FORT LAUDERDALE, Fla., July 31, 2025 /PRNewswire/ — Proximal junctional kyphosis (PJK) and failure (PJF) remain the bane of spinal deformity surgeons’ existence. While many methods (i.e. tether, cement augmentation, “ideal” alignment) have been proposed to decrease rates of PJK/PJF, they have not significantly moved the needle in this regard. Among the risk factors for PJK/PJF, the stiffness of the construct, influenced by choice of rods, is one that surgeons can control. One major limitation of rods for spinal deformity corrections is that single diameter rods (i.e. 5.5mm, 6.0mm, 6.35mm) cannot provide the needed strength to correct deformities while also providing a biomechanically friendly transition at the junction of constructs.
According to Dr. Alekos Theologis, Associate Professor, UCSF Orthopaedic Surgery, “Most recently, Spinal Resources Inc. (SRI) has developed unique rods, termed “Bezier surface-smoothed transition rods”, that vary in stiffness based on local rod section diameter. Bezier rods are patient-specific and can be designed with greater stiffness where additional stiffness is needed (i.e. across 3-column osteotomies, multilevel posterior column osteotomies, lumbosacral junction) while maintaining flexibility near the junctional vertebrae so as to promote a biomechanical “soft landing” at the junction between the instrumented spine and native/non-instrumented spine.” The biomechanical benefits of this rod have now been published in the Spine Deformity Journal in July 2025.
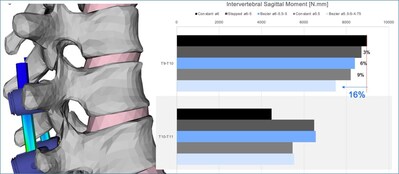
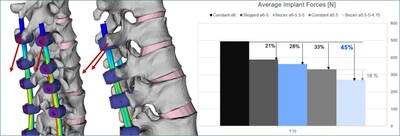
Dr. Theologis continues, “Specifically, in this finite element analysis, it was found that Bezier rods provided smoother load transitions and better offloading of proximal segments compared to constant diameter rods. The highest moment sustained by the segment adjacent to the instrumentation was observed with the constant 6mm rods, while the Bezier 5.5-5-4.75mm rod showed the lowest moment, indicating reduced stress of 16% on the upper adjacent vertebrae (Figure 1). Similarly, the Bezier rods were more effective in offloading pedicle screws up to 45% with respect to the stiffer rod construct. (Figure 2).”
Dr. Saeed S. Sadrameli, M.D., Board Certified in Neurosurgery, specializing in endoscopic and patient-specific minimally invasive/robotic spine surgery, Advent Health points out, “The underlying design principle applies equally to long, complex deformity constructs and shorter segmental fusions, where junctional stress and implant fatigue remain a concern. The Bezier technology marks a shift toward biomechanically optimized and personalized instrumentation, delivering stiffness and compliance precisely tailored to each region of the spine.”
Dr. Sadrameli continues, “Tailored to each patient’s anatomy and surgical plan, the Bezier rod shows strong potential to mitigate common mechanical complications in spinal fusion, such as Proximal Junctional Kyphosis (PJK), by optimizing load-sharing between the rod and the spine at critical spinal regions.”
According to Bernie Bedor, SRI President and CEO, “These are extremely promising and exciting findings that lend strong support for and validate SRI’s Bezier Parametric Curve Transition Rods’ potential in reducing the risk of PJK/PJF following adult and pediatric spinal deformity operations.”
About Spinal Resources Inc.
Spinal Resources Inc.® is a Ft. Lauderdale, Florida based spinal medical device company that supports cost-effective patient care with innovative mechanical and bio-mechanical products to alleviate pain, shorten recovery time, restore health, and extend quality of life.
SOURCE Spinal Resources Inc.
