ISTANBUL, TURKEY (PRWEB) SEPTEMBER 06, 2017–Inspired Spine presented research on four groundbreaking studies in Istanbul, Turkey at the World Congress of Neurosurgery Meeting. The meeting included accomplished neurosurgeons from all over the world, with Dr. Hamid Abbasi representing Inspired Spine on four studies. The four research studies looked at various innovative minimally invasive spine surgery […]
NEWS
EOS imaging Introduces Personalized Biomechanical Simulation in Spine Surgery Planning
PARIS–(BUSINESS WIRE)–EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI), the pioneer in 2D/3D orthopedic medical imaging, today announced that it will introduce a new version of spineEOS, the Company’s online 3D planning software for spine surgery based on EOS stereo-radiographic 2D/3D imaging. spineEOS is the first software to incorporate biomechanical patient data into surgery simulation and planning […]
Medtronic Launches Long Term Clinical Study Program of INFUSE Bone Graft in Two Common Spine Procedures: PLF and TLIF
DUBLIN – September 5, 2017 – Medtronic plc (NYSE: MDT) today announced the launch of a long term clinical study program to collect prospective data on INFUSE® Bone Graft in Posterolateral Fusion (PLF) and Transforaminal Lumbar Interbody Fusion (TLIF) spine procedures. The first patient has been enrolled in the PLF study at Fort Wayne Orthopedics […]
ApiFix Signs Distribution Agreement in Canada; Completes First Cases in North America at IWK Health Centre Hospital, Halifax
MISGAV, Israel, Sept. 5, 2017 /PRNewswire/ — Medical device company ApiFix Ltd., a portfolio company of The Trendlines Group (SGX: 42T) (OTCQX: TRNLY), announced it signed an exclusive distribution agreement in Canada with Joint Solutions Alliance Corp (JSA), a national Canadian medical device, and services distribution organization. JSA’s primary focus pertains to finding medical devices […]
Implanet Presents Independent Radiological Results of the JAZZ Band at the SRS 52nd Annual Meeting & Course
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–Regulatory News: IMPLANET (Euronext Growth: ALIMP, FR0010458729, PEA-PME eligible) (Paris:IMPL) (OTCQX:IMPZY), a medical Technology company specializing in vertebral and knee-surgery implants, announces the publication of a White Paper entitled “How to optimize axial correction without altering thoracic sagittal alignment in hybrid constructs with sublaminar bands: description of the ‘frame technique’”, which presents […]
joimax®, Inc. Opens Its First Education & Training Center in Irvine, California, Posts Record Sales in the US and Continues to Make Gains with Endoscopic Reimbursement
IRVINE, Calif.–(BUSINESS WIRE)–joimax® announces the grand opening of its new state-of-the-art Training and Education Center in Irvine, California. The company will be holding its first workshop at the Center on September 8-9th. As the market leader in endoscopic spine surgery, joimax® is fully dedicated to outstanding physician education, training and services worldwide. Due to the […]
Globus Medical Appoints David M. Demski as CEO
AUDUBON, Penn., Aug. 29, 2017 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced that, the Board of Directors has named David M. Demski as the Company’s Chief Executive Officer, effective immediately. Mr. Demski will report to David C. Paul, the Company’s founder, Chairman of the Board, and Chief Executive […]
Mazor Robotics and Medtronic Enter Next Phase of Strategic Partnership; Medtronic to Make a $40 million Third Tranche Investment in Mazor
CAESAREA, Israel–(BUSINESS WIRE)–Mazor Robotics Ltd. (TASE: MZOR; NASDAQGM: MZOR), a pioneer and a leader in the field of surgical guidance systems, today announced that it has entered the next phase of its strategic partnership with Medtronic earlier than planned and their existing agreements have been amended accordingly. The agreements provide for the conversion of the […]
OrthoPediatrics and Mighty Oak Medical Team Up to Bring Pediatric-Friendly Spinal Navigation to Children’s Hospitals
WARSAW, Ind., Aug. 28, 2017 (GLOBE NEWSWIRE) — OrthoPediatrics and Mighty Oak Medical are pleased to announce a navigation partnership that can provide unsurpassed screw placement accuracy in the pediatric population. Mighty Oak Medical is the manufacturer of FIREFLY® Pedicle Screw Navigation Guides, which are 3D printed and patient-specific at each planned vertebral level. FIREFLY® Guides address […]
ChoiceSpine™ Successfully Completed 1,000th TOMCAT™ ACDF Procedure
KNOXVILLE, TN (PRWEB) AUGUST 23, 2017–ChoiceSpine LP, a privately-held spinal device manufacturer based in Knoxville, TN, successfully completed its 1,000th TOMCAT Anterior Cervical Discectomy and Fusion (ACDF) procedure. The procedure was performed by Julian Price, MD of Athens Orthopedic Clinic, Athens, GA at St. Mary’s Hospital, also in Athens, on August 14, 2017. “I choose […]
EIT Emerging Implant Technologies Acquires 22 Patents for 3D Printed Expandable Cage Technology Based on Living Hinges from Dr. Morgan Lorio
WURMLINGEN, GERMANY (PRWEB) AUGUST 22, 2017–Emerging Implant Technologies GmbH (EIT), a German medical device manufacturer exclusively focused on creating innovative technologies for spinal applications by utilizing additive manufacturing (“3D-Printing”), today announced the acquisition of a full patent portfolio of 22 issued and pending patents for 3D printed expandable spinal fusion cages based on living hinges […]
Cutting Edge Spine Appoints Luke Maher as Director U.S. Sales
WAXHAW, NC (PRWEB) AUGUST 22, 2017–Cutting Edge Spine, a technology-driven spinal device company focused on the development and distribution of highly differentiated spinal implant technologies, announced today the appointment of Luke Maher as Director U.S. Sales. Luke Maher brings 15 years of spinal device industry experience to directing Cutting Edge Spine’s national sales force, including […]
Life Spine Announces First Cases Performed with Multiple Methods of In-situ Expansion
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today the first cases performed with multiple methods of in-situ expansion. “Surgical intervention for the degenerative spine is a multi-dimensional challenge and by utilizing two methods of expansion, Life Spine allowed for […]
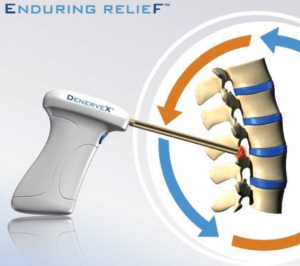
Medovex Corporation Appoints Gorlin Companies Managing Partner Jesse Crowne Director and Executive Co-Chairman of the Board
ATLANTA, GA–(Marketwired – Aug 21, 2017) – Medovex Corp. (NASDAQ: MDVX) (“Medovex” or the “Company”), the developer of the DenerveX™ System, a new and novel device designed for enduring relief of Facet Joint Syndrome related to back pain, today announced it has appointed Jesse Crowne as Director and Executive Co-Chairman of the Board. Jesse Crowne […]
Minimus Spine: Dr. Alexis Kelekis to Present an Update on European Study at CIRSE 2017 Next Month in Copenhagen
Minimus Spine announced that Dr. Alexis Kelekis, an interventional radiologist practicing at Attikon University Hospital in Athens Greece, will be presenting an update on an ongoing study comparing Triojection® to surgical discectomy at CIRSE 2017. Triojection was developed by Minimus Spine and is intended to facilitate an injection of ozone gas as a treatment for […]
Globus Medical Announces FDA 510(k) Clearance for Excelsius GPS™
AUDUBON, Pa., Aug. 17, 2017 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced that the Excelsius GPS™, a revolutionary robotic guidance and navigation system, has been 510(k) cleared by the U.S. Food and Drug Administration. This platform technology supports minimally invasive and open orthopedic and neurosurgical procedures, with screw […]
Stryker’s Spine Division Receives FDA Clearance for Serrato Pedicle Screw
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division today announced that its Serrato™ Pedicle Screw, intended for use in the non-cervical spine as part of the company’s successful Xia® 3 Spinal System, has received 510(k) clearance from the U.S. Food and Drug Administration. Serrato Pedicle Screws feature enhanced serrated cutting flutes, a unique dual-thread pattern with an increased […]
Spine Wave Announces the Commercial Launch of the Proficient® Posterior Cervical Spine System
SHELTON, Conn., Aug. 15, 2017 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce its entry into the posterior cervical fixation market with the successful completion of a limited market release of the Proficient® Posterior Cervical Spine System. The Proficient® Posterior Cervical Spine System substantially strengthens Spine Wave’s position in the cervical spine fixation market […]
NuVasive Appoints Rajesh J. Asarpota As Chief Financial Officer
SAN DIEGO, Aug. 15, 2017 /PRNewswire/ — NuVasive, Inc. (Nasdaq: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, announced today the appointment of Rajesh (Raj) J. Asarpota as the Company’s new executive vice president and chief financial officer (CFO), effective September 1, 2017. As a member of […]
Camber Spine Technologies Announces FDA Clearance And National Launch Of SPIRA™ Open Matrix ALIF
WAYNE, Pa., Aug. 15, 2017 /PRNewswire/ — Camber Spine Technologies announced today that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its SPIRA™ Open Matrix ALIF device, a unique, interbody fusion implant consisting of spiral support arches and Surface by Design™ technology. This clearance marks Camber’s tenth line […]
Expanding Orthopedics Inc. Granted Two Additional US Patents in the Expandable Interbody Domain
OR-AKIVA, Israel, August 14, 2017 /PRNewswire/ –Expanding Orthopedics Inc. (EOI), a medical device company focused on developing and commercializing innovative expandable devices for spine surgery, is excited to announce that it has been granted two additional US Patents by the USPTO covering its unique and diverse expandable cage technology, strengthening its position in the expandable […]
OrthoPediatrics Corp. Files Registration Statement for Proposed Initial Public Offering
WARSAW, Ind., Aug. 11, 2017 (GLOBE NEWSWIRE) — OrthoPediatrics Corp. (“OrthoPediatrics”) announced today that it has publicly filed an amendment to its registration statement on Form S-1 with the U.S. Securities and Exchange Commission (the “SEC”) relating to a proposed initial public offering of its common stock. OrthoPediatrics plans to list its common stock on […]
Renovis® Surgical Receives FDA Clearance for 3D-Printed Posterior Lumbar Interbody Fusion Systems
REDLANDS, Calif., Aug. 11, 2017 /PRNewswire/ — Renovis Surgical Technologies, Inc. announced today that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market posterior lumbar Tesera® porous titanium interbody fusion systems. These systems feature implants for direct posterior (PLIF) or transforaminal (TLIF) approaches in both straight and curved options. Multiple heights, widths […]
Alphatec Holdings Second Quarter 2017 Financial Highlights
CARLSBAD, Calif., Aug. 10, 2017 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“Alphatec” or the “Company”) (Nasdaq:ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, announced today recent corporate highlights and financial results for its second quarter ended June 30, 2017. Second Quarter […]
SI-BONE, Inc. Announces Publication of a Level I Study Demonstrating Strong Clinical Prediction for the Diagnosis of SI Joint Dysfunction
SAN JOSE, Calif., Aug. 9, 2017 /PRNewswire/ — SI-BONE, Inc., an innovative medical device company that pioneered the use of the iFuse Implant System® (iFuse), a triangular shaped minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced the publication of a systematic review of diagnostic accuracy studies […]
Xtant Medical Receives FDA Clearance for Calix-C Cervical Interbody Line Extension and Expanded Indications
BELGRADE, Mont., Aug. 09, 2017 (GLOBE NEWSWIRE) — Xtant Medical Holdings, Inc. (NYSE American:XTNT), a leader in the development of regenerative medicine products and medical devices, today announced that the U.S. Food and Drug Administration (FDA) has cleared product line extensions for the Calix-C family of cervical interbody cages. The clearance provides for the addition […]
The ApiFix Spinal Implant Receives TGA Certification
MISGAV, Israel, Aug. 8, 2017 /PRNewswire/ — ApiFix, a portfolio company of The Trendlines Group, received TGA certification through its distributor Orthotech Pty. Ltd. to begin marketing the ApiFix system in Australia for the treatment and correction of Adolescent Idiopathic Scoliosis (AIS) using an innovative, minimally invasive surgical approach. The ApiFix system represents a breakthrough treatment for Adolescent Idiopathic Scoliosis (AIS) as it is a […]