AKRON, OH–(Marketwired – Jul 26, 2016) – Intellirod Spine, the spinal implant company developing disposable and implantable wireless RFID sensor technology for monitoring spinal rod strains, secured additional equity financing from new and existing investors including the Queen City Angel First Fund V and JumpStart. Funds will be used to reach key milestones toward the […]
NEWS
SpineCraft Announces the Successful US release of the ASTRA SPINE SYSTEM
July 26th, 2016, Westmont, Ill. –SpineCraft would like to announce the successful US release of the ASTRA SPINE SYSTEM. The ASTRA SPINE SYSTEM is designed for spinal deformity correction as well as tumor, trauma and degenerative procedures. The ASTRA SPINE SYSTEM also includes a new revision & extension module developed for the expanding revision patient population. […]
Medtronic Receives First-Ever FDA Clearance for Cement-Augmented Pedicle Screws
DUBLIN – July 20, 2016 – Medtronic plc (NYSE: MDT) today announced U.S. Food and Drug Administration (FDA) 510(k) clearance of the CD Horizon® Fenestrated Screw Set, which can be used for patients diagnosed with advanced stage tumors involving the thoracic and lumbar spine. This marks the first U.S. clearance for cement-augmented pedicle screws. The […]
Zimmer Biomet Becomes The Latest Ortho To Go Robotic
(Seeking Alpha)–In paying $77.5m for a 59% stake in the French company Medtech – and laying plans to acquire the rest of the business for another $54m or so – Zimmer Biomet (NYSE:ZBH) has confirmed the trend for orthopedics groups to pursue surgical robotics. With deals including Smith & Nephew (NYSE:SNN) buying Blue Belt Technologies, […]
Two Clinical Studies Demonstrate Reduced Surgical Complications and Revision Surgeries With Mazor Robotic Surgical Guidance System
WASHINGTON–(BUSINESS WIRE)–Mazor Robotics, Ltd. (TASE: MZOR; NASDAQ GM: MZOR), a developer of innovative guidance systems and complementary products, today announces comprehensive data from two studies demonstrating significant clinical benefits of its surgical guidance technological platform. The data was presented during the 23rd International Meeting on Advanced Spine Techniques (IMAST) annual meeting in Washington, D.C. The […]
Zimmer Biomet enters surgical robotics with Medtech buy
HONG KONG, July 18, 2016 /PRNewswire/ — Ally Bridge Group (“ABG”), a leading China-global cross-border healthcare-focused investment group, is pleased to announce the sale of its entire stake in Medtech SA (“Medtech” or the “Company”) to Zimmer Biomet Holdings, Inc. (“Zimmer Biomet”), a leading global orthopedics company based in Warsaw, Indiana, USA. Medtech is a […]
Life Spine Achieves Unparalleled Financial Results and a 50% Sales Growth Rate in Q2 2016
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that revenues for the first half of 2016 grew by 44% over the same period in 2015. Revenues for the second quarter of 2016 grew by 50% as compared to the […]
Medtronic (MDT) Announces Receipt of FDA Approval for Prestige LP Cervical Disc
Medtronic plc (NYSE: MDT) announced the U.S. Food and Drug Administration’s (FDA) approval of the Prestige LP(TM) Cervical Disc for the treatment of cervical disc disease causing nerve or spinal cord compression at two adjacent levels between the C3-C7 segments of the neck. The Prestige LP Disc is designed to allow motion in the neck […]
SI-BONE, Inc. Announces 40th Peer-Reviewed Publication – a Multicenter Long Term Outcomes Study Showing Durable Results With the iFuse Implant System
SAN JOSE, Calif., July 18, 2016 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the use of the iFuse Implant System® (“iFuse”), a triangular-shaped minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced the publication of the 40th peer-reviewed paper on SI joint fusion with […]
Medtronic Launches Revamped Minimally Invasive Spine Fusion Procedures
DUBLIN – July 15, 2016 – Medtronic plc (NYSE: MDT) today announced the continued development of the OLIF Platform with the launch of the PivoxTM Oblique Lateral Spinal System with Lateral Plate for OLIF25TM and Divergence-LTM Anterior/Oblique Lumbar Fusion System for OLIF51TM at the International Meeting on Advanced Spine Techniques (IMAST) in Washington, D.C. Lumbar […]
Medtronic invests another $20 million in Mazor Robotics
(Star Tribune)–Medtronic PLC is preparing to invest another $20 million in Israeli med-tech company Mazor Robotics, which is expected on Tuesday to announce a new robotically assisted spine-surgery system called Mazor X. Analysts say physician interest in robotically assisted surgical tools is steadily growing, and Medtronic seems to be positioning itself to capitalize on the […]
ulrich medical USA® Announces Market Release of neon3 Posterior Cervical System
ST. LOUIS, July 13, 2016 /PRNewswire/ — ulrich medical USA, Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States, announced today the market release of the neon3 TM Posterior Cervical System, which is intended to provide immobilization and stabilization of spinal segments as an adjunct to fusion […]
Titan Spine Raises Capital for Continued U.S. Growth and Adoption of its Interbody Fusion Devices Featuring nanoLOCK™ Technology
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative surface-enhanced spinal interbody fusion implants, today announced that it has closed a substantial round of financing with investor Southlake Equity Group. Titan will use the proceeds to further expand U.S. sales growth and surgeon adoption of its Endoskeleton® line of titanium […]
SpineGuard reports 29% growth and €1.9M revenue for the second quarter 2016
PARIS and SAN FRANCISCO, July 12, 2016 – SpineGuard (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices that empower surgeons to enhance clinical outcomes and simplify surgeries, reported today that its second quarter 2016 revenue grew by 29%, to €1.9 million. Pierre Jérôme, Co-founder and CEO of SpineGuard, said: “Our […]
Are you looking for a Different Cage? Learn about 19 PLIF Expandable Cages
PLIF Expandable interbody cages is a new product category that provides the following key benefits: Most of them, can be implanted with a minimally invasive surgical procedure allowing surgeons to go through a small corridor and change the geometry of the cage to correspond to what the patient’s spine requires. These implants may help protect a patient’s nerve roots […]
Medicrea H1 Revenue Rises 7% to 14.8 Million Euros
The Medicrea Group (Alternext Paris: FR0004178572 – ALMED), PEA- PME eligible, pioneering the development and manufacture of patient-specific analytical services and implant solutions for the treatment of spinal conditions, announces sales for the first half of 2016. Medicrea First Half Sales: H1 2015 Sales = €13.8 million H1 2016 Sales = €14.8 million Increase = […]
Spine Wave Announces the Commercial Launch of the True Position® Pivoting Spacer System
SHELTON, CT–(Marketwired – July 11, 2016) – Spine Wave, Inc. is committed to the commercialization of high-quality, innovative medical devices for the treatment of spinal disorders. Spine Wave’s commitment was demonstrated by its previously announced acquisition of rights in the True Position® Pivoting Spacer System from Atlas Spine on February 23, 2016. Since then, Spine […]
Titan Spine Secures CMS New Technology ICD-10 Code for Nanotextured Surface on Interbody Fusion Devices
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative surface-enhanced spinal interbody fusion implants, reports that the U.S. Centers for Medicare & Medicaid Services (CMS) has announced the creation of a new technology ICD-10 code for a nanotextured surface on an interbody fusion device. Titan Spine’s nanoLOCK™ surface technology, which […]
Amedica Announces Closing of $12.7 Million Public Offering and Full Exercise of Over-Allotment Option
SALT LAKE CITY, UT–(Marketwired – Jul 8, 2016) – Amedica Corporation (NASDAQ: AMDA), a company that develops and commercializes silicon nitride ceramics, today announced it has closed the underwritten public offering of $12.7 million at a price to the public of $1.00 per unit of one share and one warrant, which included the exercise in […]
Precision Spine® Names Christopher A. DeNicola Chief Operating Officer as Company Enters Next Phase of Aggressive Growth
PARSIPPANY, N.J.–(BUSINESS WIRE)–Precision Spine, Inc., a medical device company dedicated to Made-in-the-USA manufacturing, announced that, in support of the company’s accelerating growth plan, it has named Christopher A. DeNicola its new Chief Operating Officer. “Chris is a seasoned leader in the orthopedic medical device industry and he has been instrumental in building our domestic and […]
Merit Medical Acquires DFINE, Inc.
SOUTH JORDAN, Utah, July 06, 2016 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ:MMSI), a leading manufacturer and marketer of proprietary disposable devices used primarily in cardiology, radiology and endoscopy, today announced that it has acquired DFINE, Inc. headquartered in San Jose, California, in a merger transaction through which DFINE has become a wholly-owned subsidiary […]
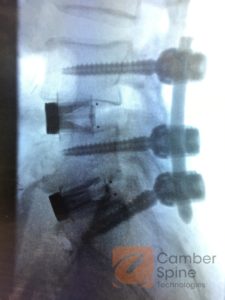
Camber Spine Technologies Announces First implantation of ENZA™ Zero-Profile ALIF Device
WAYNE, Pa., July 5, 2016, – Camber Spine Technologies, today announced the first implantation of the ENZA™ MIS Zero-Profile anterior lumbar interbody fusion (ALIF) device in a two-level lumbar procedure at levels L4/L5 and L5/S1 at Kennedy University, Washington Township, by Dr. Jeffrey Gleimer, D.O. ENZA™ is the company’s first device launched with integrated fixation […]
Amedica Announces Pricing of $11 Million Public Offering
SALT LAKE CITY, UT–(Marketwired – Jul 5, 2016) – Amedica Corporation (NASDAQ: AMDA), a company that develops and commercializes silicon nitride ceramics, today announced the pricing of an underwritten public offering of units for gross proceeds of $11 million, prior to deducting underwriting discounts and commissions and offering expenses payable by Amedica. The offering is comprised […]
SpineCraft Obtains Regulatory Clearance to market the APEX Spine System® in China, the second-largest spine market in the world.
SpineCraft announced today that it has received clearance from the China Food and Drug Administration (CFDA) to market its APEX Spine System® in China. The APEX Spine System is designed for complex deformity correction as well as tumor, trauma and degenerative procedures and has proven clinical track record with over 15,000 surgeries performed since its […]
7th Anniversary | SPINEMarketGroup
Launched in July 2009, we’re now celebrating our 7th Anniversary! Thank you very much to all our visitors and followers! In all these years, we have published more than 2.500 news and provided information related to the Spinal market including Spinal Companies, Interspinous Devices, MIS devices, LLIF, TLIF cages, Artificial Cervical Discs, Dynamic implants , SI Joint Fusion Devices, […]
Updated Spine Catalogue 2016 with 7 more Spinal Companies (347)
Today we have updated our Spine Catalogue with 7 more Companies. Now, we have 347 manufacturers in our list. Also, we have included the one more system in the list of 14 SI fusion devices. 1.- List of the 7 companies Included: Alethea-spine evospine Southern-medical CTL Medical-corporation X-spine Flospine Cardinal-spine 2.- New SI Fusion Device Included: Prolix […]
NuVasive Agrees to Settle Patent Litigation With Medtronic
SAN DIEGO, CA–(Marketwired – June 30, 2016) – NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery, today announced that the Company has agreed to settle its ongoing patent litigation with Medtronic and certain of its subsidiaries. NuVasive and Medtronic reached agreement on terms for the settlement of the previously […]