Interbody Fusion Surgery
SALT LAKE CITY, Aug. 21, 2018 (GLOBE NEWSWIRE) — Amedica Corporation (NASDAQ: AMDA), an innovative biomaterial company that develops and commercializes silicon nitride for biomedical applications, is pleased to announce the successful completion of the first spine fusion surgery using the Valeo C+CSC with Lumen Interbody Fusion Device. The first surgery was performed by Dr. […]
CoreLink, LLC Announces 510(k) Clearance for Foundation™ 3D Anterior Lumbar (ALIF)
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a vertically integrated manufacturer of spinal implant systems, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market The FoundationTM 3D Anterior Lumbar (ALIF) Interbody device. This marks the launch of the latest addition to CoreLink’s Foundation 3D Interbody Cage family. Jay Bartling, CEO, said, “The Foundation […]
NuVasive Appoints Dale Wolf To Lead Ohio Manufacturing Facility
SAN DIEGO, Aug. 16, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the appointment of Dale Wolf as vice president, Manufacturing, who will oversee the Company’s West Carrollton, Ohio manufacturing facility. Mr. Wolf will fill a newly created […]
China: Innovative Hybrid PEEK-titanium Expandable Cage for DLIF and OLIF Procedures Demonstrated
THORNTON CLEVELEYS, ENGLAND (PRWEB) AUGUST 14, 2018//The growing interest in PEEK-OPTIMA™ spinal implants in China received further impetus at the 11th Congress of the Chinese Association of Orthopaedic Surgeons (CAOS) held in partnership with the North American Spine Society (NASS). At this event, Fule Science & Technology Development, Beijing, and Invibio Biomaterial Solutions partnered to […]
Zavation Launches Facet Screw and Sacroiliac (SI) Screw Systems
FLOWOOD, Miss., Aug. 14, 2018 /PRNewswire/ — Zavation, an employee-owned medical device company that designs, develops, manufactures and distributes medical device products, announced today the launch of a fenestrated Facet Screw system and a Sacroiliac (SI) Screw system. For more information on Zavation’s complete product portfolio, visit http://zavation.com/. The Zavation Facet Screw system is a […]
7D Surgical Receives FDA Approval For Cranial Surgery
TORONTO, Aug. 13, 2018 /PRNewswire/ — 7D Surgical announced today that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Cranial Module. This achievement is a crucial step in the commercial launch of 7D Surgical’s innovative Machine-vision Image Guided Surgery (MvIGS) system for cranial surgery throughout the United States. […]