CARLSBAD, Calif., Oct. 02, 2018 (GLOBE NEWSWIRE) — In a release issued under the same headline earlier today by SeaSpine Holdings Corporation (NASDAQ: SPNE) please note that in the fifth paragraph of the release under 2018 Financial Outlook the growth should be 7% to 8% over full-year 2017 revenue, not 6% to 9% over full-year […]
2018
ApiFix Surpasses 250 Scoliosis Patients Treated and Six-Year Follow-up of Earliest Cases
BOSTON–(BUSINESS WIRE)–ApiFix Ltd., a leading motion-preservation scoliosis correction company, announced today that its patented Minimally Invasive Deformity Correction (MID-C) technology has now been used to treat more than 250 young patients diagnosed with progressive scoliosis. The company also announced that the earliest treated patients have reached their six-year follow-up milestone. ApiFix’s MID-C technology is a […]
AI And Robotics Are Taking Robotic Surgery To New Levels
(INVESTORS.COM)–With artificial intelligence now firmly entrenched in many hospital operating rooms, the field of robotic surgery is starting to get competitive.Giant companies like Alphabet (GOOGL), Johnson & Johnson (JNJ) and Medtronic(MDT) are training their sights on Intuitive Surgical (ISRG), the king of robotic surgery companies. But analysts say the booming medical technology segment has lots of room to run. Robotic surgery is […]
SCOUT:Spineology U.S. Clinical Trial for Interbody Fusion
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., an innovator in anatomy-conserving spine surgery, is excited to announce that John Chi, M.D., M.P.H., Associate Professor of Neurosurgery at Harvard Medical School and the Director of Neurosurgical Spinal Oncology at Brigham and Women’s Hospital in Boston, Massachusetts, presented 12-month outcomes data from Spineology’s SCOUT clinical trial at the recent […]
Medacta International Appoints Francesco Siccardi its Next Chief Executive Officer
CASTEL SAN PIETRO, Switzerland–(BUSINESS WIRE)–Medacta® International, the privately held, family-owned global leader in the design of innovative joint replacement and spinal surgery products, today announced that Francesco Siccardi, a noted biomedical engineer and current Executive Vice President of Medacta International, will become the company’s next chief executive officer on November 1, 2018. After nearly twenty […]
First Patient Enrolled in Vertos Medical’s New MOTION Study of MILD® Procedure, a Minimally Invasive Treatment for Lumbar Spinal Stenosis
ALISO VIEJO, Calif., Oct. 1, 2018 /PRNewswire/ — Vertos Medical Inc., a leader in the development of innovative, minimally invasive treatments for lumbar spinal stenosis (LSS), today announced the enrollment of the first patient into its nationwide, multicenter study of its mild® procedure. The novel, prospective, randomized controlled study will use clinically validated, patient-reported outcome […]
Safe Orthopaedics continues to improve results over the first six months of 2018
Eragny-sur-Oise, 28th September 2018, 6 pm CEST – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), Safe Orthopaedics (FR0012452746 – SAFOR), a company specialized in the development and marketing of implants and single-use instruments that improve the minimally invasive treatment of spinal fracture conditions, today announced its revenue for the six-month period ending on the 30th June 2018. […]
Alphatec Accelerates Organic Innovation Machine With Addition of Key Senior Leadership and Expansion of Market Development Team
CARLSBAD, Calif., Sept. 27, 2018 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, today announced the appointment of Mark Ojeda as Executive Vice President, Cervical and Biologics. The Company also […]
SPINEWAY : Presentation of its new Mont-Blanc Evo line at the Eurospine 2018 conference
In a distributor meeting at the Eurospine 2018 conference, Spineway teams presented the advantages of the new Mont-Blanc Evo line to over twenty of their international distributors. This development of Spineway’s Mont-Blanc implants and screws allows surgeons to place screws twice as fast and for more efficient placement thanks to the new design. The instruments […]
OrthoPediatrics Corp. to Sponsor Two Medical Meetings in October
WARSAW, Ind., Sept. 27, 2018 (GLOBE NEWSWIRE) — OrthoPediatrics Corp. (NASDAQ: KIDS), a company exclusively focused on advancing the field of pediatric orthopedics, announced today it will continue its Premier sponsorship for the 72nd Annual Meeting of the American Academy for Cerebral Palsy and Developmental Medicine (“AACPDM”), which will take place October 9-13 in Cincinnati, […]
4WEB Medical, TeDan Surgical Innovations and Lattus Spine Announce Exclusive Partnership To Provide Best In Class Total Lateral Solution.
LOS ANGELES, Sept. 26, 2018 /PRNewswire/ — 4WEB Medical, the industry leader in 3D printed implant technology, announced today at the North American Spine Society (NASS) Annual Meeting that the company has entered into an exclusive partnership with both TeDan Surgical Innovations and Lattus Spine, LLC to market a turnkey, best in class, Total Lateral […]
Nvision Biomedical Technologies Launches Pedicle Screw System at NASS
Wednesday, September 26, 2018–LOS ANGELES – Nvision Biomedical Technologies, a San Antonio-based medical device and biologics manufacturer, debuted its focus® Pedicle Screw System today at the North American Spine Society (NASS) Annual Meeting, held at the Los Angeles Convention Center, September 26-29. The Nvision focus Pedicle Screw System, which received clearance from the U.S. Food […]
Stryker announces 510(k) clearance of SpineJack® Implantable Fracture Reduction System
KALAMAZOO, Michigan, USA, Sept. 26, 2018 /PRNewswire/ — Stryker announced today that it has received FDA 510(k) clearance to market the SpineJack Implantable Fracture Reduction System. The SpineJack system is indicated for use in the reduction of painful osteoporotic vertebral compression fractures. In the SAKOS clinical study, the SpineJack system demonstrated superiority to balloon kyphoplasty […]
Medicrea Hosts First-Ever Spine Artificial Intelligence Focused Meeting in Lyon, France
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea® Group (Euronext Growth Paris: FR0004178572 – ALMED), pioneering the transformation of spinal surgeries through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) proprietary software platform, services and technologies, announced today that it hosted the first-ever Spine Artificial Intelligence Focused Meeting in […]
Medicrea Announces NASS 2018 Presence And Highlights a Recently Published Study Validating Its Predictive Modelling Approach for AIS Patients
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris: FR0004178572 – ALMED, PEA-PME eligible, and OTCQX: MNRTY and MNRTF), pioneering the transformation of spinal surgeries through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) technology, announced today that the Company is attending the NASS 2018 meeting […]
RTI Surgical® Features Next-Generation Spine Implant Technologies at NASS 2018
LOS ANGELES–(BUSINESS WIRE)–RTI Surgical, Inc. (Nasdaq: RTIX), a global surgical implant company, will showcase research reinforcing the strength of its spine portfolio, notably the Fortilink® IBF series featuring TETRAfuse® 3D Technology, at the North American Spine Society’s (NASS) 33rd Annual Meeting in Los Angeles, September 26-29. Additionally, RTI will feature its full line of hardware, […]
Aurora Spine Participating at North American Spine Society Annual Meeting
CARLSBAD, Calif., Sept. 26, 2018 (GLOBE NEWSWIRE) — Aurora Spine Corporation (TSXV: ASG) announced today that it will be participating at the 2018 North American Spine Society (“NASS”) Annual Meeting, September 26-29, 2018 at the Los Angeles Convention Center, Los Angeles, California at Booth 1625. The North American Spine Society is a global multidisciplinary medical […]
Alphatec Advances Clinical Distinction With OsseoScrew® Clearance
RLSBAD, Calif., Sept. 26, 2018 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, today announced that it has received 510(k) clearance from the U.S. Food & Drug Administration (FDA) for […]
SeaSpine Announces Launch of Regatta™ Lateral System
CARLSBAD, Calif., Sept. 26, 2018 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the launch of the Regatta™ Lateral System, featuring NanoMetalene® technology. Regatta is a comprehensive, minimally invasive lateral interbody fusion system designed to efficiently and reproducibly […]
K2M to Showcase First-of-its-Kind 3D-Printed Expandable Interbody System MOJAVE™ PL 3D at NASS 2018
LEESBURG, Va., Sept. 26, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced that it will showcase its first-to-market MOJAVE™ PL 3D Expandable Interbody System featuring Lamellar 3D Titanium Technology™ and several […]
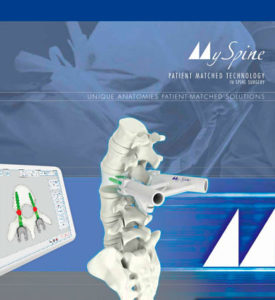
Medacta Appoints General Manager for U.S. Spine Business and Announces Full-Market Release of Customizable MySpine MC Surgical Guides for Posterior Lumbar Fusions
LOS ANGELES–(BUSINESS WIRE)–Medacta® International, the family-owned global leader in innovative joint replacement and spinal technologies, announces today the appointment of long-time commercial leader Daniel Abromowitz as General Manager, Spine Division for Medacta USA. In addition, the company announced its MySpine Midline Cortical Pedicle Screw Placement Guide (MySpine MC), a patient-matched technology for posterior lumbar fusions, […]
Centinel Spine Announces STALIF C® Integrated Interbody™ Milestone Usage in Australia
NEW YORK, Sept. 26, 2018 /PRNewswire/ — Centinel Spine, LLC announced today that Dr. Arvind Dubey, MD, FRACS, Neurosurgeon and Spine Surgeon at Hobart Brain and Spine Centre in Hobart, Tasmania, Australia, performed a milestone procedure implanting his 1,000th level of the STALIF C-Ti™ Anterior Cervical Integrated Interbody® device. The procedure was a 2-level ACDF […]
Renovis Surgical Receives FDA Clearance for Tesera Trabecular Technology 3D-Printed Lateral Interbody Fusion System
REDLANDS, Calif. and AUSTIN, Texas, Sept. 26, 2018 /PRNewswire/ — Renovis Surgical Technologies, Inc. today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the Tesera® X Lateral Interbody Fusion System. Tesera X is the latest product to utilize Renovis’ proprietary 3D-printed porous architecture, which is made […]
DePuy Synthes Expands Spine Portfolio with Nerve Assessment Platform Designed to Identify & Avoid Nerves During Spine Surgery
Los Angeles – September 26, 2018 – Johnson & Johnson Medical Devices Companies* announced today that DePuy Synthes** is introducing SENTIO MMG, a first-of-its-kind digital mechanomyography platform designed to assess nerve status and identify and avoid peripheral nerves during spine surgery. SENTIO MMG enables motor nerve monitoring for a variety of spine procedures, including non-fusion procedures […]
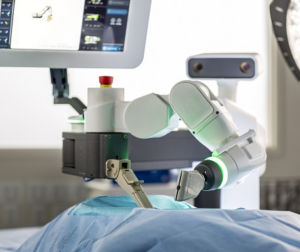
Globus Medical Sponsors NASS 2018, Hosts In-Booth Presentations on ExcelsiusGPS®
AUDUBON, Pa., Sept. 25, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, will feature its ExcelsiusGPS® revolutionary robotic guidance and navigation system along with its latest advancements in expandable interbody technology at the North American Spine Society (NASS) Annual Meeting being held September 26-29, 2018 in Los Angeles, CA. Globus […]
NuVasive To Demonstrate Pulse™ Surgical Automation Platform At NASS 2018 Annual Meeting
SAN DIEGO, Sept. 25, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ :NUVA ), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that live demonstrations of the Pulse™ surgical automation platform with integration of the Siemens Healthineers’ Cios Spin* cutting-edge mobile 3D-imaging will be held throughout the North American Spine Society (NASS) […]
SurGenTec’s ALARA™ Access Needle Awarded Best Spine Technology for 2018
BOCA RATON, Fla.–(BUSINESS WIRE)–SurGenTec ® LLC, a South Florida based medical device company has been awarded the 2018 Orthopedics This Week Spine Technology Award and will be formally receiving their award during the 2018 North American Spine Society meeting. This year’s meeting will be held Sept. 26th -28th in Los Angeles, CA. The awarded technology from SurGenTec’s product line is […]