Safe Orthopaedics (Paris:ALSAF) (FR0012452746 – ALSAF), a company specializing in the design and marketing of single-use implants and instruments for the minimally-invasive treatment of spine fracture pathologies, announces the approvals of lumbar and cervical cages SteriSpineTM LC and CC in Japan. They were received by KiSCO Co., Ltd. (KiSCO) in October 2019, and mark the […]
2019
Alphatec Enhances its Lateral Portfolio With the Commercial Launch of Approach-Specific Innovation
CARLSBAD, Calif., Oct. 22, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial release of its porous titanium line of IdentiTi™ implants for lateral interbody fusion (LIF) procedures. The launch of the IdentiTi LIF Interbody […]
Implanet Granted Patent for JAZZ Lock® Implant in Japan
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729,PEA-PME savings plans eligible; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, announces that its JAZZ Lock® technology, which is an integral part of its JAZZ™ solutions platform, has been granted a patent in Japan. Implanet CEO Ludovic Lastennet said: “Protecting our […]
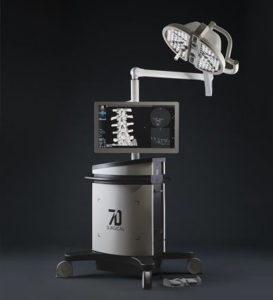
7D Surgical Completes 30th Installation Marking Key Sales Achievement
SAN FRANCISCO, Oct. 21, 2019 /PRNewswire/ — 7D Surgical announced today at the Congress of Neurological Surgeons Annual Meeting that it has completed 30 sales since commercial launch of its innovative Machine-vision Image Guided Surgery (MvIGS) platform for spinal and cranial surgery. This achievement represents installations throughout the United States, Canada, Australia and New Zealand. “These sales results validate the years of […]
There are more than 25 Sacroiliac Joint Fusion Devices… WHY? The Global market will reach 450 million US$ by the end of 2025
Sacroiliac joint fusion is a surgical procedure which fuses the iliac bone (pelvis) to the spine (sacrum) for stabilization. It is performed for a variety of conditions including trauma, infection, cancer, and spinal instability. Sacroiliac joint fusion may be performed as a minimally invasive surgical (MIS) procedure or as an open surgical procedure requiring a […]
Xtant Medical to Feature Innovative Product Offering at the CNS Annual Meeting
BELGRADE, Mont., Oct. 18, 2019 (GLOBE NEWSWIRE) — Xtant Medical Holdings, Inc. (NYSE American: XTNT), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, will feature its recently announced Intice™-C Titanium Cervical Interbody Spacer, Atrix-C Union™ Cervical Interbody Spacer and Calix®-C PC Plasma Coated PEEK Implant products at the […]
Synaptive Medical Expands Availability of Modus V™ into Spine Market
TORONTO–(BUSINESS WIRE)–Synaptive Medical, a leader in automation and robotics, announced today a strategic commercial expansion into the spinal surgery market. As part of this expansion, Synaptive is also announcing a co-marketing collaboration with Stryker’s Advanced Guidance Technologies business for the company’s robotically controlled digital microscope, Modus V™. Synaptive’s initial product offering, the BrightMatter™ suite of […]
CarboFix Extends Its CarboClear® Carbon Fiber Spinal Product Line With Its FDA Cleared Vertebral Body Replacement (VBR) System
DOVER, Delaware, Oct. 17, 2019 /PRNewswire/ — CarboFix has announced today that the U.S. Food and Drug Administration (FDA) has cleared its CarboClear® Carbon Fiber Vertebral Body Replacement (VBR) System to replace a collapsed, damaged, or unstable vertebral body due to tumor or trauma. The CarboClear® VBR System joins the FDA cleared CarboClear® Pedicle Screw and CarboClear® Fenestrated Pedicle Screw Systems. […]
EOS imaging TO HIGHLIGHT EOS SPINE SOLUTIONS AT EUROSPINE 2019
PARIS–(BUSINESS WIRE)–Regulatory News:EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI – Eligible PEA – PME), a leader in 2D/3D orthopedic medical imaging and software solutions for 3D anatomical modeling and surgical planning, today announced it will present its spine solutions, including the EOS System and spineEOS online 3D planning software in conjunction with EOSlink™, at the […]
Safe Orthopaedics: Efficacy of the Next-generation SteriSpine PS Solution Demonstrated by Several Dozen Surgeries
ERAGNY-SUR-OISE, France–(BUSINESS WIRE)–Regulatory News: Safe Orthopaedics (Paris:ALSAF) (FR0012452746 – ALSAF), a company specialized in the design and marketing of always new and sterile single-use implants and instruments improving the minimally invasive treatment of spinal fracture conditions, announces today that the efficacy of the second generation of the SteriSpine PS pedicle screw system has been demonstrated […]
Ozop Surgical Corp. Announces an Update to the Exclusive License Agreement with Spinal Resources Inc. for Spinal Implants
WEST PALM BEACH, FL, Oct. 16, 2019 (GLOBE NEWSWIRE) — via NEWMEDIAWIRE — Ozop Surgical Corp. (OTCQB: OZSC), a provider of premium surgical devices in the rapidly growing field of minimally invasive spine surgery (“Ozop” or the “Company”), today released the following corporate update on its recent progress and current developments. Ozop believes the license […]
SpineGuard Reports Third-quarter 2019 Revenue
SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that designs, develops, and markets disposable medical devices intended to make spine surgery safer by bringing real-time digital technology into the operating room, announced today that its third-quarter revenue was €1.7 million. Stéphane Bette, CEO and co-founder of SpineGuard, said: « These results are in line with our […]
Centinel Spine Named to The Silicon Review’s Top 50 Innovative Companies to Watch in 2019
NEW YORK, Oct. 15, 2019 /PRNewswire/ — Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced that it has been recognized as one of the Top 50 Innovative Companies to Watch in 2019 by The Silicon Review magazine. Centinel Spine began operations in 2008 through the merger-acquisition of two […]
Spineway : Approval of Mont-Blanc MIS in Japan
Spineway, specialist in surgical implants and instruments for treating disorders of the spinal column (spine), has just obtained approval for its Mont-Blanc MIS line from the Japanese authorities, authorizing it to sell these products for minimally invasive surgeries on this territory. Spineway is now in discussions with several Japanese companies specialized in importing and distributing […]
Eurospine 2019:The List of All the Sponsors&Exhibitors
Which Are the companies that you will find in Eurospine 2019? Please find enclosed the complete list: In’Tech Medical: Booth No.: 5G Paonan Biotech Co., Ltd.: Booth No.: 34B Global Biomedica s.r.o.: Booth No.:1B SpineVision SpineGuard: Booth No.: 21A 3D Systems AxioMed B. Braun Aesculap BAIDE MEDICAL BAUI SPINE BEIJING FITO MEDICAL CO., LTD. Beijing […]
Amplify Surgical Inc. Announces 100th Level Treated with dualX Expanding Interbody Fusion System
Amplify™ Surgical, Inc., is pleased to announce that only months since the initial alpha launch the dualX™ Expanding Interbody Fusion System has been implanted in 100 surgical levels. The milestone two level case was performed by Dr. Yong Kim at NYU Langone Health in New York, NY. After the case Dr. Kim stated, “dualX is […]
Medicrea Reports Third Quarter 2019 Sales
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris – FR0004178572 – ALMED; OTCQX Best Market – MRNTF), pioneering the digital transformation of spinal surgery through artificial Intelligence, predictive modeling and patient specific implants with its UNiD ASI™ (Adaptive Spine Intelligence) proprietary software platform, services and technologies, publishes sales for the third quarter […]
Safe Orthopaedics announces record revenues for the third quarter of 2019: €1,159k, an increase of 41%
ÉRAGNY-SUR-OISE, France–Safe Orthopaedics (Paris:SAFOR) (FR0012452746 – SAFOR), a company specializing in the design and marketing of single-use implants and instruments for the minimally invasive treatment of spinal fracture conditions, is today announcing its revenues for the third quarter of 2019 and its cash position at September 30, 2019. thousands euros Q3 2019(3 months) Q3 2018(3 […]
ChoiceSpine™ Announces Changes in Key Growth Leadership Positions
KNOXVILLE, TENN. (PRWEB) OCTOBER 08, 2019–ChoiceSpine LLC, a privately-held spinal device manufacturer based in Knoxville, TN, today announced several executive leadership changes designed to grow the company’s expertise, deepen its knowledge base, and drive sales growth. The following changes and new additions have been made in the sales and marketing teams. KC Gilbert promoted to […]
NuVasive to Participate as Gold Sponsor at the EUROSPINE 2019 Annual Meeting
SAN DIEGO, Oct. 9, 2019 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced it will participate as a gold sponsor at the EUROSPINE 2019 Annual Meeting held October 16-18, 2019 in Helsinki, Finland. “NuVasive is driven to deliver […]
Implanet Reports Q3 2019 Revenue of €1.7 Million, Up 12%
IMPLANET (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, today announces its revenue for the third quarter and first nine months to September 30, 2019. Ludovic Lastennet, CEO of Implanet, commented: “In line with our objectives, the third quarter continues the […]
BIOMECH Paonan to Showcase New Spinal Solutions at EuroSpine Congress 2019
Biomech (Paonan Biotech) will join EUROSPINE Annual Meeting in Helsinki next week. Biomech will provide the brand new spinal solution for our valued guests and partners. Eurospine 2019 Biomech Booth No.: 34B Date: Oct 16-18, 2019 Venue: Messukeskus Helsinki, Finland About BIOMECH Paonan The world 1st case of spinal implant was carried out in 1984, […]
Intralink-Spine, Inc. Announces ISO-13485 Certification
LEXINGTON, Ky.–(BUSINESS WIRE)–Intralink-Spine, Inc. (ILS) announces that it has received ISO 13485:2016 certification for its Quality Management System (QMS) following review by its notified body, MedCert GmbH. ISO 13485:2016 is an internationally recognized quality standard specific to the medical device industry and signifies Intralink-Spine’s commitment to the highest level of medical device quality controls and […]
Viseon Inc. Announces the First Worldwide Use of Its High Definition Visualization Device in a Multi-Level Lateral Lumbar Deformity Procedure
IRVINE, Calif.–(BUSINESS WIRE)–Viseon, Inc. announces the first clinical use of the Voyant Lateral System, a visualization technology to enable real-time imaging during minimally invasive lateral lumbar spine surgery. The system provides the surgeon with intraoperative visualization allowing an accurate, precise and unobstructed view of the operative field offering optimized procedural workflow. The device is the […]
Xtant Medical Names Sean Browne President and Chief Executive Officer
BELGRADE, Mont., Oct. 07, 2019 (GLOBE NEWSWIRE) — Xtant Medical Holdings, Inc. (NYSE American: XTNT), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, today announced the appointment of Sean Browne as President and Chief Executive Officer, effective immediately. “Sean has an extensive track record of success as a […]
Is the Artificial Cervical Disc Market still Alive?Did you know that Today there are more than 40 Competitors?
Artificial Disc Market to exceed USD 4.5 billion by 2024 as per a new research report.Steadily growing number of osteoarthritis and rheumatoid arthritis procedures around the world is a primary factor responsible for artificial disc market growth. Increasing incidences of degenerative disc diseases and adult patient’s willingness to opt for artificial disc are predominant factors […]
Centinel Spine® Celebrates 18 Years of Usage of prodisc® L Total Disc Replacement in the U.S.
NEW YORK, Oct. 3, 2019 /PRNewswire/ –Centinel Spine, LLC, the largest privately-held spine company focused on anterior column reconstruction, today celebrated the 18th year of clinical usage of the prodisc® L Lumbar Total Disc Replacement in the United States. prodisc L was first implanted in the United States during concurrent IDE studies to obtain FDA approval for one- and two-level use. The first […]