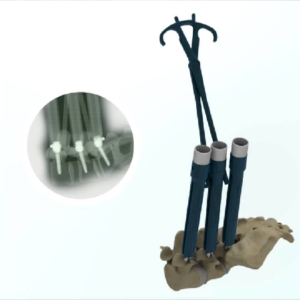
AUDUBON, Pa., Dec. 06, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, recently announced the entrance of ExcelsiusGPS®, a revolutionary robotic guidance and navigation system, to the international market with system installations completed in several European countries. This past October, the first European Excelsius GPS® procedure was performed at the […]
NEWS
Medtronic to pay $31M in DoJ settlement of medical device probes
Two companies now owned by Medtronic agree to pay $31M to resolve a U.S. Department of Justice probe into the marketing of a medical device meant to treat a vascular defect in the brain for unapproved purposes and paying kickbacks to hospitals to get them to use a second product. The U.S. Food and Drug Administration in […]
joimax® Rolls Out Its Endoscopic Generation 4 Devices and Showcases New 3D-Printed Titanium Implants at the German Spine Society (DWG) Convention
KARLSRUHE, Germany & IRVINE, Calif.–(BUSINESS WIRE)–joimax®, the Germany-based market leader of technologies and training methods for full-endoscopic minimally-invasive spinal surgery, is well positioned for Germany’s principal spine convention, German Spine Society (DWG), taking place December 6 to 8 in Wiesbaden, Germany. This year, joimax® is focusing on their Endoscopic Tower Generation 4 Devices, an expert solution for […]
SpineGuard Announces a Partnership with Carnot Interfaces for the Combination of its DSG® Technology with Surgical Robots
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that designs, develops, and markets disposable medical devices intended to make spine surgery safer by bringing real-time digital technology into the operating room, announced today a partnership agreement with Carnot Interfaces for the combination of its DSG technology with surgical robots. In November […]
Nexxt Spine Launches SAXXONY® Posterior Cervical Thoracic Spine System
NOBLESVILLE, Ind.–(BUSINESS WIRE)–Nexxt Spine LLC, a medical device company focused on designing, manufacturing, and distributing innovative spinal solutions, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) and full commercial launch of its SAXXONY® Posterior Cervical Thoracic System—a system designed to stabilize cervical (C1 to C7) and thoracic (T1 to T3) spinal […]
Spineology Inc. Announces New Board Members
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., the innovator in anatomy-conserving spine surgery, announced today the appointment of two new members to its Board of Directors, Bob Paulson and Phil Soran. Mr. Soran is a successful serial entrepreneur who has founded several technology companies and was inducted into the Minnesota Business Hall of Fame in 2016. He […]
Safe Orthopaedics organizes a workshop on spinal fracture treatment at DWG 2018
Eragny-sur-Oise, France, December 4, 2018 – 5:45pm CET – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company specialized in the design and marketing of single-use implants and instruments improving the minimally invasive treatment of spinal fracture pathologies, will be present at the DWG 2018 Annual Conference taking place in Wiesbaden (Germany) from December 6th to 8th, 2018. […]
IMPLANET to attend the 13th Annual Meeting of the German Society for Spine Surgery (DWG)
Bordeaux, Boston, December 4, 2018 at 8:00am: IMPLANET (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, is announcing that it will attend the 13th Annual Meeting of the German Society for Spine Surgery (DWG), which will take place December 6-8 in Wiesbaden, Germany. Implanet’s presence at […]
How many MIS systems are now in the market? Only 90 Minimally Invasive Pedicle Screw Systems!
According to iData Research, the U.S. market, valued at $7.7 billion in 2017. Of this total market share, 78% can be attributed to the traditional spine market, and the remaining, to the minimally invasive (MIS) segment. Growth in the former has slowed as newer, minimally invasive innovations gradually cannibalize the traditional spine market. Growth, at […]
Carmel spine surgeon wins $112M verdict in royalty battle with Medtronic
(www.ibj.com)–Dr. Rick C. Sasso, an Indiana spine surgeon and inventor, has won a sweeping, five-year legal battle against medical-device giant Medtronic, with a jury this week awarding him $112 million in damages.Sasso, 58, president of Carmel-based Indiana Spine Group, had claimed that Medtronic had violated a contract by not paying royalties he was due for […]
WishBone Medical, Inc. acquires Response Ortho
WARSAW, Ind., Nov. 28, 2018 (GLOBE NEWSWIRE) — WishBone Medical, Inc. announces the acquisition of Response Ortho. Response Ortho has a complete pediatric line of external and internal fixation and deformity correction products. Response Ortho has been in business for fourteen years and has built a modern, high-tech manufacturing facility with access to an experienced […]
Swiss implant makers come under the microscope
(swissinfo.ch)–The Implant Files revelations of faulty implants and weak safety standards are raising alarm bells for Switzerland’s rapidly growing medical device industry. Manufacturers and industry bodies say that safety standards have improved in the last decade, but do they go far enough? The market for implanted medical devices is booming globally. Tens of thousands of […]
Massive investigation of medical device industry highlights Memphis-based Medtronic Spine, kickback allegations
(commercialappeal.com)–An international journalism investigation of the medical device industry describes dangerous devices, kickbacks to doctors and weak regulations – and shines a spotlight on a medical company with a big presence in Memphis. That company is Medtronic, which has its main operations in Minneapolis and annual sales of about $30 billion. One of its units, […]
UK firm sold spinal implants that disintegrated
(theguardian.com)–A UK company’s spinal implants that allegedly moved and eroded in patients, and which are at the centre of legal action, have highlighted potential weaknesses in the way in which some medical devices enter the market, an investigation has revealed.Documents seen by the Guardian show the plastic discs were approved for sale by the British […]
Independent evaluation confirms savings generated for hospitals by using Safe Orthopaedics’s technology
Eragny-sur-Oise (France), November 27, 2018 5:45pm CET – SAFE ORTHOPAEDICS (FR0012452746 –SAFOR), a company specialized in the design and distribution of implants and single-use instruments improving minimally-invasive treatments of pathologies from spinal fractures, announces today the publication of an independent economic evaluation by the CHU of Saint-Etienne (France) in 2018 on the use of instruments […]
Medtronic slips 2% premarket on device injuries
(SEEKINGALPHA.COM))–Medtronic (NYSE:MDT) is down 2% premarket, albeit on only 2,000 shares, on the heels of a Bloomberg report about a study conducted by a group of journalists that showed a surprisingly high number of injuries and deaths from medical devices over the past 10 years. The company’s devices appeared prominently in the study, accounting for 20% of reported adverse […]
Stryker announces pricing of €2.25 billion senior notes offering
Kalamazoo, Michigan, Nov. 27, 2018 (GLOBE NEWSWIRE) — Stryker (NYSE:SYK) announced today that it has priced the following notes: (i) €550 million aggregate principal amount of the Company’s 1.125% Notes due 2023 (the “2023 Notes”), (ii) €750 million aggregate principal amount of the Company’s 2.125% Notes due 2027 (the “2027 Notes”), (iii) €650 million aggregate […]
Positive Payor Policy Covering coflex® by AmeriHealth Caritas for the Surgical Treatment of Lumbar Spinal Stenosis
NEW YORK, Nov. 27, 2018 /PRNewswire/ — Paradigm Spine, LLC, a leader in providing solutions for the treatment of lumbar spinal stenosis, today announced the issuance of an AmeriHealth Caritas Clinical Policy, dated September 1, 2018, for the exclusive coverage of the coflex® interlaminar stabilization device for the treatment of lumbar spinal stenosis. Lumbar spinal stenosis (“LSS”), affecting 1.6 million […]
Viseon, Inc. Announces First US Clinical Use of the Voyant System for Minimally Invasive Spine Surgery Access, Illumination and Visualization
IRVINE, Calif.–(BUSINESS WIRE)–Viseon, Inc. today announced the first U.S. clinical human use of the Voyant System for Minimally Invasive Spine Surgery access, illumination and visualization. The case was performed by Neurosurgeon John J. Knightly, MD of the Atlantic NeuroSurgical Specialists in Morristown, New Jersey. The Voyant System is composed of a sterile single-use, disposable retractor […]
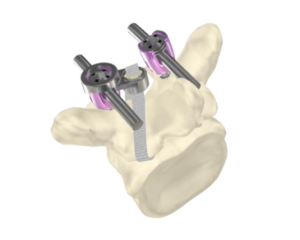
Astura Medical Receives FDA 510(k) Clearance For OLYMPIC MIS Posterior Spinal Fixation System
CARLSBAD, CA – November 26, 2018 – Astura Medical, a high-growth, innovative spine technology company, today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Olympic Minimally Invasive Surgery (MIS) Posterior Spinal Fixation System. The Olympic MIS system delivers a new level of intraoperative flexibility and efficiency by allowing surgeons […]
Medtronic to Acquire Nutrino Health
DUBLIN and TEL AVIV, Israel – November 21, 2018 – Medtronic plc (NYSE:MDT), a global leader in medical technology, and Nutrino Health Ltd., a leading provider of nutrition-related data services, analytics, and technologies, today announced the companies have entered into a definitive agreement under which Medtronic will acquire Nutrino. Given that food and nutrition are […]
Interbody Fusion Cage Market to Reach $2.30 Bn, Globally, by 2023 at 3.4% CAGR, Says Allied Market Research
PORTLAND, Oregon, November 21, 2018 /PRNewswire/ –Rise in geriatric population, increase in incidence of spinal and sports-related injuries, and benefits associated with interbody fusion cage surgeries would drive the growth of the global interbody fusion cage market Allied Market Research published a report, titled, Interbody Fusion Cage Market by Product (Lumbar, Cervical, Thoraco-lumbar, and Thoracic), […]
Vertiflex® Announces Publication of Data Analysis Showing 85% Reduction in Patients Using Opioids After Treatment With Superion® Indirect Decompression System
CARLSBAD, Calif.–(BUSINESS WIRE)–Vertiflex® Inc., a leading innovator of advanced, minimally invasive interventions for spinal stenosis, announced additional results from a randomized, controlled trial of its Superion® Indirect Decompression System in patients with lumbar spinal stenosis (LSS). The results, published in the Journal of Pain Research (https://www.dovepress.com/articles.php?article_id=42435), showed an 85 percent decrease in the proportion of patients who were […]
CE mark clearance for the Jazz Cap System®, a screw securing solution for vertebral fusion
Bordeaux, Boston, November 20, 2018 at 8:00am CET – IMPLANET (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral and knee surgery implants, is announcing the award of CE marking for the Jazz Cap System®, developed to meet the constraints of vertebral fusion indications in adults. Jazz Cap System®, […]
Altus Capital Partners, Inc. Acquires ChoiceSpine, LP
WILTON, Conn.–(BUSINESS WIRE)–Altus Capital Partners, Inc. (“Altus”), an investment firm focused on the North American manufacturing sector, today announced it has acquired ChoiceSpine, LP, a leading designer, manufacturer, and marketer of specialized spinal implants, instrumentation and biologics for the surgical treatment of complex spine disorders. Financial terms of the transaction were not disclosed. Based in […]
MEDICREA® Achieves 3,000 Surgery Milestone with UNiD ASI™ and Patient Specific Implants
LYON, France & NEW YORK–(BUSINESS WIRE)–The MEDICREA® Group (Euronext Growth Paris: FR0004178572-ALMED ; OTCQX Best Market – MRNTY & MRNTF), pioneering the digital transformation of spinal surgery through Artificial Intelligence, predictive modeling and patient specific implants with its UNiD ASI™ (Adaptive Spine Intelligence) proprietary software platform, services and technologies, announced today that the 3,000th surgery […]
Mazor Robotics Shareholders Approve Merger Agreement With Medtronic
CAESAREA, Israel, Nov. 19, 2018 /PRNewswire/ — Mazor Robotics Ltd. (TASE: MZOR; NASDAQGM: MZOR), a pioneer and a leader in the field of robotic guidance systems, today announced that at a Special General Meeting of Shareholders held earlier today, Mazor Robotics shareholders approved the previously announced definitive merger agreement with wholly-owned subsidiaries of Medtronic plc (NYSE: MDT). Approximately […]