Extreme lateral interbody fusion (XLIF) (NuVasive Inc., San Diego, CA) and direct lateral interbody fusion (DLIF) (Medtronic Sofamor Danek, Memphis, TN) are novel minimally invasive transpsoas approaches to the lumbar spine for performing interbody fusions. Read More
NEWS
Bone Therapeutics and Kasios develop 3D bioprinted structures
Bone Therapeutics, a Belgian biotech company specializing cell therapy for bone fracture repair, alongside French bone substitute specalist Kasios, are now working to develop a kind of ‘waffle’—one that is 3D bioprinted, and could be used to significantly improve next-generation bone disease therapy.This biotech collaboration will see Bone Therapeutics combine its proprietary ALLOB osteoblastic cell […]
Add an Apple Icon to the desktop for an easy access to our site!
To add an Apple Icon to the desktop of an iPad or iPhone device for an easy access to our site is simple and fast.When browsing online with an Apple mobile device (iPod Touch, iPad, and iPhone), just press the button shown in the following image (Image 1). Then there is the option of saving web […]
Lateral Lumbar Interbody Fusion Market in the US 2016-2020
Last week, Technavio announced the launch of report focused on the LLIF market (minimally invasive surgery recommended for treating lumbar spinal disorders such as spondylolisthesis, lumbar degenerative disc disease, scoliosis, and recurrent lumbar disc herniation). According to Technavio’s analysts forecast the lateral lumbar interbody fusion market in the US to grow at a CAGR of 7.05% […]
Surgical Robots for the Spine Market Worth $2.77 Billon by 2022: Radiant Insights, Inc.
SAN FRANCISCO, CA–(Marketwired – Feb 3, 2016) – Back conditions can result in instability and compression of the spinal nerves, causing back pain and/or radiating pain in the legs. Robotic procedures offer significant cost savings in terms of pre- and post-operation care costs and length of stay at hospitals. Technological advances and breakthroughs leverage new […]
Amendia Inc. Launches the OmegaLIF™ Expandable Interbody Cage
MARIETTA, GA (PRWEB) FEBRUARY 02, 2016–Amendia, Inc. (http://www.amendia.com), a leading provider of balanced, innovative medical device solutions for the spine industry, announces today the launch of the OmegaLIF™ (ΩLIF™) expandable lumbar interbody device, an expandable interbody cage especially designed for use in intervertebral body spinal fusion procedures. The OmegaLIF™ was created to provide a solution […]
MEDTECH Announces Two New ROSA Sales in the U.S.
MONTPELLIER, France, Feb. 02, 2016 (GLOBE NEWSWIRE) — MEDTECH (Euronext, FR0010892950 – ROSA), a company specialized in designing, developing and marketing innovative surgical assistance robots, is pleased to announce continued commercial expansion with two new sales of its ROSA™ Brain robot in the U.S., to Brigham and Women’s Hospital and the University of Colorado Medical […]
Exactech Australia Becomes Exactech Subsidiary and Ninth Direct Operation
GAINESVILLE, Fla.–(BUSINESS WIRE)–Exactech, Inc. (Nasdaq: EXAC), a developer and producer of bone and joint restoration products for extremities, knee, hip, spine and biologics, announced today the acquisition of its independent distribution partner, Exactech Australia Pty Ltd, effective Feb 1, 2016. The purchase included all the stock and assets of Exactech Australia, a Sydney-based importer and […]
EOI Announces Dr. Jonathan Lewin is First to Implant the FLXfit™ 3D Expandable Cage in New Jersey
OR AKIVA, Israel, February 1, 2016 /PRNewswire/ — Expanding Orthopedics Inc. (EOI), a privately held medical device company focused on developing and commercializing innovative expandable devices for spine surgery, is excited to announce that Dr. Jonathan Lewin of North Shore Forest Hills Hospital in New York is the first surgeon in New Jersey to adopt the […]
45 Dynamic stabilization Devices to know
Dynamic stabilization products include interspinous process spacers, pedicle screw systems with semirigid rods and facet replacement technologies (Learn about the most relevant in :www.spinemarketnews.com/dynamic).No dynamic stabilization devices have received approval from the U.S. Food and Drug Administration (FDA) for use other than as an adjunct to spinal fusion. The FDA has specified that separate approval is required for […]
We are also on Facebook! Join us!
The SPINEMarketNews Facebook group (https://www.facebook.com/SPINEMarketNewscom-275622739188641) is open to any interested person. If you are on Facebook, add this one to your groups. Also feel free to share it with your facebook friends. It is also a great forum for sharing the latest news and discussions.If you like us, please give a positive feedback.We will appreciate […]
Use of Surface-Modified Titanium in Cervical Spine Implants Set to Drive the Global Cervical Artificial Discs Market, Says Technavio
LONDON–According to the latest research report released by Technavio, the global cervical artificial discs marketis predicted to reach over USD 1,082 million by 2020.This report titled ‘Global Cervical Artificial Discs Market 2016-2020‘, provides an in-depth analysis of the market in terms of revenue and emerging trends. This market study also presents up to date analysis […]
Amedica to Present Research Supporting Favorable Silicon Nitride Findings at the Orthopaedic Research Society Annual Meeting
SALT LAKE CITY, Jan. 28, 2016 (GLOBE NEWSWIRE) — Amedica Corporation (Nasdaq:AMDA), a company that develops and commercializes silicon nitride ceramics as a biomaterial platform, is pleased to announce all four submissions to the Orthopaedic Research Society (“ORS”) Annual Meeting were accepted for presentation occurring March 5-8, 2016 in Orlando, Florida. “Our presentations will demonstrate […]
LDR Announces First Peer-Reviewed Publication of Five-Year Results for Mobi-C Cervical Disc
AUSTIN, Texas, Jan. 27, 2016 (GLOBE NEWSWIRE) — LDR Holding Corporation (Nasdaq:LDRH), a global medical device company focused on designing and commercializing novel and proprietary surgical technologies for the treatment of patients suffering from spine disorders, today announced that “Subsequent surgery rates after cervical total disc replacement using a Mobi-C® Cervical Disc Prosthesis versus anterior […]
Paradigm Spine, LLC Announces Publication Of 5-Year Long-Term Results Of Level 1 Data For coflex® Interlaminar Stabilization® Compared To Fusion
NEW YORK, Jan. 27, 2016 /PRNewswire/ — Paradigm Spine, LLC, a leader in providing solutions for the treatment of lumbar spinal stenosis announces publication on January 26, 2016 by Michael Musacchio, M.D., et al of “Evaluation of Decompression and Interlaminar Stabilization Compared with Decompression and Fusion for the Treatment of Lumbar Spinal Stenosis: 5-year Follow-up […]
IMPLANET: 2015 Sales Growth (Excluding Hip): +6% to €6.7 Million
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–Regulatory News: IMPLANET (Paris:IMPL) (Euronext: IMPL, FR0010458729, PEA-PME eligible), a medical technology company specializing in vertebral and knee-surgery implants, today announces its sales for the 4th quarter and full year to 31 December 2015. Ludovic Lastennet, CEO of Implanet, says: “Our annual performance shows buoyant sales growth in Spine, thanks to market […]
Amedica Enhances Valeo II™ Product Family With the Addition of Its Second Generation Cervical System
SALT LAKE CITY, Jan. 25, 2016 (GLOBE NEWSWIRE) — Amedica Corporation (Nasdaq:AMDA), a company that develops and commercializes silicon nitride ceramics as a biomaterial platform, is pleased to announce the release of its Valeo II™ C interbody fusion device system. The second generation cervical system will be commercially available mid-February 2016. The Valeo II C […]
SpineGuard Expands Senior Management Team
PARIS and SAN FRANCISCO, Jan. 25, 2016 – SpineGuard (FR0011464452 – ALSGD), a fast-growing company that develops and markets disposable medical devices designed to make spine surgery safer, today announced the addition of an experienced spine industry executive to the company’s senior management team. Steve McAdoo has joined SpineGuard as Vice President of Business Development […]
How Medical Device Companies Can Strategically Sell to Customers
(January 2016 Medical Device Business )- Economic pressures on the health system have been mounting for years, pushed by demographics, shrinking reimbursements, increasingly expensive therapies, and, ultimately, payer consolidation and reforms. Hospitals are the fulcrum and the pinch point for these pressures. Medtech companies no longer can afford to focus on clinical benefits without seeking other avenues […]
Spine Market Overview: Which are the Spinal Companies?
We have updated the list of the most important Spinal Companies. Please visit: www.spinemarketnews.com/companies According to MarketsandMarkets, the Spinal market is expected to reach USD 15.73 Billion by 2020, growing at a CAGR of 5.6% from 2015 to 2020. Medtronic, plc (U.S.), DePuy Synthes (U.S.), Stryker Corporation (U.S.), NuVasive, Inc. (U.S.), Zimmer Biomet Holdings, Inc. (U.S.), […]
K2M Receives CE Mark for Its Next-Generation RHINE(TM) Cervical Disc System
LEESBURG, Va., Jan. 21, 2016 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (Nasdaq:KTWO), a global medical device company focused on designing, developing and commercializing innovative and proprietary complex spine technologies and techniques, today announced it has received a CE Mark for the RHINE™ Cervical Disc System, allowing for sale and distribution of the product within […]
LDR Holding Partners With Poly-Shape to Build Innovative Spinal Implants With Additive Manufacturing Technology
AUSTIN, Texas, Jan. 21, 2016 (GLOBE NEWSWIRE) — LDR Holding Corporation (Nasdaq:LDRH), a global medical device company focused on designing and commercializing novel and proprietary surgical technologies for the treatment of patients suffering from spine disorders, today announced that, through its wholly-owned subsidiary LDR Médical, SAS, it has made a minority investment in Poly-Shape, SAS, […]
SpineFrontier, Inc. Announces Newest Member to Join its Board of Directors
BOSTON, MA (PRWEB) JANUARY 21, 2016=SpineFrontier (http://www.spinefrontier.com) announced today that Jerry Goldstein, Founder and former CEO of Advanced Magnetics (AMAG), has joined the SpineFrontier Board of Directors. Jerry is a pioneer in the MRI industry. He started AMAG in 1981 and took the company public in 1986. AMAG is best known for its MR imaging […]
Centinel Spine™ Announces the FDA Clearance and First Implantation of ALTOS™
NEW YORK, Jan. 21, 2016 /PRNewswire/ — Centinel Spine, Inc., (Centinel Spine) announces the U.S. Food and Drug Administration (FDA) clearance of ALTOS™, a posterior cervical thoracic stabilization system indicated for use in either the lateral masses of the cervical spine or the pedicles of the cervical-thoracic spine. The first surgical implantation of the ALTOS […]
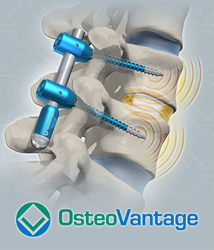
Orthopedic Innovator OsteoVantage, Inc. Awarded SBIR Grant by National Science Foundation
Fayetteville, Arkansas (PRWEB) JANUARY 20, 2016-OsteoVantage, Inc. has recently been awarded a Small Business Innovation and Research (SBIR) Phase I grant by the National Science Foundation. The $150,000 grant, awarded January 10, 2016, supports the development of enhancements to the company’s INDOS™ system of spinal instrumentation. The INDOS™ system simultaneously facilitates both mechanical stabilization of […]
Globus Medical Announces Litigation Settlement Agreement
AUDUBON, Pa., Jan. 19, 2016 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal implant manufacturer, today announced DePuy Synthes Products, Inc., DePuy Synthes Sales, Inc. and Globus Medical, Inc. have settled four patent infringement lawsuits concerning spinal implant technologies. The terms of the settlement have not been disclosed but were to the parties’ […]
SI-BONE, Inc. Announces European Expansion with Formation of SI-BONE UK Ltd and Key Additions to Management Team
SAN JOSE, Calif., Jan. 20, 2016 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the use of the iFuse Implant System®, a minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced the expansion of its European operations with the formation of SI-BONE UK Ltd, along […]