ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division will introduce the Aero-C Cervical Stability System, the only straight forward ACDF device that offers uniform compression across the interbody space, at the American Academy of Orthopaedic Surgeons (AAOS) annual meeting, March 2-4, 2016, in Orlando, Fla. (booth number 3845). Aero-C is the latest addition to Stryker’s Aero platform of […]
NEWS
Precision Spine™ Reaffirms Commitment to Made-in-the-USA Manufacturing
PEARL, Miss.–(BUSINESS WIRE)–While many U.S.-headquartered medical device manufacturers are moving their manufacturing operations to China and other overseas locations, Precision Spine, Inc. has reaffirmed its commitment to Made-in-the-USA manufacturing with the addition of a third building to its Pearl, Mississippi campus, expanding the total manufacturing facility footprint by 30% to just under 30,000 square feet […]
50 HeadHunters to Know
Are you looking for a job in the Medical Devices or Spinal market? Is your Company searching for Spine professionals? This list of 50 Recruiters can help you. We have included relevant Headhunters focused or specialised in these fields.We hope that it will work for you! Please find the list of 50 Headhunters: https://thespinemarketgroup.com/headhunters/ Why Headhunters are Important? Executive recruiters—or […]
Atlas Spine Licenses True Position® Pivoting Spacer System Patents to Spine Wave
JUPITER, FL–(Marketwired – February 29, 2016) – Medical device company Atlas Spine, Inc. today announced the exclusive license of its True Position® Pivoting Spacer system patents and transfer of related assets to Spine Wave, Inc. The novel and patented Center Pivot Point Technology of the True Position® Spacer provides a powerful and controlled mechanical advantage, […]
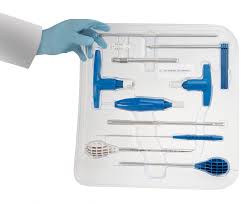
Single-use Sets are gaining traction in Spine
Single-use sets are gaining traction in Spine since their introduction in late 2014 . Physicians, like most human beings, are creatures of habit, preferring the familiar over the unknown. Most clinicians, for example, resisted the idea of electronic medical records in the late 1990s, deeming the time-saving tool an impersonal addition to their practices and an affront […]
Tyber Medical Announces a Statistically Significant Reduction of Bacteria
MORRISTOWN, N.J., Feb. 24, 2016 /PRNewswire/ — Tyber Medical, LLC, a privately held medical device company focused on developing innovative orthopedic and spine devices for private label opportunities, announces findings from research ongoing at Northeastern University that demonstrate a statistically significant reduction in bacteria on implant surfaces processed with BioTy™, a Modified Surface Treatment. The […]
Amedica to Present Advantages of Silicon Nitride Implants at 2016 Brazilian Spine Congress
SALT LAKE CITY, Feb. 23, 2016 (GLOBE NEWSWIRE) — Amedica Corporation (Nasdaq:AMDA), a company that develops and commercializes silicon nitride ceramics as a biomaterial platform, is pleased to announce its participation in the 2016 Brazilian Spine Congress occurring March 3-4, 2016 in Sao Paulo, Brazil. “Brazil has been an emerging growth opportunity for us since achieving […]
40 Cervical Artificial Discs
40 Cervical Artificial Discs (https://thespinemarketgroup.com/adr/) list include most of these devices that are already in the market and even some products that have discontinued. The Cervical Artificial Disc Market The global cervical artificial discs market to grow at a CAGR of 22.53% during the period 2016-2020.An artificial disc is a prosthetic device inserted between the […]
RTI Surgical® Announces Limited Launch and First Implantation of Unison™-C System
RTI Surgical® Inc. (RTI) (Nasdaq: RTIX), a leading global surgical implant company, is pleased to announce the limited launch of the Unison™-C System, an anterior cervical fixation system. The first human implantation of the Unison-C System took place during an anterior cervical discectomy and fusion (ACDF) procedure last week. The Unison-C System will expand RTI […]
50 Minimally Invasive Spine Fusion Systems
Learn about the 50 Minimally Invasive Spine Fusion Systems:thespinemarketgroup.com/mis/. MIS is an increasing trend today in the Spinal Market. The advantages of MIS over traditional surgery are shorter hospital stays, lower infection risk, reduced bleeding, shorter scars, and earlier return to work. MIS is currently in its early stages and is not uniformly covered by insurance […]
Dealing with Commoditization and Price in Spine
Dealing with commoditization and price in Spine has become in recent years, the key issues and most important trends for manufacturers and distributors in this spinal industry. This process fundamentally changes the nature of competition in the spine segment. Instead of competing on attributes such as brand, and quality, products in a commoditized industry compete largely along […]
LDR Holding Corporation Reports Record Revenue for Fourth Quarter and Fiscal Year 2015
AUSTIN, Texas, Feb. 17, 2016 (GLOBE NEWSWIRE) — LDR Holding Corporation (NASDAQ:LDRH), a global medical device company focused on designing and commercializing novel and proprietary surgical technologies for the treatment of patients suffering from spine disorders, today reported its financial results for the fourth quarter and fiscal year ended December 31, 2015. Fourth Quarter and Fiscal […]
ulrich medical USA® Announces Strong Financial Performance of U.S. Spine Business
ST. LOUIS, Feb. 17, 2016 /PRNewswire/ — ulrich medical USA Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States, today reported a fifth consecutive year of profitable, double-digit growth for the U.S. spine business and a 20% top line growth for the year ending December 31, 2015. Equally […]
Hospital Corporation of America (HCA) Awards National Vendor Contract to SpineGuard
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–SpineGuard (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices designed to make spine surgery safer, today announced that it has been awarded a national vendor contract with the HCA network of hospital systems. The HCA network includes over 167 hospitals and 113 free-standing surgery centers in […]
Titan Spine Continues Substantial Growth with 51% Sales Increase in 2015
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced the Company continues to achieve substantial growth, with a 51% increase in sales of its Endoskeleton® interbody fusion devices in 2015 compared to the previous year. The Company has also reported over $33.5 million in […]
Single-use Spinal Products? A Succesful Trend?
Single-use or disposable instruments, kits and products are not new to the medical and biopharma markets but they are already in the Spinal Business. This technology will likely succeed as result of cost reduction demands by hospitals and healthcare reimbursement agencies and insurance companies. In 2010 Safe Orthopaedics, a French medical technology company was founded and soon started to market the Single-use […]
All Alphatec Spine needs is Time!
Alphatec Holdings (NASDAQ:ATEC), is a perfect example of a high reward, high risk investment opportunity. There is a ton of debt on the balance sheet, that needs to get refinanced in the next 3-4 months, and the company is still unprofitable on an EPS basis. However, the company is very close to being at breakeven […]
Safe Orthopaedics: 2015 revenue: +23% to €2.5 million
SAFE ORTHOPAEDICS (Paris:SAFOR) (Euronext: FR0012452746 – SAFOR), a company developing and marketing an innovative range of sterile implants combined with their single-use surgical instruments, presents its business update for the year to December 31, 2015. Yves Vignancour, Chief Executive Officer of Safe Orthopaedics, says: “The fact that we exceeded our guidance of 2,200 surgical procedures, […]
RTI Surgical® Announces Agreement with Oxford Performance Materials
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical® Inc. (RTI) (Nasdaq: RTIX), a leading global surgical implant company, announced a new agreement with Oxford Performance Materials, Inc. (OPM), a pioneer in advanced materials and additive manufacturing (3-D printing) for biomedical applications. The agreement, effective Feb. 4, 2016, grants RTI an exclusive license to OPM’s OsteoFab® technology platform for spinal […]
Wenzel Spine, Inc. Announces FDA Clearance to Launch VariLift
AUSTIN, Texas–(BUSINESS WIRE)–Wenzel Spine, Inc., a pioneer medical device company focused on providing minimally invasive, stand-alone alternatives to traditional spinal fusion, announced today that it received clearance from the U.S. Food and Drug Administration (FDA) to market VariLift®-LX as an interbody fusion device for stand-alone use in the lumbar spine. VariLift®-LX represents the next generation […]
SI-BONE, Inc. Announces Two Economic Publications Showing iFuse Implant System is Cost Effective and Saves Money
SAN JOSE, Calif., Feb. 11, 2016 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the use of the iFuse Implant System, a minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced the publication of two economic studies showing the cost benefits of MIS SI joint […]
Implanet announces the appointment of Brian T. Ennis to head its US subsidiary
Bordeaux, Boston, 8 February 2016 – IMPLANET (Euronext: IMPL, FR0010458729, PEA-PME eligible), a medical technology company specializing in vertebral and knee-surgery implants, announces the appointment of Brian T. Ennis as President of Implanet America. As such, he will head Implanet America and his objective will be to optimize its organisational structure in order to accelerate the […]
SPINEMarketNews.com is on Google Plus! Join us!
Our group plus.google.com/104646636881784161804 open to any interested person. Also feel free to share it with your colleagues. It is also a great forum for sharing the latest news and discussions.If you like us, please give us a positive feedback.We will appreciate very much your Follows.
LDR Announces Publication of Five-Year Results of Cost Utility Analysis of Mobi-C Cervical Disc
AUSTIN, Texas, Feb. 09, 2016 (GLOBE NEWSWIRE) — LDR Holding Corporation (Nasdaq:LDRH), a global medical device company focused on designing and commercializing novel and proprietary surgical technologies for the treatment of patients suffering from spine disorders, today announced that NEUROSURGERY® has published a peer-reviewed article that evaluated the cost-effectiveness of two-level cervical Total Disc Replacement […]
Amedica Regains Compliance With Nasdaq Listing Requirements and Announces Capitalization Improvements
SALT LAKE CITY, Feb. 09, 2016 (GLOBE NEWSWIRE) — Amedica Corporation (Nasdaq:AMDA), a company that develops and commercializes silicon nitride ceramics as a biomaterial platform, is pleased to announce that it has regained compliance with Nasdaq’s minimum bid price requirement. On February 8, 2016, the Company received a letter from the Nasdaq Listing Qualifications department […]
VGI Medical Announces 1st Implantation of the VerteLP® Lateral Lumbar Cage with Bi-Cortical Fixation
ST. PETERSBURG, FL (PRWEB) FEBRUARY 08, 2016–VGI Medical, a privately held medical device company offering innovative spinal implants, announces that it has successfully implanted the 1st patent pending VerteLP Lateral Lumber Cage. VerteLP represents the latest of lateral lumbar fusion, featuring enhanced bi-cortical plating technology to facilitate simplified cage insertion, and integrated fixation all through […]
Congratulations to The SPINE GROUP on reaching 11,000 LinkedIn members
Congratulations, The SPINE Group, on topping 11000 members in this awesomegroup. Since 2008, The SPINE Group has been supporting the Spine Industry providing relationships, discussions, events and information .Thank you for your partnership and we encourage others to keep joining! About The SPINE Group This group is for all the people involved in the SPINAL business willing to receive fresh […]