SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices to make spine surgery safer, announced today the successful completion of an experimental feasibility study in collaboration with the Institut des Systèmes Intelligents et de Robotique (UPMC /CNRS), Paris, France. The study demonstrates how DSG™ technology can stop a robot automatically when an impending […]
NEWS
Is the Lumbar Total Disc Replacement (TDR) Back? Learn about the 10 most relevant Lumbar Artificial Discs
The lumbar artificial disc (LAD) market has experienced significantly lower procedural growth than other market segments as a result of a lack in clinical efficacy data and poor reimbursement. In 2014, the reimbursement environment severely limited the number of LAD procedures performed. However, reimbursement policies are expected to change because Medicare and other organizations are considering […]
NASS 2017 Best Paper Sessions Provide Significant Insights into the Long-Term Results of activL® Artificial Disc
CENTER VALLEY, Pa., Nov. 9, 2017 /PRNewswire/ — Aesculap Implant Systems, LLC today announced the significance of the long-term data presented at the NASS 2017 podium during the NASS Annual Meeting in Orlando, FL, October 25-27, 2017. At the meeting, three key presentations were made that greatly increase the body of evidence in support of lumbar total disc replacement (TDR) […]
François-Henri Reynaud Appointed as Safe Orthopaedics’ Chief Financial Officer
ERAGNY-SUR-OISE, France– SAFE ORTHOPAEDICS (Paris:SAFOR) (FR0012452746 – SAFOR), a company offering innovative ranges of sterile implants combined with their single-use instruments for back surgery, today announces the appointment of François-Henri Reynaud as Chief Financial and Administrative Officer. François-Henri has 13 years’ experience in finance. For five years, he was initially involved in financing innovative business companies in a […]
Mazor Robotics Reports Record Third Quarter 2017; Revenue More Than Doubles to $17.2 Million
CAESAREA, Israel–(BUSINESS WIRE)–Mazor Robotics Ltd. (TASE: MZOR; NASDAQGM: MZOR), a pioneer and a leader in the field of robotic guidance systems, reported record third quarter revenue of $17.2 million. As previously announced, the Company received purchase orders for 22 systems in the 2017 third quarter, including 19 Mazor X systems. Recent Significant Highlights Entered the […]
SPINEWAY :Joseph Brigneaud nominated to Spineway’s Board of Directors
The Board of Directors of Spineway, specialist in surgical implants and instruments for treating disorders of the spinal column (spine), has co-opted Mr. Joseph Brigneaud as an independent Board member. Joseph Brigneaud, a partner at MAELO Capital – a private-equity investment firm, has over 15 years’ experience in supporting and financing companies. Having opened and […]
SAGICO Announces North American Launch of Expandable Spinal Implants
TAMPA, Fla., /PRNewswire-iReach/ — Spinal Analytics & Geometrical Implant Co, (SAGICO), an innovative spinal implant company, announced today the North American debut of its proprietary spinal restoration technology. The North American debut of the innovative SAGICO IBF System, Aria Lumbar and Arion Cervical, introduces expandable interbody fusion devices during the North American Spine Society (NASS) annual […]
Amedica Submits 510(k) Application to FDA for Valeo C+CSC with Lumen
SALT LAKE CITY, Nov. 06, 2017 (GLOBE NEWSWIRE) — Amedica Corporation (Nasdaq:AMDA), an innovative biomaterial company that develops and manufactures silicon nitride as a platform for biomedical applications, announced that the company made a 510(k) submission to the U.S Food and Drug Administration for its Valeo C+CSC with Lumen spinal implant. The Valeo C+CSC with Lumen […]
Medical Device Sales Reps in the Operating Room: An Asset or a Liability?
November 1, 2017 (mddionline.com)–In this age of rapidly developing technologies, medical device sales representatives fill a unique role. In the business of surgery, sales reps not only deliver medical devices to hospitals as needed, but they ensure that surgical teams know precisely when and how a device and its components are to be used. Often knowing […]
SeaSpine Reports Third Quarter 2017 Financial Results
CARLSBAD, Calif., Nov. 02, 2017 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ:SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today financial results for the third quarter ended September 30, 2017. Third Quarter 2017 Financial Highlights and Recent Accomplishments Revenue of $31.7 million, unchanged compared to the prior […]
New Studies Look At Cost And Benefits Of Robotic Surgery
As robotics in medicine becomes more widely adopted, two new studies look at the cost and advantages and disadvantages of robotic surgery versus freehand surgery. University of Stanford researchers conducted a multiyear analysis and study with 24,000 patients with kidney cancer who needed laparoscopic surgery to remove a patient’s kidney indicated that the two approaches had comparable patient outcomes and […]
SeaSpine Announces Limited Commercial Launch of OsteoStrand™ Demineralized Bone Fibers
CARLSBAD, Calif., Nov. 01, 2017 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ:SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the limited commercial launch and completion of initial surgeries for its OsteoStrand™ Demineralized Bone Fibers. The first implantations were completed by Dr. Khalid Abbed, Vice Chair […]
Atlas Spine Receives FDA Clearance for Ortus Expandable Posterior Lumbar Interbody System
PR Newswire11.01.17–Atlas Spine Inc., a high technology, spinal implant and instrumentation company based in Jupiter, Fla., has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Ortus Expandable Posterior Lumbar Interbody System. The device is designed to be delivered using either minimally invasive or open surgical approach. The Ortus PL is clearly differentiated […]
K2M Group Holdings, Inc. Reports Third Quarter 2017 Financial Results
LEESBURG, Va., Nov. 01, 2017 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (Nasdaq:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today reported financial results for its third fiscal quarter ended September 30, 2017. Third Quarter 2017 Financial Summary: Total Q3 revenue […]
Surgeon sues J&J’s DePuy in royalties dispute
OCTOBER 31, 2017 (massdevice.com)–A surgeon who helped Johnson & Johnson (NYSE:JNJ) subsidiary DePuy Synthes Spine develop instruments and implants for spine procedures wants a Massachusetts federal court to overturn an arbitrator’s ruling that the company doesn’t owe royalties on the products, according to a lawsuit filed last week in the Bay State. Dr. Carl Lauryssen and his company, Jordan […]
Pinnacle Spine Group Files Application with FDA for 510(k) Clearance to Market their InFill® Lateral Fusion System with PEEK-OPTIMA™ HA Enhanced Polymer
DALLAS & THORNTON CLEVELEYS, UK (PRWEB) OCTOBER 31, 2017–Pinnacle Spine Group, LLC, the pioneer of in situ graft delivery in spinal fusion, today announced it has filed an application with the Food and Drug Administration (FDA) seeking 510(k) clearance to market its lateral InFill® Lumbar Fusion Systems with PEEK-OPTIMA HA Enhanced polymer from Invibio Biomaterial […]
Exactech to be acquired by private equity firm
(Gainesville.com)–After 21 years as a public company, Exactech, a Gainesville-based medical device company, announced Monday it agreed to be acquired by TPG Capital, a global private equity platform of alternative asset firm TPG — making it private. Exactech’s board of directors approved the agreement, which says TPG Capital will acquire all common stock shares of […]
4WEB Medical Demonstrates Increased Subsidence Resistance for Lateral Spine Truss System Compared to Annular Implants
ORLANDO, Fla., Oct. 26, 2017 /PRNewswire/ — 4WEB Medical, the industry leader in 3D printed implant technology, announced the results of a new study demonstrating reduced subsidence for its Lateral Spine Truss System (LSTS) as compared to annular interbody implant designs today at the annual meeting of the North American Spine Society. The study was […]
EOI Announces Initial Surgeries With the FLXfit™15, the First Articulated and Expandable Cage With 15 Degrees of Lordosis Correction
OR AKIVA, Israel, Oct. 26, 2017 /PRNewswire/ –Expanding Orthopedics Inc. (EOI), a privately held medical device company focused on developing and commercializing innovative expandable devices for spine surgery, today announced first surgeries with its enhanced 3D expandable cage, the FLXfit™15. The FLXfit™15 offers enhanced performance and versatility by providing additional device sizes with the ability […]
NuVasive Unveils Latest Innovation At NASS 2017
SAN DIEGO, Oct. 25, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced it will highlight the Company’s latest disruptive spine technology innovation at the North American Spine Society (NASS) Annual Meeting held October 25-28, 2017 in Orlando, Fla. At the event, NuVasive will also celebrate […]
AxioMed Announces Release of Lateral Viscoelastic Total Disc Replacement at NASS 2017
BOSTON, MASS. (PRWEB) OCTOBER 25, 2017–AxioMed announces the release of its lateral total disc replacement at the North American Spine Society (NASS) 2017 Annual Meeting. The company is expecting the first surgery to be done in Jamaica by Dr. Kingsley R. Chin before the end of this year. AxioMed has hired two top executives – Chief […]
GS Medical USA Announces New Product Portfolio of Spinal Solutions
IRVINE, Calif., Oct. 25, 2017 /PRNewswire/ — Today, GS Solutions Inc., DBA GS Medical USA (GSM), a U.S. supplier of spinal implants and instrumentation and provider of high-quality surgical solutions, announced the full launch of the AnyPlus® Direct Lateral Interbody Fusion (DLIF) System after three months of an alpha launch period, and the Anterior and […]
K2M & Brainlab Cooperate Towards an Adapted Version of K2M Spinal Systems Compatible with Brainlab Navigation
LEESBURG, Va., Oct. 25, 2017 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, and Brainlab, a leader in medical technology, today announced a global compatibility and co-marketing agreement. The two companies will collaborate in […]
ChoiceSpine™ Granted FDA Clearance for Revolutionary Cervical Spine Interbody Fusion Device
KNOXVILLE, TN (PRWEB) OCTOBER 25, 2017–ChoiceSpine LP, a privately-held spinal device manufacturer based in Knoxville, TN, today announced the U.S. Food and Drug Administration (FDA) clearance (510k) of the revolutionary Blackhawk integrated anterior cervical fusion device featuring self-locking anchors and a large center graft chamber. “The Blackhawk clearance expands the ChoiceSpine cervical fusion product portfolio […]
New DePuy Synthes Spine Technologies Help Simplify Minimally Invasive Surgery by Enhancing Efficiency and Reducing Steps
ORLANDO, Fla., Oct. 25, 2017 /PRNewswire/ — DePuy Synthes* today is launching two solutions for minimally invasive spinal fusion surgery (MIS) designed to simplify the procedure, particularly as MIS gains traction and shows reduced complications compared to open surgery in treating degenerative disc disease in the back.1,2 Research shows the number of MIS fusion surgeries […]
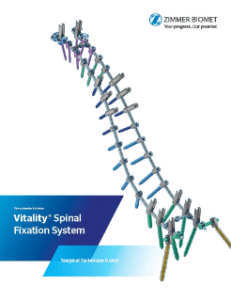
Zimmer Biomet Announces U.S. Launch of Vitality®+ and Vital™ Spinal Fixation Systems
WARSAW, Ind., Oct. 25, 2017 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced the official launch of its Vitality®+ and Vital™ Spinal Fixation Systems in the United States at the 2017 North American Spine Society (NASS) Annual Meeting. The comprehensive Vitality+ Spinal Fixation System consists […]
RTI Surgical® Launches Fortilink®-C IBF System, the First 3D Printed Polymer-based Interbody Device
ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (RTI) (Nasdaq: RTIX), a global surgical implant company, today announced the commercial launch of Fortilink-C IBF System with TETRAfuse® 3D Technology. The Fortilink-C system is the first in a family of devices to incorporate RTI Surgical’s TETRAfuse 3D Technology. “TETRAfuse 3D Technology combines the best characteristics of titanium, allograft bone […]