IRVING, TX – September 22, 2021 – Astura Medical, a high-growth, innovative spine technology company, announced that it will be attending the 2021 North American Spine Society (NASS) Annual Meeting being held in Boston, MA from September 29th through October 1st. The Company’s current technology that is already widely recognized as best-in-class will be on […]
NEWS
Life Spine to Showcase One of the Industry’s Largest Expandable Offerings and Full Spine Product Portfolio at NASS 2021
Huntley, IL, September 21, 2021 –Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that it would be showcasing its complete line up of Micro Invasive procedural solutions, which includes 13 Expandable Interbody devices making it one of the largest and most […]
Horizon Technology Finance Leads $15 Million Venture Loan Facility to Spineology
FARMINGTON, Conn., Sept. 20, 2021 /PRNewswire/ — Horizon Technology Finance Corporation (NASDAQ: HRZN) (“Horizon”), a leading specialty finance company that provides capital in the form of secured loans to venture capital backed companies in the technology, life science, healthcare information and services, and sustainability industries, announced today it led a $15 million venture loan facility to Spineology, Inc. (“Spineology”), of which […]
AxioMed LLC, A KICVentures Group Portfolio Company, Achieved Landmark Patent For Lateral Lumbar Viscoelastic Disc Replacement Medical Device
MALDEN, MASS. (PRWEB) SEPTEMBER 18, 2021–AxioMed continues to press its advantages in the artificial total disc replacement market with its investment in lateral lumbar viscoelastic disc technology. At a time when revenues and margins are driving the spine industry, AxioMed chose to venture alone into developing a lateral lumbar viscoelastic disc replacement. We thank the United […]
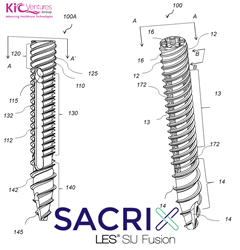
Sacrix LLC To Be Issued A 3D Porous Patent On Its Threaded Implant And Lateral-Oblique Sacroiliac Joint Fusion Technique On September 28th, 2021
MALDEN, MASS. (PRWEB) SEPTEMBER 18, 2021–Vito Lore, the VP of innovations at KICVentures Group, said “the patent includes a device claim of our threaded implant with the fusion channel; also an embodiment with a 3D porous structure”. “Clinically the 3D porous structure is expected to enhance the threaded implant’s performance in fixating the sacroiliac joint and […]
CoreLink Surpasses 5,000 Entasis SI Joint Fusion Implantations With Patented Stackable Guide Wires
ST. LOUIS, Sept. 16, 2021 /PRNewswire/ — CoreLink, LLC, a leading designer and manufacturer of spinal implant systems, today announced the implantation of over 5,000 Entasis® Lateral SI Joint Fusion System compression screws to fuse the sacroiliac joint. CoreLink also announced the company has been awarded patent number 11,052,229 by the United States Patent and Trademark Office (USPTO). The patent, titled, […]
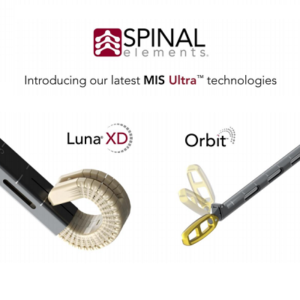
Spinal Elements® Announces Full Commercial Launch of Luna® XD and Orbit™ Systems
Carlsbad, CA, September 15, 2021 – Spinal Elements, a spine technology company, today announced the full commercial launch of the Luna XD multi-expandable lumbar interbody fusion device and Orbit articulating discectomy systems. Luna XD and Orbit have been integrated into Spinal Elements as the newest technologies in its MIS Ultra™ platform of products and procedural solutions. The […]
ATEC to Feature Comprehensive Procedural Solutions at NASS 2021
CARLSBAD, Calif.–(BUSINESS WIRE)– Alphatec Holdings, Inc. (Nasdaq: ATEC), a provider of innovative solutions dedicated to revolutionizing the approach to spine surgery, announced today that the Company’s comprehensive procedural portfolio will be featured at the North American Spine Society (NASS) Annual Meeting, which will be held at the Boston Convention and Exhibition Center from September 29 […]
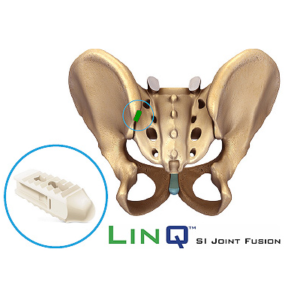
PainTEQ Acquires Three U.S. Patents for Innovative Sacroiliac (SI) Joint Procedure
TAMPA, Fla., Sept. 15, 2021 /PRNewswire/ –PainTEQ, a fast-growing,Tampa-based medical device company focused on treating sacroiliac (SI) joint dysfunction, announced the award of three new U.S. patents unique to the LinQ procedure. The LinQ™ process is a minimally invasive, novel procedure designed to help patients suffering from SI joint dysfunction. These patents solidify PainTEQ as an industry leader in minimally invasive […]
Aurora Spine Corporation Announces C$6.5 Million Private Placement with Institutional Investors
CARLSBAD, Calif., Sept. 15, 2021 (GLOBE NEWSWIRE) — Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSXV: ASG) (OTCQB: ASAPF), a designer and manufacturer of innovative medical devices that improve spinal surgery outcomes, today announced that it has entered into securities purchase agreements for a private placement of the Company’s common shares (or common share […]
Centinel Spine® Announces First International Use of 3D-Printed Porous Titanium FLX™ Platform
WEST CHESTER, Pa., Sept. 14, 2021 /PRNewswire/ — Centinel Spine®, LLC, a leading global medical device company addressing cervical and lumbar spinal disease through anterior surgical access, today announced the first international implantation of the 3D-Printed Porous Titanium FLX™ platform. Dr. Alfredo Guiroy and Dr. Jose Ricardo Prina in Argentina were the first surgeons outside the United States to implant the STALIF C FLX™ and STALIF M FLX™ […]
Zimmer Biomet Announces Key Additions to “NewCo” Leadership Team; Transaction to Form New Independent, Publicly Traded Company On Track
WARSAW, Ind., Sept. 14, 2021 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global medical technology leader, today announced key appointments to the “NewCo” leadership team, including Richard J. Heppenstall as Executive Vice President and Chief Financial Officer. Additionally, the Company announced ZimVie as the name for the planned spin-off company for the Spine and Dental businesses. […]
Accelus Announces Addition of TiHawk9™ Titanium-Bonded Multidirectional Implant to its FlareHawk® Family of Expandable Interbody Fusion Cages
PALM BEACH GARDENS, Fla., Sept. 14, 2021 (GLOBE NEWSWIRE) — Accelus, a privately held medical technology company focused on accelerating the adoption of minimally invasive surgery (MIS) as the standard of care in spine, today announced the introduction and commercial availability of the TiHawk9 expandable interbody cage, the latest addition to its FlareHawk® family of spinal […]
Sacrix Announces Publication on Successful Fusion Using Their Sacrofuse Device
BOSTON (PRWEB) SEPTEMBER 13, 2021 Sacrix LLC (Boston, MA) is pleased to announce the positive results of the study was performed at the LES Clinic “Clinical Outcomes of Novel Lateral-Oblique Percutaneous Sacroiliac Joint (SIJ) Fixation”, a technique perfected by Professor Dr. Kingsley R Chin, MD, a board-certified orthopedic spine surgeon. The peer-reviewed publication highlights effectiveness and […]
SPINEWAY – 2021 half-year results
Ecully, 13 September 2021 –Spineway’s Board of Directors, at a meeting held on 13 September 2021 chaired by Stephane Le Roux, closed the half-year results as of 30, June 2021. Spineway confirmed a sharp 153% 2021 half-year turnover increase compared with 2020 same period despite an international economic context still disrupted by the worldwide pandemic. The sales recovery in its main territories allowed […]
+60 Lateral Lumbar Cages to Know!
Today, we present the update of our Lateral Lumbar Cages section. Following our updating schedule for all of our product listings, we have taken the following actions: We have included all the lateral cages that are somehow differentiated because they are expandable, stand-alone, because of the material or due to the retractor device. The standard PEEK […]
KICVentures Group Successfully Meets ISO 13485 Recertification for its medical device subsidiary
BOSTON (PRWEB) SEPTEMBER 10, 2021–KICVentures Group Inc., a Boston-based investment firm with a portfolio of leading healthcare technology companies focused on spine surgery, announced that portfolio company LESspine Innovations has successfully completed its ISO 13485 recertification audit. What is ISO 13485? It is an internationally recognized standard for a Quality Management System (“QMS”) for the design […]
Spinal Elements® Announces 100th Case with Sapphire X™Anterior Cervical Fixation System
Carlsbad, CA, September 10, 2021 — Spinal Elements today announced it has reached the milestone of completing 100 Sapphire X procedures after only a few weeks of commercialization. This minimally invasive surgical anterior cervical plate system features streamlined integrated instrumentation designed to increase procedural efficiency while reducing procedural steps and complexity. As one of the […]
OrthoPediatrics Announces Entry into a Distribution Agreement with SeaSpine to Exclusively Distribute the 7D Surgical FLASH Navigation Platform for Pediatric Applications
WARSAW, Ind., Sept. 09, 2021 (GLOBE NEWSWIRE) — OrthoPediatrics Corp. (“OrthoPediatrics” or the “Company”) (Nasdaq: KIDS), a company focused exclusively on advancing the field of pediatric orthopedics, today announced they have entered into a distribution agreement with SeaSpine® Orthopedics Corporation, a wholly-owned subsidiary of SeaSpine Holdings Corporation (Nasdaq: SPNE), to exclusively distribute the 7D Surgical FLASH™ Navigation […]
NuVasive Simplify Cervical Disc Data Published In International Journal Of Spine Surgery
SAN DIEGO, Sept. 9, 2021 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced the NuVasive Simplify® Cervical Disc one-level U.S. Food and Drug Administration (FDA) Investigational Device Exemption (IDE) study data was recently published in the International Journal of Spine Surgery. The data […]
We have Updated our List of Product Videos! Now, we have more than 900!
Since last year, we have been constantly uploading products videos in our Youtube Channel. Now we have more than 900 videos and we have gained more than 500 subscribers. Thank you very much to all of them! Also, a few months ago we launched the Brochures section (where we have included more than 700 Surgical Techniques) and the VIDEOS section. Both […]
Spinal Elements® Announces Executive Appointments to Commercial Team
Carlsbad, CA, September 8, 2021 – Spinal Elements, a spine technology company, today announced recent additions to its commercial team. André Potgieter will be leading Spinal Elements’ sales initiatives and scale up as the company’s Chief Commercial Officer. Mr. Potgieter has over 25 years of experience in spine technology sales. He was responsible for building […]
80 Anterior Lumbar Cages (ALIF) to Know!
Today, we are finally publishing the updated ALIF list of devices. We have included videos, brochures, surgical techniques. The list contains most of the systems that are differentiated either by material or design. We have left out of the list most of the Peek cages that are very similar. We hope you find this compilation […]
Robotic detection of bone breach via SpineGuard’s DSG® technology: 100% efficacy proven in lab experiment
PARIS and BOULDER (CO), September 7th, 2021 – 08:30 am CEST – SpineGuard (FR0011464452 – ALSGD), an innovative company that deploys its DSG (Dynamic Surgical Guidance) sensing technology to secure and streamline the placement of bone implants, announced today a key milestone reached in the development of its robotic application. DSG is based on the local measurement of […]
SeaSpine Announces Initial Launch of Mariner Adult Deformity System
CARLSBAD, Calif., Sept. 07, 2021 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch and completion of initial surgeries of the Mariner Adult Deformity Pedicle Screw System. SeaSpine continues to build upon the strength and […]
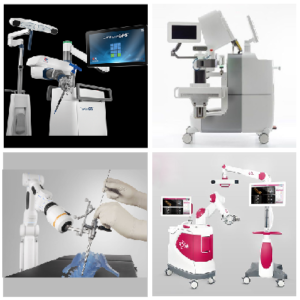
I want a robot for spinal surgery! What are the best options?
The growing awareness among the physicians and patients regarding the benefits associated with the usage of spine surgery robots has been directly impacting the growth of the market. As we have already commented in another article, Spine surgery robots market account to USD 330.85 million by 2027 growing at a CAGR of 16.0% 2020-2027. It […]
Updated Interspinous (IPD) Section! More than 60 Devices!
Today, we present the update of our Interspinous (IPD) section. Following our updating schedule for all of our product listings, we have taken the following actions: We have eliminated those implants whose companies no longer exist. In some cases, although the products are obsolete, we have preferred to leave them. We consider that they continue […]