SAN JOSE, Calif., July 18, 2016 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the use of the iFuse Implant System® (“iFuse”), a triangular-shaped minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced the publication of the 40th peer-reviewed paper on SI joint fusion with […]
2016
Medtronic Launches Revamped Minimally Invasive Spine Fusion Procedures
DUBLIN – July 15, 2016 – Medtronic plc (NYSE: MDT) today announced the continued development of the OLIF Platform with the launch of the PivoxTM Oblique Lateral Spinal System with Lateral Plate for OLIF25TM and Divergence-LTM Anterior/Oblique Lumbar Fusion System for OLIF51TM at the International Meeting on Advanced Spine Techniques (IMAST) in Washington, D.C. Lumbar […]
Medtronic invests another $20 million in Mazor Robotics
(Star Tribune)–Medtronic PLC is preparing to invest another $20 million in Israeli med-tech company Mazor Robotics, which is expected on Tuesday to announce a new robotically assisted spine-surgery system called Mazor X. Analysts say physician interest in robotically assisted surgical tools is steadily growing, and Medtronic seems to be positioning itself to capitalize on the […]
ulrich medical USA® Announces Market Release of neon3 Posterior Cervical System
ST. LOUIS, July 13, 2016 /PRNewswire/ — ulrich medical USA, Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States, announced today the market release of the neon3 TM Posterior Cervical System, which is intended to provide immobilization and stabilization of spinal segments as an adjunct to fusion […]
Titan Spine Raises Capital for Continued U.S. Growth and Adoption of its Interbody Fusion Devices Featuring nanoLOCK™ Technology
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative surface-enhanced spinal interbody fusion implants, today announced that it has closed a substantial round of financing with investor Southlake Equity Group. Titan will use the proceeds to further expand U.S. sales growth and surgeon adoption of its Endoskeleton® line of titanium […]
SpineGuard reports 29% growth and €1.9M revenue for the second quarter 2016
PARIS and SAN FRANCISCO, July 12, 2016 – SpineGuard (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices that empower surgeons to enhance clinical outcomes and simplify surgeries, reported today that its second quarter 2016 revenue grew by 29%, to €1.9 million. Pierre Jérôme, Co-founder and CEO of SpineGuard, said: “Our […]
Are you looking for a Different Cage? Learn about 19 PLIF Expandable Cages
PLIF Expandable interbody cages is a new product category that provides the following key benefits: Most of them, can be implanted with a minimally invasive surgical procedure allowing surgeons to go through a small corridor and change the geometry of the cage to correspond to what the patient’s spine requires. These implants may help protect a patient’s nerve roots […]
Medicrea H1 Revenue Rises 7% to 14.8 Million Euros
The Medicrea Group (Alternext Paris: FR0004178572 – ALMED), PEA- PME eligible, pioneering the development and manufacture of patient-specific analytical services and implant solutions for the treatment of spinal conditions, announces sales for the first half of 2016. Medicrea First Half Sales: H1 2015 Sales = €13.8 million H1 2016 Sales = €14.8 million Increase = […]
Spine Wave Announces the Commercial Launch of the True Position® Pivoting Spacer System
SHELTON, CT–(Marketwired – July 11, 2016) – Spine Wave, Inc. is committed to the commercialization of high-quality, innovative medical devices for the treatment of spinal disorders. Spine Wave’s commitment was demonstrated by its previously announced acquisition of rights in the True Position® Pivoting Spacer System from Atlas Spine on February 23, 2016. Since then, Spine […]
Titan Spine Secures CMS New Technology ICD-10 Code for Nanotextured Surface on Interbody Fusion Devices
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative surface-enhanced spinal interbody fusion implants, reports that the U.S. Centers for Medicare & Medicaid Services (CMS) has announced the creation of a new technology ICD-10 code for a nanotextured surface on an interbody fusion device. Titan Spine’s nanoLOCK™ surface technology, which […]
Amedica Announces Closing of $12.7 Million Public Offering and Full Exercise of Over-Allotment Option
SALT LAKE CITY, UT–(Marketwired – Jul 8, 2016) – Amedica Corporation (NASDAQ: AMDA), a company that develops and commercializes silicon nitride ceramics, today announced it has closed the underwritten public offering of $12.7 million at a price to the public of $1.00 per unit of one share and one warrant, which included the exercise in […]
Precision Spine® Names Christopher A. DeNicola Chief Operating Officer as Company Enters Next Phase of Aggressive Growth
PARSIPPANY, N.J.–(BUSINESS WIRE)–Precision Spine, Inc., a medical device company dedicated to Made-in-the-USA manufacturing, announced that, in support of the company’s accelerating growth plan, it has named Christopher A. DeNicola its new Chief Operating Officer. “Chris is a seasoned leader in the orthopedic medical device industry and he has been instrumental in building our domestic and […]
Merit Medical Acquires DFINE, Inc.
SOUTH JORDAN, Utah, July 06, 2016 (GLOBE NEWSWIRE) — Merit Medical Systems, Inc. (NASDAQ:MMSI), a leading manufacturer and marketer of proprietary disposable devices used primarily in cardiology, radiology and endoscopy, today announced that it has acquired DFINE, Inc. headquartered in San Jose, California, in a merger transaction through which DFINE has become a wholly-owned subsidiary […]
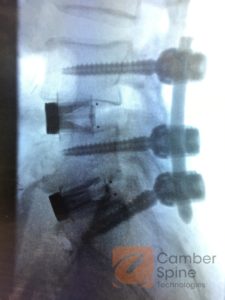
Camber Spine Technologies Announces First implantation of ENZA™ Zero-Profile ALIF Device
WAYNE, Pa., July 5, 2016, – Camber Spine Technologies, today announced the first implantation of the ENZA™ MIS Zero-Profile anterior lumbar interbody fusion (ALIF) device in a two-level lumbar procedure at levels L4/L5 and L5/S1 at Kennedy University, Washington Township, by Dr. Jeffrey Gleimer, D.O. ENZA™ is the company’s first device launched with integrated fixation […]
Amedica Announces Pricing of $11 Million Public Offering
SALT LAKE CITY, UT–(Marketwired – Jul 5, 2016) – Amedica Corporation (NASDAQ: AMDA), a company that develops and commercializes silicon nitride ceramics, today announced the pricing of an underwritten public offering of units for gross proceeds of $11 million, prior to deducting underwriting discounts and commissions and offering expenses payable by Amedica. The offering is comprised […]
SpineCraft Obtains Regulatory Clearance to market the APEX Spine System® in China, the second-largest spine market in the world.
SpineCraft announced today that it has received clearance from the China Food and Drug Administration (CFDA) to market its APEX Spine System® in China. The APEX Spine System is designed for complex deformity correction as well as tumor, trauma and degenerative procedures and has proven clinical track record with over 15,000 surgeries performed since its […]
7th Anniversary | SPINEMarketGroup
Launched in July 2009, we’re now celebrating our 7th Anniversary! Thank you very much to all our visitors and followers! In all these years, we have published more than 2.500 news and provided information related to the Spinal market including Spinal Companies, Interspinous Devices, MIS devices, LLIF, TLIF cages, Artificial Cervical Discs, Dynamic implants , SI Joint Fusion Devices, […]
Updated Spine Catalogue 2016 with 7 more Spinal Companies (347)
Today we have updated our Spine Catalogue with 7 more Companies. Now, we have 347 manufacturers in our list. Also, we have included the one more system in the list of 14 SI fusion devices. 1.- List of the 7 companies Included: Alethea-spine evospine Southern-medical CTL Medical-corporation X-spine Flospine Cardinal-spine 2.- New SI Fusion Device Included: Prolix […]
NuVasive Agrees to Settle Patent Litigation With Medtronic
SAN DIEGO, CA–(Marketwired – June 30, 2016) – NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery, today announced that the Company has agreed to settle its ongoing patent litigation with Medtronic and certain of its subsidiaries. NuVasive and Medtronic reached agreement on terms for the settlement of the previously […]
Amendia Launches an Innovative Anterior Cervical Plate, the Ceres™ Midline
Amendia (http://www.amendia.com), a leading provider of innovative spinal technologies, is pleased to announce the commercial launch of its Ceres™- Midline Cervical Plate. The Ceres Midline Cervical Plate System is intended for anterior screw fixation of the cervical spine (C2-C7) as an adjunct to fusion. It is indicated for degenerative disc disease, spondylolisthesis, trauma, spinal stenosis, […]
Pierre Dumouchel’s interim appointment as Chief Executive Officer made permanent
SAFE ORTHOPAEDICS (Paris:SAFOR) (FR0012452746 – SAFOR), a company offering innovative ranges of sterile implants combined with their single-use instruments, is today announcing that Pierre Dumouchel’s interim appointment as Chief Executive Officer has been made permanent and that Alain Coudray has been named Chairman of the Board of Directors. Following his accomplishments since he was first […]
Amendia Promotes Chris Fair to President & CEO
Amendia, Inc., a leading designer, developer, manufacturer and marketer of medical devices used in spinal surgical procedures, today announced that Chris Fair, its current Chief Operating Officer, has been named President and Chief Executive Officer. Additionally, Amendia announced the appointment of Scott Bruder, MD, PhD, a current Amendia Director, to be the Chairman of its […]
Phygen, LLC Receives Notice of Issuance of Its ISP Device Under U.S. Patent Number 9,387,016
IRVINE, Calif., June 29, 2016 /PRNewswire/ — Phygen, LLC, a company focused on the development of devices used in the stabilization and repair of spinal disease and trauma, today announced that it has received a Notice of Issuance for its latest interspinous device as an additional device for use in the treatment of spinal disease […]
500th Spinal Fusion Performed with Benvenue Medical’s Luna® 3D Interbody Fusion System
SANTA CLARA, Calif.–(BUSINESS WIRE)–Benvenue Medical, Inc., a developer of minimally invasive solutions for spine repair, today announced that more than 500 patients have been treated with the company’s Luna® 3D Interbody Fusion System. The 500th case was performed by Richard Fessler, M.D., Ph.D., of Rush University Medical Center in Chicago. “The Luna System is an […]
Orthobion Interbody Devices with the unique FGOIC Ti technology
Orthobion has developed a complete range of cervical and lumbar interbody devices with the unique FGOIC Ti technology (Fine Grained Osseo Integrative Coating of Titanium). FGOIC Ti is a unique surface technology that forms the perfect blend of PEEK and Titanium.. Orthobion combines the best of both worlds. They have a unique microstructured topography on the […]
Trial begins in lawsuit over spinal bone cement
SEATTLE (AP) — The physician who used a non-FDA-approved bone cement during a woman’s spinal surgery and the company that ran an illegal test market to promote the dangerous product should be held accountable for her death, a lawyer for the woman’s daughter told a jury Monday. Reba Golden was vibrant and healthy when she […]
Do you like PEEK Cages? Are you satisfied? Learn about 9 Titanium Coated TLIF implants!
Learn about 9 Titanium Coated Peek TLIF Cages: https://thespinemarketgroup.com/tlif-coated/ Surgeons have historically preferred interbody spacers made of titanium because of their strength and long clinical history.Over the last 10 years PEEK has largely replaced titanium because it has a modulus of elasticity similar to human cortical bone and for its radiolucency. But recently, it has been discussed the difficulties […]