Boca Raton, FL, Sept. 27, 2024 (GLOBE NEWSWIRE) -- 33 Medical, Inc., a leader in cutting-edge spinal health innovations, is proud to announce that its NuvoDisc® medical device has been awarded the prestigious 2024 Best Technology in Spine Award. This accolade recognizes the NuvoDisc® as a revolutionary advancement in the treatment of … [Read More...] about 33 Medical Wins 2024 Best Technology in Spine Award for Innovative NuvoDisc® Device
Main Content
FEATURED ARTICLE
BREAKING NEWS

Over 7,500 Procedures Completed in Two Years with Centinel Spine’s prodisc® C Vivo and prodisc C SK Cervical Total Disc Replacement System Since U.S. Launch
WEST CHESTER, Pa., Sept. 26, 2024 /PRNewswire/ -- Centinel Spine®, LLC ("the Company"), the leading global medical device company focused exclusively on treating cervical and lumbar spinal disease with the most complete and clinically-proven total disc replacement (TDR) technology platform in the world (prodisc®), today announced the completion of 7,500 U.S. cases with the prodisc C … [Read More...] about Over 7,500 Procedures Completed in Two Years with Centinel Spine’s prodisc® C Vivo and prodisc C SK Cervical Total Disc Replacement System Since U.S. Launch

Synergy Spine Solutions announces completion of patient enrollment in the Synergy Disc® 2-Level IDE clinical trial
LOUISVILLE, Colo., Sept. 25, 2024 /PRNewswire/ -- Synergy Spine Solutions Ltd, an innovative orthopedic medical device developer focused on artificial cervical disc replacement, today announced that it has completed patient enrollment in its U.S. 2-Level IDE clinical trial. Conducted under a U.S. Investigational Device Exemption (IDE), the 2-Level trial will evaluate the safety … [Read More...] about Synergy Spine Solutions announces completion of patient enrollment in the Synergy Disc® 2-Level IDE clinical trial

Medtronic expands AiBLE™ spine surgery ecosystem with new technologies and Siemens Healthineers partnership
GALWAY, Ireland and CHICAGO, Sept. 25, 2024 /PRNewswire/ -- Medtronic plc (NYSE: MDT), a global leader in healthcare technology, announced today at the North American Spine Society (NASS) 39th Annual Meeting in Chicago the commercial launch of several software, hardware, and imaging innovations. These enhancements are designed to advance AiBLE™, the Medtronic smart ecosystem of innovative … [Read More...] about Medtronic expands AiBLE™ spine surgery ecosystem with new technologies and Siemens Healthineers partnership

elliquence and Implanet Form Partnership for US Distribution of Ultrasonic Bone Scalpel Olea in Endoscopic Spine Market
Spine tech innovators elliquence and Implanet to exhibit at NASS in Chicago, September 25-28, 2024 (elliquence LLC Booth 2419 Implanet Booth 2412) elliquence LLC, specializing in advanced minimally invasive surgical technologies, and Implanet (Euronext Growth: ALIMP, FR0013470168, eligible for PEA-PME), a medical technology company specializing in implants for orthopedic surgery and … [Read More...] about elliquence and Implanet Form Partnership for US Distribution of Ultrasonic Bone Scalpel Olea in Endoscopic Spine Market

OrtoWay-US Inc. Announces Granting of U.S. Patent for OrtoWell® Distractor System for Spinal Surgery Corpectomy
HORSHAM, Pa., September 23, 2024 (Newswire.com) - OrtoWay-US Inc., inventors and developers of the world’s first hydraulically-powered OrtoWell® Distractor for spinal surgery, announced today the receipt of a new patent from the U.S. Patent Office for its OrtoWell® Distractor system. The patent, numbered US 12,023017 B1, entitled “Apparatus, Methods and Systems for Spine Surgery,” was issued … [Read More...] about OrtoWay-US Inc. Announces Granting of U.S. Patent for OrtoWell® Distractor System for Spinal Surgery Corpectomy

Cerapedics’ i-FACTOR P-15 Peptide Enhanced Bone Graft, a Trusted Choice in Bone Graft Solutions, Now Approved for Expanded Indications for Use
WESTMINSTER, Colo., Sept. 24, 2024 /PRNewswire/ -- Cerapedics Inc., a global, commercial-stage orthopedics company dedicated to redefining the standard of care for bone repair, today announced the U.S. Food and Drug Administration (FDA) approval for an expansion to the indications for use and labeling for i-FACTOR P-15 Peptide Enhanced Bone Graft. With this label expansion, i-FACTOR … [Read More...] about Cerapedics’ i-FACTOR P-15 Peptide Enhanced Bone Graft, a Trusted Choice in Bone Graft Solutions, Now Approved for Expanded Indications for Use

NGMedical presents its MOVE®-C Cervical Artificial Disc Prosthesis at NASS and Eurospine
NONNWEILER, SAARLAND, GERMANY, September 18, 2024 /EINPresswire.com/ -- NGMedical GmbH, a medical device manufacturer exclusively focused on creating innovative technologies for spinal application, will present its unique articulating viscoelastic cervical artificial disc MOVE®-C as well as further unique spinal solutions at this year’s annual NASS meeting in Chicago and Eurospine in … [Read More...] about NGMedical presents its MOVE®-C Cervical Artificial Disc Prosthesis at NASS and Eurospine

Spineology® Announces Organization Rebrand
ST. PAUL, Minn.--(BUSINESS WIRE)--Spineology Inc., the pioneer of Conform and Expand™ Technology, today unveiled a new brand and visual identity, logo, and product positioning as part of an extensive rebranding initiative. This new brand defines Spineology’s flagship interbody fusion portfolio, OptiMesh®, and establishes it within a category of its own. OptiMesh is the only conforming, patient … [Read More...] about Spineology® Announces Organization Rebrand

SIGNUS Receives FDA Approval for TETRIS® ST & TETRIS® R ST PLIF Cages
Alzenau, 09/12/2024 – SIGNUS Medizintechnik GmbH is proud to announce the successful FDA approval of TETRIS ® ST and TETRIS ® R ST PLIF cages. These cages, made from structured titanium (ST), combine cutting-edge technology and materials, further expanding the innovative SIGNUS spinal surgery portfolio. SIGNUS is dedicated to continuously developing products with passion and precision, offering … [Read More...] about SIGNUS Receives FDA Approval for TETRIS® ST & TETRIS® R ST PLIF Cages

SC MEDICA wins 2024 Best Technology in Spine Award for its FFX® Facet FiXation system
Strasbourg, France, September 18, 2024 – SC MEDICA, a leader in innovative spinal solutions, is pleased to announce that it has received the prestigious 2024 Best Technology in Spine Award, organizedby Orthopedics This Week, for its innovative FFX® Facet Fixation system. This award honors the relentless dedication of the engineers, inventors, and medical professionals who strive to advance … [Read More...] about SC MEDICA wins 2024 Best Technology in Spine Award for its FFX® Facet FiXation system

Nexxt Spine Launches Struxxure® Modular Cervical System
NOBLESVILLE, IN, UNITED STATES, September 18, 2024 /EINPresswire.com/ -- Nexxt Spine, LLC is proud to announce the launch of the Struxxure® Modular Cervical System (MCS). Struxxure MCS is built on more than ten years of proven clinical use of the Struxxure Anterior Cervical Plate system. MCS is designed to offer intraoperative flexibility and modularity, including two-, three-, and … [Read More...] about Nexxt Spine Launches Struxxure® Modular Cervical System
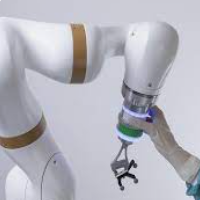
eCential Robotics Appoints Experienced Leadership Team to Drive U.S. Market Expansion
NASHVILLE, Tenn., Sept. 19, 2024 /PRNewswire/ -- eCential Robotics, a growth company transforming bone surgery by designing and manufacturing an open robotic and navigation system with unique capabilities, is pleased to announce the addition of Lisa Jacobs and Matthieu Ville to its leadership team. These strategic hires come as part of eCential Robotics' … [Read More...] about eCential Robotics Appoints Experienced Leadership Team to Drive U.S. Market Expansion

Avicenna.AI lands FDA clearance for AI tool that detects cervical spine fracture
La Ciotat, FRANCE – September 19, 2024 – Medical imaging AI company Avicenna.AI today announced that it has received 510(k) clearance from the US Food and Drug Administration for its CINA-CSpine tool. Using a combination of deep learning and machine learning technologies, the company develops AI solutions that automatically detect and prioritize life-threatening conditions within … [Read More...] about Avicenna.AI lands FDA clearance for AI tool that detects cervical spine fracture

Amnovis Marks Milestone of 50,000 Implants Delivered with Game-Changing Titanium 3D Printing Process
Aarschot, Belgium – September19, 2024.--Amnovis, a leader in 3D printed titanium implant manufacturing, has reached a significant milestone, delivering over 50,000 titanium implants since 2021 using its groundbreaking, heat treatment-free 3D printing process. This innovative technology is transforming the production of spinal, orthopedic, and cranio-maxillofacial (CMF) implants, allowing customers … [Read More...] about Amnovis Marks Milestone of 50,000 Implants Delivered with Game-Changing Titanium 3D Printing Process

SurGenTec Wins Two Spine Technology Awards for 2024
BOCA RATON, Fla.--(BUSINESS WIRE)--SurGenTec LLC, a privately held medical device company, was awarded two 2024 Spine Technology Awards, hosted by Orthopedics This Week. These prestigious awards recognize the company’s standout innovations in a highly competitive field of submissions. The company's TiLink-P Posterior Sacroiliac (SI) Joint Fusion System received the Gold Award, while the OsteoFlo … [Read More...] about SurGenTec Wins Two Spine Technology Awards for 2024

Life Spine Announces Published Article on Discectomy Performance During Transforaminal Lumbar Interbody Fusion Approach Using Endoscopic Technique vs MIS Tubular Approach
HUNTLEY, Ill.--(BUSINESS WIRE)--Life Spine, Inc., a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that Spine, an international, peer-reviewed journal has published a new study examining Discectomy performance during TLIF approaches using an Endoscopic technique versus a MIS Tubular approach1. The … [Read More...] about Life Spine Announces Published Article on Discectomy Performance During Transforaminal Lumbar Interbody Fusion Approach Using Endoscopic Technique vs MIS Tubular Approach

First Augmented Reality Spine Surgery Performed in Michigan Using OnPoint AR Spine System
CONCORD, Mass., Sept. 17, 2024 /PRNewswire/ -- Neurosurgeons in Detroit became the first in in the State of Michigan, to use the OnPoint AR Spine System on August 8, 2024. The OnPoint AR Spine System is an augmented-reality based platform that offers a cost-effective and highly accurate alternative to current navigation and robotic systems. Neurosurgeon … [Read More...] about First Augmented Reality Spine Surgery Performed in Michigan Using OnPoint AR Spine System

Neo Medical Secures Landmark Equity Investment of $68 million to Accelerate Global Growth in Spine Surgery
LAUSANNE, Switzerland--(BUSINESS WIRE)--Neo Medical SA (Neo), a Swiss technology company specializing in spine surgery, has announced the successful completion of its Series B financing round, including the conversion of existing shareholder loans. With this financing round, Neo has secured an additional equity investment of up to $68 million (CHF 58 million) to accelerate its global … [Read More...] about Neo Medical Secures Landmark Equity Investment of $68 million to Accelerate Global Growth in Spine Surgery

X-Bolt Receives Prestigious ‘Best Technology in Spine’ Award from Orthopedics This Week
DUBLIN, Sept. 16, 2024 /PRNewswire/ -- X-Bolt Orthopedics, an emerging innovator of bone fixation implants in orthopedics, has been awarded the Best Technology In Spine Award for 2024/2025 by Orthopedics This Week. This award honors the unsung heroes in the world of medical technology — the inventors, engineers, and innovators who dedicate countless hours to advancing … [Read More...] about X-Bolt Receives Prestigious ‘Best Technology in Spine’ Award from Orthopedics This Week
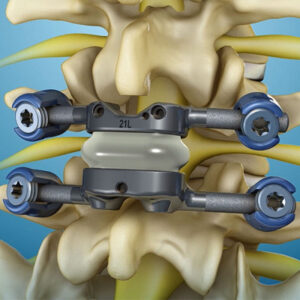
Premia Spine Wins Best Paper Award at SMISS 2024 for Study on Lumbar Facet Arthroplasty and the TOPS™ System
NORWALK, Conn., Sept. 13, 2024 /PRNewswire-PRWeb/ -- Premia Spine, a leader in motion-preserving spinal technologies, is proud to announce that its study, titled "Lumbar Facet Arthroplasty: An Analysis Comparing Two-Year Outcomes from a Prospective Randomized Clinical Trial Among Patients with Unstable vs. Stable Spondylolisthesis," has been awarded the Best Paper Award at the Society for … [Read More...] about Premia Spine Wins Best Paper Award at SMISS 2024 for Study on Lumbar Facet Arthroplasty and the TOPS™ System

Stryker announced the addition of two new products recently acquired from 4WEB Medical
Stryker (NYSE: SYK), a global leader in medical technology, has announced the expansion of its Foot & Ankle portfolio with the addition of two new products – the Osteotomy Truss System™ (OTS) and the Ankle Truss System™ (ATS). These products were recently acquired from 4WEB Medical. Stryker will showcase the OTS and ATS, along with the newly acquired Artelon portfolio and other offerings, at … [Read More...] about Stryker announced the addition of two new products recently acquired from 4WEB Medical

Zavation Medical Products, LLC announces launch of Varisync® ALIF System
FLOWOOD, Miss., Sept. 10, 2024 /PRNewswire/ -- Zavation Medical Products, LLC ("Zavation"), an innovator in spinal device technology, proudly announces the launch of its latest product—the Varisync® ALIF System. Engineered to streamline ALIF procedures, the Varisync system offers customization and efficiency with two spacer fixation options: anchors and screws. Innovative … [Read More...] about Zavation Medical Products, LLC announces launch of Varisync® ALIF System




![[GS Medical] Banner Ad_2-2](https://thespinemarketgroup.com/wp-content/uploads/2024/02/GS-Medical-Banner-Ad-1_ver.2-jpg-750x100.webp)












