BETHLEHEM, PA, UNITED STATES, August 27, 2025 /EINPresswire.com/ — In a bold move to elevate performance and accelerate innovation across the MedTech industry, the combined organization of Intech, Tyber Medical, and Resolve Surgical Technologies today announced a strategic reorganization into two distinct operational business units:
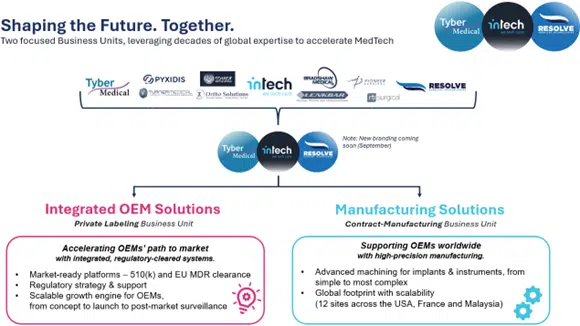
• Manufacturing Solutions (‘MS BU’) – Supporting OEMs worldwide with design and manufacturing of mission-critical components and complex assemblies for broad Medtech devices from implants to instruments to enabling technologies.
• Integrated OEM Solutions (’IOS BU’) – Accelerating OEMs’ path to market with integrated, regulatory-cleared systems, effectively supporting product launches within 6-12 months with all relevant regulatory approvals
As part of this transformation, the company welcomes two highly accomplished executives: Mohamed Chakroun, Chief Operating Officer of Manufacturing Solutions with deep expertise in operational excellence, and Andrew Thomas, Chief Operating Officer of Integrated OEM Solutions with a proven track record in driving end-to-end commercialization of integrated, regulatory-cleared systems. With this transition, Laurent Pruvost will move from day-to-day operations into a strategic role on the Board of Directors.
In addition, all Commercial and Innovation efforts are now unified under the leadership of Jeff Tyber, Group President. Through this alignment, NewCo will be able to put its customers at the center of all activities: creating a powerful engine for growth, streamlining the customer journey, providing a comprehensive offering matching market needs and accelerating innovation for customers to new levels.
“Our transformation into two focused operational business units is about more than efficiency, it’s about delivering the full spectrum of solutions that OEMs are looking for,” said Olivier Wolber, CEO of the combined entity. “With Mohamed’s customer-centric expertise in production and Andrew’s track record in delivering OEM solutions at speed, we are poised to raise the bar for collaboration, innovation, and speed in MedTech. As we put customers more and more at the center of everything we do, their leadership will help us take customer satisfaction to the next level across the globe.”
This reorganization reflects the company’s commitment to deliver sharper focus, faster execution, and deeper integration, enabling OEM partners to innovate with confidence and bring life-changing devices to patients more efficiently than ever before. As the company gears up to unveil its new brand in the coming weeks, this transformation sets the stage for an exciting next chapter, defined by a commitment to accelerate MedTech and elevate patient care.
About Intech, Tyber and Resolve
With headquarters in Bethlehem, PA, and operations across multiple continents, the new organization is a global solutions provider for the MedTech industry. Through its Manufacturing Solutions and Integrated OEM Solutions business units, the company supports leading OEMs in delivering breakthrough medical devices that improve patient outcomes worldwide.
Francois Samson
Intech
fsamson@intech-medical.com
