IRVINE, Calif., April 5, 2016 /PRNewswire/ — Interventional Spine, Inc. today announced the launch of a new line of products that will be showcased at this year’s International Society for the Advancement of Spine Surgery (ISASS) Annual Meeting in Las Vegas, NV. The new products, available at exhibit booth #229, include recent size additions to […]
2016
Premia Spine Launches ProMIS™ Fixation System
PHILADELPHIA, April 5, 2016 /PRNewswire/ — Premia Spine, a spinal implant company focused on commercializing innovative lumbar solutions, today announced the launch of the ProMIS™ Fixation System with its Advanced MIS screw placement at the International Society for the Advancement of Spine Surgery Meeting in Las Vegas. Dr. Steven DeLuca, from the Orthopedic Institute of […]
Green light for a new implant: JAZZ Lock
Bordeaux, Boston, 5 April 2016 – IMPLANET (Euronext: IMPL, FR0010458729, PEA-PME eligible), a medical technology company specializing in vertebral and knee-surgery implants, today announces that it has been granted 510k clearance in the United States and the CE mark in Europe to market its new Jazz Lock® implant. Jazz Lock® is the first of an […]
NuVasive Launches Integrated Global Alignment (iGA) Platform at BritSpine 2016
LONDON, UNITED KINGDOM–(Marketwired – April 04, 2016) – NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that it will participate in BritSpine 2016 held April 6-8, 2016 at Nottingham Conference Centre, in Nottingham, England. NuVasive’s participation at BritSpine will include numerous education workshops […]
German Patient First To Receive 3D Printed Titanium Spinal Fusion Implant
Spinal fusion surgeries in the United States have increased by 70 percent between 2001 and 2011. While those numbers are due in large part to the aging of the American population, they’re also due to advances in technology like additive manufacturing. Now for what Dr. Uwe Spetzger, a Professor and Chairman of the Department of […]
Spine Rep of Tomorrow: Adaptation is Key
April 3, 2016– “…but the species that survives is the one that is able best to ADAPT and adjust to the changing environment in which it finds itself” said Darwin. Spine Market: Changing Environment In the recent years, the Spinal Industry has started a transformation .Governments and health insurers worldwide are implementing measures to control costs, public hospitals are operating […]
Top Emerging Trends Impacting the Spine Market
The top four emerging trends influencing the global spine surgery market according to Technavio’s healthcare and life sciences research analysts are: Increased use of surface-modified titanium in spinal implants The use of titanium to manufacture spinal implants has increased in recent years. Spinal implants such as cages, rods, screws, hooks, wires, plates, and bolts are […]
Preparing For The New EU Medical Device Regulations
By Ronald Boumans and Stewart Eisenhart, Emergo–When European lawmakers and regulators first indicated plans to overhaul legislation on how the union oversees medical devices and in vitro diagnostics (IVDs) in 2010 and 2011, industry participants knew such changes to the world’s second-largest device market would be anything but sudden. Indeed, the wait for final legislation, […]
Implanet announces 2015 annual results
Bordeaux, Boston, March 30, 2016 – IMPLANET (Euronext: IMPL, FR0010458729, PEA-PME eligible), a medical technology company specializing in vertebral and knee-surgery implants, today announces its annual results for the financial year ending December 31, 2015, as approved by the Board on March 24, 2016. Ludovic Lastennet, CEO of Implanet, says: “We are continuing the sales […]
FDA Clears Xspan Laminoplasty Fixation System
Xtant Medical Holdings, Inc. (NYSE: XTNT) announced that its wholly owned subsidiary, X-spine Systems, received FDA clearance of the Xspan™ Laminoplasty Fixation System. The Xspan™ System represents the latest generation of laminoplasty fixation. Spinal laminoplasty is a surgical procedure performed to remove pressure from the spinal cord. Xtant Medical estimates the worldwide market for laminoplasty […]
DIPLOMAT screw system is double award winner
Alzenau, March 30, 2016. The DIPLOMAT pedicle screw system has been awarded the globally acclaimed „Red Dot“ seal of quality. The implant was also awarded the senetics Innovation Award on 1 March 2016. DIPLOMAT is a modular system with in-situ interchangeable tulips and self-tapping thread, which is suitable for all indications and makes implantation more efficient […]
NuVasive – Same Opportunities, Same Concerns
(Seekingalpha.com)–Not a lot has really changed for NuVasive (NASDAQ:NUVA). The third-largest spine company in the U.S. by market share, NuVasive continues to take share from larger players on the back of innovative products that offer meaningful advantages in terms of patient outcomes and surgeon convenience. On the other hand, concerns remain about the company’s weaker […]
LDR Shows Superiority of Two-Level Mobi-C Cervical Disc to ACDF
LDR Holding Corporation (Nasdaq:LDRH), a global medical device company focused on designing and commercializing novel and proprietary surgical technologies for the treatment of patients suffering from spine disorders, today announced that “Five-year clinical results of cervical total disc replacement compared with anterior discectomy and fusion for treatment of two-level symptomatic degenerative disc disease: a prospective, […]
SpinalCyte New Results from Phase II Animal Trials of Cell Therapy to Regenerate the Spinal Disc
HOUSTON–(BUSINESS WIRE)–SpinalCyte, LLC, a Texas-based tissue engineering technology company focused on regrowth of the spinal disc nucleus using Human Dermal Fibroblasts (HDFs), today announced the preliminary results of Phase II Animal Studies for Spinal Disc Height Improvement. The study began in 2014 with Rush University and Howard An, M.D., The Morton International Endowed Chair Professor […]
Spinal Non-Fusion Market to Almost $750 Million by 2021
The global market for spinal non-fusion treatments will expand from just under $521 million in 2014 to $749 million by 2021, at a Compound Annual Growth Rate (CAGR) of 5.3%, according to research and consulting firm GlobalData. The company’s latest report* states that this moderate growth will be driven primarily by the promising Cervical Total […]
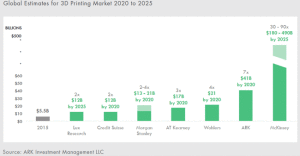
3-D Printing Market Expected to Grow 40% Annually
3-D Printing technology will make good on its promise to enable the Third Industrial Revolution, says the investment firm Ark Invest (New York City). Sure, 3-D printing has been hyped. But in many ways, the technology is undervalued, maintains ARK Invest, which anticipates that the annual growth rate for the technology will be approximately 40% […]
Xtant Medical Announces Distribution Agreement with Vivex Biomedical
BELGRADE, Mont., March 24, 2016 (GLOBE NEWSWIRE) — Xtant Medical Holdings, Inc. (NYSE:XTNT), a leader in the development of regenerative medicine products and medical devices, is expanding its biologics portfolio through a distribution relationship with Vivex Biomedical, Inc., a leading processor of human tissue, synthetics, mimetic patterning technology, and biocellular products. Through this agreement, Xtant […]
SpineGuard Reports Full-Year 2015 Financial Results
PARIS & SAN FRANCISCO–SpineGuard (FR0011464452 – ALSGD), an innovative company that develops and markets disposable medical devices designed to make spine surgery safer, reported its full-year 2015 financial results as approved by the Board of Directors on March 22, 2016. Pierre Jérôme, CEO and co-founder of SpineGuard, said: “Whether you look at sales, financing or […]
VEXIM: 2015 Financial Results
VEXIM (Paris:ALVXM) (FR0011072602 ‐ ALVXM / PEA-PME), a medical device company specializing in the minimally invasive treatment of vertebral fractures, announced its consolidated financial results for the year that ended on December 31st, 2015. Fast and sustained business growth In 2015, VEXIM had a turnover of €13.9 million, up 34% compared to 2014. This increase […]
NJ Doctor Escapes Most Claims In Suit Over Spinal Implants
Law360, New York (March 2016, ) — A New Jersey doctor escaped most of the claims brought against him in federal court by a patient who accused him of medical malpractice after devices implanted in her spine failed, but he’s still facing negligence charges in the heavily trimmed case. U.S. District Judge Noel L. Hillman […]
Amedica First to 3D Print Silicon Nitride for Medical Applications
SALT LAKE CITY, UT–(Marketwired – Mar 23, 2016) – Amedica Corporation (NASDAQ: AMDA), a company that develops and commercializes silicon nitride ceramics as a biomaterial platform, is pleased to announce its first fabrication of complex, three-dimensional structures by a 3D printing process called robotic deposition, or robocasting. The final products have been examined under scanning […]
IMPLANTS 2016 Conference Paris – June 7th
“IMPLANTS” in France, is an international conference on innovations and solutions for orthopedic implants and orthobiologics. This conference is the most important European event dedicated to the orthopedic implants industry, along with its European and international manufacturers and subcontractors.Top level conference for contract manufacturers and the orthopedic companies that will focus on changes in manufacturing […]
Blue Cross Blue Shield of Michigan Announces Positive Coverage Policy for iFuse Implant System® MIS SI Joint Fusion
SAN JOSE, Calif., March 21, 2016 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the use of the iFuse Implant System® (“iFuse”), a minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced that Blue Cross Blue Shield (BCBS) of Michigan, the largest commercial insurer in […]
Orthofix Appoints Michael Paolucci to Board of Directors
Orthofix International N.V. (NASDAQ:OFIX), a diversified, global medical device company, announced that Michael E. Paolucci has been named to its Board of Directors and appointed to the Compensation Committee. A seasoned Human Resource (HR) executive, Paolucci has more than 20 years of global experience working directly with Boards of Directors and C-level executives to improve […]
Add our Apple Icon to your Desktop
If you visit our website frequently we recommend you to bookmark our site so that you can access to it without typing in the web address every time. However, you can quickly and easily access www.TheSPINEMarketGroup.com by adding an icon directly on the iPhone or iPad Home screen. How to do it? To add an Apple Icon to the desktop of an iPad or […]
Promimic and Danco in a Strategic Partnership
WARSAW, IN, USA AND GOTHENBURG, SWEDEN – MARCH 17, 2016. Promimic and Danco today signed a license agreement where Danco will invest in a production line for the HAnano Surface process developed by Promimic. Danco will be the preferred process partner for Promimic for the US and China medical implant market. Since 1971, Danco Anodizing has […]
Titan Spine Executive Appointed to Executive Operations Committee for FDA-Sponsored MDEpiNet
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced the appointment of Jeff Dunkel, Titan Spine Vice President, Strategic Partnering, to the Executive Operations Committee of the Medical Device Epidemiology Network Initiative (MDEpiNet). The initiative is a collaborative program of the FDA’s Center for […]