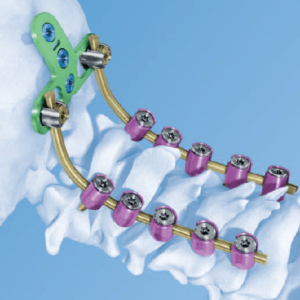
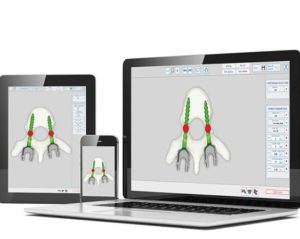
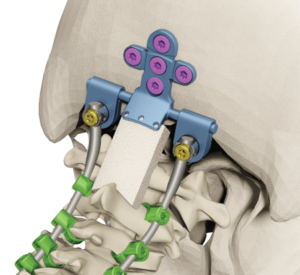
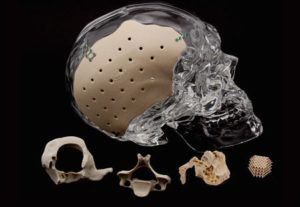
LEESBURG, Va., May 30, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced U.S. Food and Drug Administration (FDA) 510(k) clearance for BACS® Patient-Specific devices. With the BACS Surgical Planner, surgeons can […]
2018
Titan Spine ‘turns it to eleven’
(BIZTIMES.COM)–With a proprietary surface technology and a runway of private equity investment, Titan Spine LLC has, to borrow a phrase from the cult classic film “This is Spinal Tap,” turned its growth up to 11.The Mequon-based medical device developer was established after surgeon Dr. Peter Ullrich came up with the idea to make interbody spinal […]
Posterior Cervical Fusion Market (Part2) 10 More Systems to Know!
The posterior cervical fusion is a type of cervical fusion surgery, that accounts the 15% to the cervical devices market share. Although it is still an small segment of the overall Spinal market, it is growing due to the increasing prevalence of osteoarthritis and rheumatoid and the rising aging population. The estimated market value of the posterior […]
In’Tech Medical enters into agreement to acquire Bradshaw Medical, industry leader in silicone overmolded solutions, ratchet and torque devices
Memphis, Tenn. May 23, 2018–In’Tech Medical SAS (http://www.intech-medical.com), the leading Contract Manufacturer of medical devices in Orthopedics, announced today that it has entered into a binding agreement to acquire world renowned instruments and silicone overmold company, Bradshaw Medical Inc. The transaction, supported by Eurazeo PME, has been approved by the Board of Directors of both organizations. Based in […]
Spinal Elements® Announces Expanded Commercial Release of its Clutch® Interspinous Process Device
CARLSBAD, Calif.–(BUSINESS WIRE)–Spinal Elements, a spine technology company, today announced the release of its Clutch interspinous process device. This new product further enhances the breadth of Spinal Elements’ thoracolumbar portfolio and offers surgeons more options for treatment of various posterior thoracolumbar pathologies. Clutch has been in use since shortly after its FDA 510(k) clearance in […]
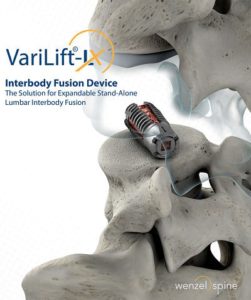
Wenzel Spine Announces Publication of Case Series Detailing Endoscopic Transforaminal Decompression and Fusion Technique with VariLift-LX
AUSTIN, Texas–(BUSINESS WIRE)–Wenzel Spine, Inc., a medical technology company focused on providing minimally invasive solutions for the treatment of spinal disorders, today announced a 24 Consecutive Patient Case Series detailing an endoscopic, stand-alone TLIF surgical technique used with the VariLift-LX System has been published in the Journal of Spine, Volume 7, Issue 2. The Case […]
Medacta International Introduces New Approach to Posterior Lumbar Midline Cortical Screw Placement with Customizable MySpine MC Surgical Guides
CASTEL SAN PIETRO, Switzerland–(BUSINESS WIRE)–Medacta International, the family-owned global leader in innovative joint replacement and spinal technologies, announced clearance from the U.S. Food and Drug Administration (FDA) and the addition of MySpine Midline Cortical Pedicle Screw Placement Guide (MySpine MC) to its MySpine portfolio. The first two stateside surgeries utilizing MySpine MC were conducted this […]
Nuvasive SpineTRACK Registry Reaches Significant Enrollment Milestone Of 10,000 Patients
NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that the Company’s SpineTRACK Registry has reached a significant enrollment milestone of 10,000 patients from more than 50 surgeons in the United States since its inception in 2011. This milestone illustrates not only the continued […]
IMPLANET Announces the Launch of JAZZ Evo® for the Promising Market of Degenerative Spine Disease in Adults
BORDEAUX, France & BOSTON–IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, announces the launch of JAZZ Evo, a new evolution of the JAZZ® implant developed to respond to the constraints of vertebral fusion indications in adults. Developed primarily […]
Zimmer Biomet Announces 510(k) Clearance for Zyston® Strut Open Titanium Interbody Spacer System
WARSAW, Ind., May 22, 2018 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Zyston® Strut Open Titanium Interbody Spacer System. This marks Zimmer Biomet’s first titanium spinal implant manufactured via […]
Life Spine Announces Line Extension of PRO-LINK® Ti Stand-Alone Cervical Spacer System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that the U.S. Food and Drug Administration (FDA) has given 510(k) marketing clearance to the PRO-LINK Ti Stand-Alone Cervical Spacer System for spinal fusions. PRO-LINK featuring Life Spines OSSEO-LOC™ surface treatment […]
NuVasive PRECICE STRYDE™ System Used In First Patient For Stature Lengthening By International Limb Lengthening Expert
SAN DIEGO, May 22, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the new PRECICE STRYDE™ system from NuVasive Specialized Orthopedics™ (NSO) was used in the first set of patients by internationally recognized limb lengthening and reconstruction expert Dr. Dror Paley. Built on […]
Providence Medical Technology Announces FDA 510(k) Clearance for DTRAX® Spinal System
PLEASANTON, Calif., May 22, 2018 /PRNewswire/ — Providence Medical Technology, Inc., a manufacturer of innovative instruments and implants for cervical fusion surgery, today announced it has received 510(k) clearance from the United States Food and Drug Administration (FDA) for its DTRAX® Spinal System (“DTRAX”) to be specifically indicated for use in posterior cervical fusion in […]
Xtant Medical Receives FDA 510(K) Clearance for InTice™-C Porous Titanium Cervical Interbody System
BELGRADE, MT / ACCESSWIRE / May 21, 2018 / Xtant Medical Holdings, Inc. (NYSE American: XTNT), a leader in the development of regenerative medicine products and medical devices, announces U.S. Food and Drug Administration (FDA) 510(k) clearance for InTice™-C Porous Titanium Cervical Interbody System. InTice-C is designed using OsteoSync Ti, a best-in-class, cost-effective, highly porous titanium […]
Globus Medical Announces Appointment of Orthopedic Trauma Division’s Vice President of Sales
AUDUBON, Pa., May 21, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the appointment of Daniel Gregoris to Vice President of Sales for the company’s emerging Orthopedic Trauma division. Gregoris will be responsible for leading the growth and expansion of Globus Medical’s direct Trauma sales force and the […]
Cerapedics Announces Presentation of Results from the IVANOS Study of i-FACTOR™ Peptide Enhanced Bone Graft in Uninstrumented Lumbar Posterolateral Spine Surgery
WESTMINSTER, Colo., May 21, 2018 /PRNewswire/ — Cerapedics, a privately-held orthobiologics company, today announced results from the IVANOS study evaluating i-FACTOR™ Peptide Enhanced Bone Graft in lumbar posterolateral spine surgery. Results were presented by Mikkel O Andersen, MD, from the Sector for Spine Surgery and Research at Middelfart Hospital in Denmark, at the 7th Annual […]
Is the Posterior Cervical Fusion Market Attractive? 7 Systems to Know
Posterior Cervical fusion technique involves the fusion of more than two vertebrae to prevent unwanted motion of the discs or the compression of posterior spaces between the cervical vertebrae. Posterior Cervical Fusion is a procedure intended for the stabilization of the cervical spine through a posterior approach. The procedure is commonly performed using hooks, plates, […]
NuVasive Sponsors SOLAS 2018, Hosts Workshop On Latest Lateral Innovations
SAN DIEGO, May 16, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that the Company is a sponsor of the Society of Lateral Access Surgery (SOLAS) and its annual meeting being held May 17-19, 2018, in San Diego. NuVasive was recently recognized by SOLAS […]
K2M Announces US Launch of First-to-Market 3D-Printed Expandable Interbody MOJAVE™ PL 3D Expandable Featuring Lamellar 3D Titanium Technology™
LEESBURG, Va., May 16, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced the U.S. commercial launch of its MOJAVE™ PL 3D Expandable Interbody System. Designed with K2M’s Lamellar 3D Titanium Technology™, […]
LinkSPINE Names David Capallo Vice President of Sales
Columbia, MD — May 8, 2018 — LinkSPINE, a medical device company focused on the development of less invasive techniques for spine surgery, has named David Capallo Vice President of Sales. Capallo joins LinkSPINE from NuVasive, where he worked since 2006 and most recently served as Director, Commercial Strategy. Prior to this, he held positions […]
Globus Medical Announces Positive Clinical Results for Lateral Expandable Technology
AUDUBON, Pa., May 15, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the results of the study “Clinical and radiographic analysis of expandable versus static lateral lumbar interbody fusion devices with two-year follow-up” published in the Journal of Spine Surgery, supporting benefits of Globus’ expandable lateral spacers. The […]
Spine Industry: Growth Through Acquisitions! 7 Relevant Acquisitions in the last 12 months
During the last year, the spinal market has seen more than 7 acquisitions for many different reasons. Buy Sales or Market Share: Although the global spinal implants market is still growing at an expected rate of 6.6% CAGR 2022, acquisitions are an importante alternative way for Spinal Companies to achieve more than single digit organic spine […]
Oxford Performance Materials and JSR Corporation Announce Partnership to Advance OPM Technology Platform Throughout Asia
SOUTH WINDSOR, Conn. and TOKYO, May 14, 2018 (GLOBE NEWSWIRE) — Oxford Performance Materials, Inc. (OPM), a leader in advanced materials science and high performance additive manufacturing (HPAM®), and Tokyo-based JSR Corporation (JSR), a leading materials supplier in a variety of technology driven markets, today announced a partnership to advance OPM’s poly-ether-ketone-ketone (PEKK)-based 3D-printed orthopedic […]
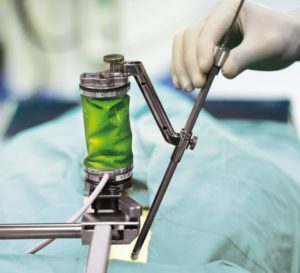
Mazor Robotics Reports Record First Quarter Results
CAESAREA, Israel, May 14, 2018 /PRNewswire/ — Mazor Robotics Ltd. (TASE: MZOR) (NASDAQ-GM: MZOR), a pioneer and a leader in the field of robotic guidance systems, reported record first quarter revenue of $15.5 million compared to $11.7 million in the first quarter of 2017. “Our record first quarter revenue reflects clinical adoption, as we surpassed […]
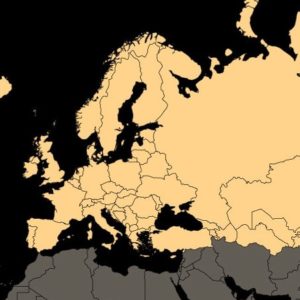
Europe spinal implants market: (Part IV) The Top 20 European Manufacturers Of Spinal Implants
Spine surgery is a relatively mature market — all segments of the spine can be treated, significant share has been garnered by a few major companies, and there are no radically new technologies disrupting current market positions. Yet, trends in the patient population, the active innovation by all companies (see further, below, on market shares […]
3D-Printing in Spine: 17 Additively Manufactured Lumbar Cages to Know
In the past decade, the orthopedic implant industry has been quietly revolutionized through the use of additive manufacturing. Today, the penetration rate of additive production of standard-sized implants is expanding rapidly. In the future the researcher believes the majority of implants will be produced additively, creating a new revenue stream for printers and materials as […]
Amedica Corporation Announces Pricing of $15,000,000 Public Offering of Units Consisting of Convertible Preferred Stock and Warrants
SALT LAKE CITY, May 2018 (GLOBE NEWSWIRE) — Amedica Corporation (NASDAQ: AMDA) an innovative biomaterial company that develops and commercializes silicon nitride for biomedical applications, today announced the pricing of an underwritten public offering of units, consisting of convertible preferred stock and warrants, for gross proceeds of $15,000,000, which excludes underwriting discounts and commissions and […]