AUDUBON, Pa., Oct. 25, 2016 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal implant manufacturer, today announced the introduction of the INDEPENDENCE MIS™ System, an integrated ALIF plate-spacer system designed to simplify implantation and fixation of a commonly challenging procedure. The INDEPENDENCE MIS™ system features advanced instruments that deploy three pre-loaded anchors through […]
NEWS
Zimmer Biomet’s Mobi-C® Cervical Disc Becomes Most Widely Covered Intervertebral Disc Prosthesis for Cervical Disc Replacement
WARSAW, Ind., Oct. 25, 2016 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced that its Mobi-C® Cervical Disc Prosthesis is now the most widely covered device for one- and two-level cervical disc replacement by commercial health insurers in the United States. Mobi-C, acquired as part […]
Globus Medical Launches QUARTEX™ OCT Stabilization System
AUDUBON, Pa., Oct. 25, 2016 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal implant manufacturer, continues to innovate in the treatment of complex spinal instability and deformity with today’s announcement of the launch of the QUARTEX™ OCT Stabilization System. QUARTEX™ offers a variety of solutions to the challenges associated with posterior OCT fusion […]
Vertera Spine Reports Successful Early Outcomes with the Porous PEEK COHERE Fusion Device
ATLANTA, Oct. 24, 2016 /PRNewswire/ — Vertera Spine, a developer of medical devices using advanced biomaterial technologies, today announced the first early successful outcomes using the COHERE® Cervical Interbody Fusion Device. COHERE is Vertera Spine’s first device featuring the company’s patented porous PEEK (polyetherether ketone) Scoria® biomaterial technology. Scoria’s porous architecture seamlessly integrates with the […]
Spine Wave Announces the Commercial Launch of the Velocity® P Expandable Interbody Device
SHELTON, CT–(Marketwired – October 24, 2016) – Spine Wave, Inc. is committed to the commercialization of high-quality, innovative medical devices for the treatment of spinal disorders. Spine Wave is pleased to announce that it will be fully launching the Velocity® P Expandable Interbody Device system in mid-November with the arrival of commercial-scale inventory quantities. The […]
Spinal Elements® Announces Initial Procedures with Katana™ MIS Lateral System
CARLSBAD, Calif.–(BUSINESS WIRE)–Spinal Elements, a spine technology company, announced that the first procedures with its minimally invasive (MIS) Katana Lateral System were successfully completed. The Katana Lateral System is a muscle-splitting system that is designed to overcome some of the inherent challenges in MIS lateral surgery. Traditionally, spine surgery has been performed from the back […]
DeGen Medical Launches Latitude-C™ Porous Ti Cervical Interbody Spacer
FLORENCE, SC–(Marketwired – October 24, 2016) – DeGen Medical introduces Latitude-C™ Porous Ti Cervical Interbody Spacer, an extension of our popular and effective Latitude-C™ product line, is designed to consider the uncinate process of the cervical vertebra. The lateral angled shape is a shift from traditional rectangular shape and mimics patient anatomy. The variety of […]
Medicrea Announces 2 FDA Clearances of New Specialized Components for Pediatric Deformity on PASS® and LigaPASS® Platforms for Complex Spine in Younger Patients
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Alternext Paris: FR0004178572 – ALMED), worldwide leader pioneering the development and manufacture of personalized analytical services and implant solutions for the treatment of complex spinal conditions, today announced that it has received two unique 510(k) clearances from the U.S. Food and Drug Administration (FDA) for its PASS® […]
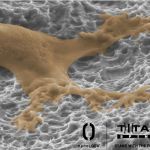
Titan Spine Expands Sales Force to Meet Growing Demand, Support Recent Full Launch of its New nanoLOCK® Surface Technology
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced that it has doubled its sales management team with the addition of nine new members to meet the growing demand for its Endoskeleton® line of titanium interbody fusion devices and to support the recent full […]
Benvenue Medical’s Luna 3D Interbody Fusion System to Be Featured in Technology Innovation Session at NASS 2016
SANTA CLARA, Calif.–(BUSINESS WIRE)–Benvenue Medical, Inc., a developer of minimally invasive solutions for spine repair, today announced key presentations and company events featuring the Luna® 3D Multi-Expandable Interbody Fusion System and Kiva® VCF Treatment System at the North American Spine Society (NASS) 31st Annual Meeting taking place Oct. 26-29 in Boston. Benvenue is one of […]
A Randomized Study Confirms the Benefits of Safe Orthopaedics’ Products in a Leading French Teaching Hospital
Safe Orthopaedics (Paris:SAFOR) (FR0012452746 – SAFOR), a company offering innovative ranges of sterile implants combined with their single-use instruments for back surgery, is today announcing the presentation of a randomized study confirming the benefits of its single-use instruments for spine surgery. This 1:1 randomized study of 40 surgeries was conducted by teams at CHU Bicêtre, […]
Tritanium® Posterior Lumbar Cage To Be Demonstrated at NASS 2016
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division will demonstrate its 3D-printed Tritanium Posterior Lumbar (PL) Cage, an interbody fusion device designed to aid in lumbar spinal fixation for patients with degenerative disc disease, including up to Grade 1 spondylolisthesis, and degenerative scoliosis, at the North American Spine Society (NASS) Annual Meeting, Oct. 26-29, 2016, in Boston (booth […]
Amendia Partners with Global Spine Outreach
MARIETTA, GA (PRWEB) OCTOBER 19, 2016–Leading provider of innovative medical devices Amendia, Inc. is pleased to announce its partnership with and support of Global Spine Outreach (“GSO”), a charitable organization dedicated to establishing first-rate scoliosis treatment centers worldwide. Amendia is the primary industry sponsor of the Third Annual Spinal Deformity Symposium, to be held October […]
Precision Spine® to introduce the MD-Vue™ Lateral Access System at the North American Spine Society’s 31st Annual Meeting in Boston
PARSIPPANY, N.J.–(BUSINESS WIRE)–Precision Spine, Inc., a medical device company dedicated to Made-in-the-USA manufacturing, will introduce its MD-Vue™ Lateral Access System at the North American Spine Society (NASS) Annual Meeting October 26-29 in Boston, MA. The strong, lightweight MD-Vue System was designed in collaboration with prominent lateral approach spine surgeons and is the only lateral retractor […]
SpineGuard® expands “PediGuard®” franchise, will launch “PediGuard Threaded” drilling device at North American Spine Society (NASS) annual meeting
PARIS & SAN FRANCISCO: SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD) announced today that it will launch its next-generation PediGuard “Threaded” device enabled by DSG™ (Dynamic Surgical Guidance) technology at next week’s 31st annual meeting of the North American Spine Society (NASS) in Boston on October 26-29. The innovative PediGuard Threaded device with DSG is a drilling instrument […]
Alphatec Spine to Showcase New Minimally Invasive (MIS) and Complex Spine Products at the Upcoming North American Spine Society (NASS)
CARLSBAD, Calif., Oct. 18, 2016 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (Nasdaq:ATEC), the parent company of Alphatec Spine, Inc., a provider of spinal fusion technologies, announced today that it will introduce several new products, as well as its current comprehensive portfolio at the upcoming North American Spine Society (NASS) meeting at the Boston Convention and […]
InterMed Resources distributes for Z-Medical and Announce Agreement with HealthTrust
InterMed Resources will distribute spinal implants manufactured by Z-Medical, a company based in Tuttlingen, Germany to HealthTrust (legally known as HealthTrust Purchasing Group, L.P.), a leading group purchasing and total cost management organization that serves nearly 1,600 acute care facilities nationwide. Under the agreement, Z-Medical will sell its MIS Z-Pedicle Screw System at a contracted […]
Spineology Completes Limited Release for Elite™ Expandable Interbody Fusion System
MINNEAPOLIS & ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology is excited to announce the successful completion of the Elite™ Expandable Interbody Fusion System limited release. The Elite Expanding Interbody Implant is inserted into the disc space in a contracted minimal height to minimize dissection and neural retraction. It is then incrementally expanded to restore the anatomical height of […]
Nexxt Spine: Taking Action to Reduce Post-Operative Surgical Infection
NOBLESVILLE, Ind.–(BUSINESS WIRE)–Nexxt Spine, LLC, a privately held medical device company dedicated to increasing procedural efficiency and clinical outcomes for patients with debilitating spinal conditions, further demonstrates the company’s on-going commitment toward reducing post-operative surgical infections with the addition of an ISO Class 10,000 (International Standards Organization) Certified Cleanroom for pre-packaging sterile spinal implants within […]
Zyga Releases 12-month Fusion and Clinical Results of Sacroiliac Joint Fusion with Decortication Following Presentation by Dr. William Cross at 2016 SMISS Annual Meeting
MINNETONKA, Minn., Oct. 17, 2016 /PRNewswire/ — Zyga Technology, Inc., a medical device company focused on the design, development and commercialization of minimally invasive devices to treat underserved conditions of the lumbar spine, today released 12-month fusion and clinical results for patients receiving SImmetry® Sacroiliac (SI) Joint Fusion with decortication. Study results were presented on Saturday […]
Vexim: +37% Sales Increase to €13.3 Million as of September 30, 2016 (9 Months)
VEXIM (Paris:ALVXM) (FR0011072602 – ALVXM / PEA‐PME), a medical device company specializing in the minimally invasive treatment of vertebral fractures, today announces its consolidated sales results for the third quarter 2016. « Vexim continues to develop according to its plan. Our results confirm the objectives set as the SpineJack® is becoming a reference on the […]
EOS imaging Enters the North African Market with the First Two EOS Systems in Tunisia
PARIS–(BUSINESS WIRE)–EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI), the pioneer in 2D/3D orthopedic medical imaging, announced today the recent entry into the North African market with the opening of two EOS sites in Tunisia. The systems are operated by the public Institut Mohamed Kassab d’Orthopédie and the private Centre d’Imagerie Médicale Ariana in the Tunis […]
Titan Spine Initiates Full U.S. Launch of New nanoLOCK® Surface Technology
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced that it has expanded the distribution of its line of Endoskeleton®titanium implants featuring the company’s new proprietary nanoLOCK® surface technology nationwide. The full U.S. launch follows the successful alpha introduction of the nanoLOCK® technology initiated […]
Medovex Reports Successful Demonstration of Expanding EMEA Business at EuroSpine 2016, Berlin
ATLANTA, GA–(Marketwired – Oct 13, 2016) – Medovex Corp. (NASDAQ: MDVX), a developer of medical technology products, today announced that the Company successfully exhibited its’ DenerveX™ System during EuroSpine 2016 Tradeshow last week in Berlin, Germany. As part of the product exhibition, Medovex highlighted its innovative DenerveX™ System, a minimally invasive surgical (MIS) procedure pioneered […]
Which are the Trends and the 365 players operating in the Spinal market?
In 2015, North America held the largest share of the spinal implants and devices market accounting for nearly 63%. The presence of advanced healthcare facilities, strong reimbursement support, and a large number of people suffering with spinal disorders are some of the major factors accounting for this region’s dominance. The key players operating in the […]
SpineGuard reports €1.7M revenue for 3Q 2016, and 9 months growth of 15%
PARIS and SAN FRANCISCO, Oct. 12, 2016 – SpineGuard (FR0011464452 – ALSGD), an innovative company that designs, develops, and markets disposable medical devices intended to make spine surgery safer, announced today that its third quarter revenue grew to €1.7 million, a 2% increase compared with 3Q 2015. Pierre Jérôme, CEO and co-founder of SpineGuard, said: […]
ChoiceSpine Announces the Appointment of Christy Cote as Vice President of Biologics
KNOXVILLE, TN, October 5, 2016 – ChoiceSpine, a Knoxville, TN based spinal implant company, today announced it has appointed Christy Cote as Vice President of Biologics. She is a business leader with 14+ years’ experience in stem cell technology, surgical implants, and regenerative therapies. She’s had an award winning sales career and brings with her […]