This year at the North American Spine Society’s 33rd Annual Meeting (NASS), NuVasive unveiled new spine innovation and clinical insights at the Los Angeles Convention Center. For those of you that missed NASS, we’ll highlight the six key takeaways from NuVasive. The Unveiling of Pulse™. The first surgical automation platform designed for spine, Pulse was […]
2019
Robotics, navigation systems create buzz at spine meeting
(medtechdive.com)—As in orthopaedic reconstruction, companies looking to move the needle in the spine device market are investing in robotics to compete. Modest growth in the sector is a driving factor. Medtronic told investors in a session at the meeting that the U.S. spine market has returned to growth, with an increase in procedures offsetting pricing […]
Spine Surgery Robots Market to hit US$ 320 million by 2026.Which are Today the most advanced Robots for Spine ?
In spine surgery robots market an innovative tool is essentially used to assists spinal surgeons. With these tools’ surgeons are able to perform various surgeries with ease. And high accuracy and precision. These sorts of surgery have several benefits over the conventional type. Along with this it can also lead to quicker recovery period, this […]
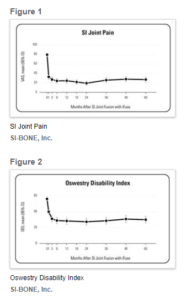
SI-BONE, Inc. Announces Publication of Five-Year Prospective Study Results on 103 Patients Treated with Triangular Titanium iFuse Implant System
SANTA CLARA, Calif., Sept. 26, 2019 (GLOBE NEWSWIRE) — SI-BONE, Inc., (Nasdaq: SIBN), a Silicon Valley-based medical device company dedicated to solving musculoskeletal disorders of the sacro-pelvic anatomy, today announced the publication of 5-year results from a long-term prospective study called LOIS (Long Term Outcomes from INSITE and SIFI). The publication, titled Long-term prospective clinical and radiographic outcomes after […]
Safe Orthopaedics announces its results for the first half of 2019
Éragny-sur-Oise, France, September 27, 2019 – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company specializing in the design and marketing of single-use implants and instruments for the minimally-invasive treatment of spine fracture pathologies, today announced its results for the first half of 2019 to June 30. Safe Orthopaedics’ half-year financial report will be available in the […]
DeGen Medical Receives Clearance of F1 MPS™ System for JOUST™ Minimally Invasive Surgical Procedure
Florence, SC, Sept. 26, 2019 (GLOBE NEWSWIRE) — DeGen Medical has received clearance from the FDA for its F1 MPS™ Modular Pedicle Screw System for JOUST™ Minimally Invasive Surgery (MIS) procedure. The JOUST™ MIS is a modular pedicle screw with cobalt-chrome alloy head for percutaneous procedures. The JOUST™ MIS technology includes MIS Towers for percutaneous […]
MEDICREA® Hosts Third Annual User Group Meeting (MAIA) in Lyon, France Followed by Attendance at SRS in Montreal, Canada
LYON, France & NEW YORK–(BUSINESS WIRE)–The MEDICREA® Group (Euronext Growth Paris: FR0004178572 – ALMED ; OTCQX Best Market – MRNTF), pioneering the digital transformation of spinal surgery through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) proprietary software platform, services and technologies, announced today that it hosted the […]
DeGen Medical Receives Clearance of Cyclops™ Anterior Cervical Plate System
Florence, SC, Sept. 26, 2019 (GLOBE NEWSWIRE) — DeGen Medical has received clearance from the FDA for its cervical spine implant, Cyclops™ Anterior Cervical Plate System. The Cyclops™ system is an anterior intervertebral fixation device that is designed to aid in stabilizing the cervical spine. The system provides an exceptionally short, low profile plate with […]
Life Spine Announces 510(k) Clearance of the Hinged Laminoplasty System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today the FDA 510(k) clearance of the Hinged Laminoplasty System. “The laminoplasty procedure is an excellent alternative to a laminectomy. I am excited to continue to launch systems that are designed for […]
Medtronic Announces New Clinical Trial to Study Infuse Bone Graft in TLIF Spine Procedures
DUBLIN and CHICAGO, Sept. 26, 2019 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT) today announced U.S. FDA approval of a prospective, randomized pivotal clinical trial for the use of Infuse™ Bone Graft in Transforaminal Lumbar Interbody Fusion (TLIF) spine procedures. A TLIF is a type of surgery that fuses – or joins – bones of the […]
EOS imaging to Showcase Breakthrough Spine Solutions at North American Spine Society (NASS) 34th Annual Meeting
EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI – Eligible PEA – PME), a leader in 2D/3D orthopedic medical imaging and software solutions for 3D anatomical modeling and surgical planning, will present its differentiated Advanced Orthopedic Solutions for spine care, including spineEOS, EOSlink™ and Auto 3D modeling prototype, with a focus on personalized patient care during […]
4WEB Medical Announces Completion of Cell Differentiation and Gene Expression Study
CHICAGO, Sept. 25, 2019 /PRNewswire/ — 4WEB Medical, an orthopedic device company focused on developing innovative implants utilizing its proprietary Truss Implant Technology™ announced today the completion of Phase I in vitro testing that demonstrates the surface roughness on its truss implants stimulates a superior osteogenic response when compared to smooth titanium and PEEK. The study, conducted by David […]
CTL Amedica to unveil MONET™ Integrated ACIF Cage System during NASS Sept. 25 – 28
DALLAS (PRWEB) SEPTEMBER 25, 2019–CTL Amedica Corporation will officially launch its MONET™ ACIF Integrated Cage System with Supplementary Fixation during the North American Spine Society (NASS) annual meeting and exhibition Sept. 25-28, 2019. NASS will be held at McCormick Place, West Building, located at 2301 S. King Drive in Chicago, IL. CTL Amedica representatives will […]
DePuy Synthes Expands Comprehensive Offering to Treat Degenerative Spine Disease with Launch of 3D Printed Implant Portfolio for Spine Surgery
CHICAGO, Sept. 25, 2019 /PRNewswire/ — The Johnson & Johnson Medical Devices Companies* today announced that DePuy Synthes has launched the CONDUIT Interbody Platform with EIT Cellular Titanium Technology, further expanding its comprehensive offering to treat degenerative spine disease. The innovative portfolio, which includes 3D printed titanium interbody implants for spinal fusion surgery is designed […]
Stryker announces SAKOS pivotal trial results for SpineJack System published in The Spine Journal
KALAMAZOO, Michigan, USA, Sept. 25, 2019 /PRNewswire/ — Stryker announced today the publication of the SAKOS pivotal trial results for the SpineJack implantable fracture reduction system (SpineJack System) in The Spine Journal.1 The SAKOS trial was a prospective, multicenter, randomized study designed to support a non-inferiority finding for use of the SpineJack system versus balloon […]
Alphatec Announces Commercial Launch of IdentiTi™ TLIF Posterior Oblique Porous Titanium Interbody Implant System
CARLSBAD, Calif., Sept. 25, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of its IdentiTi TLIF Posterior Oblique Titanium Interbody Implant System (“IdentiTi PO”) for transforaminal lumbar interbody fusion procedures. The launch of […]
Biedermann Motech To Further Expand Its iMAS360™ Procedural Solution And To Launch MOSS VRS® – The Next Generation Pedicle Screw Technology™ To The US Market
MIAMI & DONAUESCHINGEN, Germany–(BUSINESS WIRE)–Biedermann Motech, the leader in pedicle screw technology and pioneer in the spinal market for more than 30 years, today announced the further expansion of its product portfolio of advanced solutions for spinal surgery with the integration of the DELTA XS™ Shank Based Retractor System into its iMAS360™ Procedure as well […]
RTI Surgical® Announces Commercial Launch of the CervAlign® Anterior Cervical Plate System
DEERFIELD, Ill., Sept. 24, 2019 (GLOBE NEWSWIRE) — RTI Surgical Holdings, Inc. (Nasdaq: RTIX), a global surgical implant company, today announced the full commercial launch of the CervAlign® Anterior Cervical Plate (ACP) System to add to the growth of its Established Therapies spine portfolio. The CervAlign ACP System is designed to promote cervical fusion by providing […]
SeaSpine® Announces Exclusive Development and Licensing Agreement with restor3D®
CARLSBAD, Calif., Sept. 24, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced that it has entered into an exclusive (field-based) development and licensing agreement with restor3d, Inc., a privately-held medical device company co-founded by Ken Gall, Professor […]
Innovasis:Company continues to innovate spine portfolio with partnership to advance the next interbody material; Porous HA PEEK
SALT LAKE CITY, September 25, 2019 –Innovasis, Inc. an integrated research, development and manufacturing spinal device company, signs exclusive deal with Pore Matrix to utilize the next interbody technology, Porous HA PEEK. Anthony Ruggiero, CEO of Pore Matrix, stated, “It’s a pleasure to be partnering with Innovasis and its innovation-driven culture to bring Porous HA […]
Life Spine Announces First Cases With the Micro Invasive PROLIFT® Lateral Expandable Spacer System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced the initial surgeries of the PROLIFT Lateral Expandable Spacer System. “I have been utilizing PROLIFT for posterior lumbar approaches in my practice for the past few years and have been eager to […]
Empirical Spine to Present Data from Study of the LimiFlex™ Paraspinous Tension Band at the Society for Minimally Invasive Spine Surgery (SMISS) Annual Forum 2019
SAN CARLOS, Calif., Sept. 24, 2019 /PRNewswire/ — Empirical Spine, Inc., a Silicon-Valley based medical technology company, announced today that data from the IDE study of the company’s LimiFlex Paraspinous Tension Band for patients suffering from degenerative spondylolisthesis with lumbar spinal stenosis, will be presented at the Society for Minimally Invasive Spine Surgery (SMISS) Annual Forum 2019. The […]
Centinel Spine® Celebrates 30 Years of Clinical Excellence with prodisc® Total Disc Replacement Technology
NEW YORK, Sept. 24, 2019 /PRNewswire/ — Centinel Spine, LLC, the largest privately-held spine company focused on anterior column reconstruction, today celebrated thirty years of clinical use of the prodisc total disc replacement technology. The prodisc family of devices includes the most frequently implanted lumbar total disc replacement in the United States, prodisc L, and the most frequently implanted cervical total […]
Global BMD will join Eurospine 2019 in Helsinki!
Welcome to join Global BMD in Eurospine 2019 (Booth 1B Hall 4) from October 16-18! At Eurospine annual meeting, Global BMD will display their 3D printed spinal and orthopedics implants. For more information, please contact us soon! About GLOBAL biomedica ISO 13485 certified, Czech Republic-based company specialize in producing interbody spinal fusion cages designed and […]
CarboFix Extends Its CarboClear® Carbon Fiber Pedicle Screw System Product Line With Its FDA Cleared Fenestrated Screws
DOVER, Delaware, Sept. 24, 2019 /PRNewswire/ — CarboFix has announced today that the U.S. Food and Drug Administration (FDA) has cleared its CarboClear® Carbon Fiber Fenestrated Pedicle Screw System used in the treatment of patients with advanced stage spinal tumors. CarboClear® Fenestrated Pedicle Screws are used in conjunction with a high viscosity, radiopaque, PMMA bone cement (Teknimed’s High V+ […]
Benvenue Medical Releases MIS TLIF Clinical Data on Luna Device Showing Improved Patient Outcomes, Decreased Back and Leg Pain at 12 Months Post-Lumbar Fusion Surgery
CHICAGO & SANTA CLARA, Calif.–(BUSINESS WIRE)–Benvenue Medical, Inc., an innovative developer of minimally invasive surgery (MIS) expandable implant solutions for lumbar fusions, today announced data demonstrating clinical results of improved relief of back pain and leg pain, as well as lowered Oswestry Disability Index (ODI) scores at 12 months post-op with its Luna™ product. Richard […]
NuVasive LessRay® Platform Wins 2019 Spine Technology Award from Orthopedics This Week
SAN DIEGO, Sept. 23, 2019 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced its LessRay® radiation reduction and workflow enhancement platform won the 2019 Spine Technology Award presented by Orthopedics This Week, a widely read publication in the […]