Medtronic PLC has hired Karen L. Parkhill as its new chief financial officer, recruiting her from the same role at Comerica Inc.Medtronic said Wednesday that Ms. Parkhill would fill the CFO post, after The Wall Street Journal reported Tuesday that Ms. Parkhill had been hired away. She will start in the role June 20. Comerica, […]
2016
Updated Interspinous Page with the 52 most Important Devices
SpineMarketGroup announces the launch of an Updated Interspinous Page with 52 devices, including the main players and products. We have included more information and features. Here, (https://thespinemarketgroup.com/interspinous), you will find the main Static, Dynamic, and Percutaneous Devices as well as the most important interspinous Fusion devices. Interest in interspinous devices has emerged again with a great focus […]
SpinalCyte, LLC Receives New United States Patent for Spinal Disc Tissue Engineering
HOUSTON, Texas – SpinalCyte, LLC, a Texas-based tissue engineering technology company focused on regrowth of the spinal disc nucleus using human dermal fibroblasts, announced today the issuance of U.S. Patent No. 9,320,776, “Methods And Compositions For Repair Of Cartilage Using An In Vivo Bioreactor.” The technology described in the patent involves a device which is […]
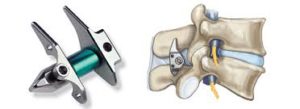
CoreLink Releases the Entasis SI Joint Fusion System
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a fast growing designer and manufacturer of spinal implant systems, is pleased to announce the U.S. launch of the Entasis™ Sacroiliac Joint Fusion System. Entasis was designed to better address the challenges of SI joint fusion procedures through innovation in the delivery of autograft and compression across the joint. Further innovations […]
Life Spine Announces Twenty New Innovations in 2016, Record Growth in First Quarter
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced that revenues for the first quarter of 2016 grew by 38% over the same period last year and that twenty new products are scheduled for release in 2016. These releases include best […]
MEDTECH Announces First ROSA Spine Surgery in the United States
MONTPELLIER, France, May 03, 2016 (GLOBE NEWSWIRE) — MEDTECH (Euronext, FR0010892950 – ROSA), a company specialized in designing, developing and marketing innovative surgical assistance robots, today announced the first surgical operation with the ROSA™ Spine in the U.S. was performed at Miami Valley Hospital, a nationally recognized medical facility in Dayton, Ohio. Miami Valley Hospital […]
Amedica Completes Debt Exchange and Reduces Debt Obligations by 47% Year-Over-Year
SALT LAKE CITY, UT–(Marketwired – May 3, 2016) – Amedica Corporation (NASDAQ: AMDA), a company that develops and commercializes silicon nitride ceramics as a biomaterial platform, is pleased to announce it has reduced its total debt obligations to $12.9 million, a decrease of 47% from the prior year period, with the completion of the previously […]
Spinal Simplicity Lands Investor In Nueterra Capital, Advances Opportunity For FDA-Cleared Minuteman Fusion Device
LEAWOOD, Kan., May 3, 2016 /PRNewswire/ — Spinal Simplicity, a medical device company based in Overland Park, Kan., that develops innovative solutions to treat complex surgical problems, recently announced the closing of a financing round with an investment from private equity and advisory firm Nueterra Capital. After successfully introducing the Minuteman fusion device for lower back […]
SI-BONE Surpasses 20,000 Surgeries With Triangular iFuse Implant System
SAN JOSE, Calif., May 3, 2016 /PRNewswire/ — SI-BONE, Inc., a medical device company that pioneered the use of the iFuse Implant System® (“iFuse”), a minimally invasive surgical (MIS) device indicated for fusion for certain disorders of the sacroiliac (SI) joint, announced that iFuse has now been used in over 20,000 procedures worldwide by more than […]
Bone Therapeutics completes recruitment of its ALLOB Phase IIA spinal fusion study
BONE THERAPEUTICS, (Brussels:BOTHE) (Paris:BOTHE) (Euronext Brussels and Paris: BOTHE), the bone cell therapy company addressing high unmet medical needs in the field of bone fracture repair and prevention and spinal fusion, today announces that it has completed recruitment of its Phase IIA spinal fusion study for its allogeneic cell therapy product ALLOB®ahead of schedule. The […]
joimax Celebrates 15 Years During AANS Meeting
CHICAGO, IL–(Marketwired – April 29, 2016) – joimax®, the global acting German developer and marketer of technologies and treatment methods for minimally invasive endoscopic spinal surgery, celebrates its 15th anniversary during the American Association of Neurological Surgeons (AANS) annual scientific meeting in Chicago, held in combination with the company’s senior faculty meeting. The aim of […]
Stryker’s Spine Division Debuts 3D-Printed Tritanium® Posterior Lumbar Cage at AANS Meeting
ALLENDALE, N.J.–(BUSINESS WIRE)–Stryker’s Spine division will introduce the Tritanium Posterior Lumbar (PL) Cage, a 3D-printed intervertebral body fusion device that aids in lumbar spinal fixation for patients with degenerative disc disease, at the American Association of Neurological Surgeons Annual Meeting April 30–May 4, 2016, in Chicago (booth No. 3611). The Tritanium PL Cage is manufactured […]
Wenzel Spine Announces Commercial Launch of the VariLift-LX
AUSTIN, Texas–(BUSINESS WIRE)–Wenzel Spine, Inc., a pioneer medical device company focused on providing minimally invasive, expandable stand-alone alternatives to traditional spinal fusion, announced today the full commercial availability of the VariLift-LX System throughout the U.S. VariLift®-LX represents the next generation of Wenzel Spine’s innovative product lines of Expandable Stand-Alone interbody fusion devices. The zero-profile VariLift®-LX […]
Amendia Announces Transaction with Kohlberg & Company
MARIETTA, GA (PRWEB) APRIL 29, 2016–Amendia, Inc., a leading designer, developer, manufacturer and marketer of medical devices used in spinal surgical procedures, today announced the completion of a significant transaction and partnership that positions Amendia for accelerated growth and an enhanced value proposition for current and future customers and partners. Kohlberg & Company, L.L.C. (“Kohlberg”), […]
Intelligent Implant Systems Receives 510(k) Clearance for Two-Level Revolution
Intelligent Implant Systems announced today that the two-level components for the Revolution™ Spinal System were cleared by the FDA via 510(k) for sale in the United States. These components expand the capabilities of the system and allow Revolution™ to be utilized for two-level posterior thoraco-lumbar fusions. With one-level sales beginning in October of 2015, the […]
K2M Hosts Leading Spine Surgeons at the 2016 Meeting of Minds
LEESBURG, Va., April 28, 2016 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (Nasdaq:KTWO) (the “Company” or “K2M”), a global medical device company focused on designing, developing and commercializing innovative and proprietary complex spine and minimally invasive spine technologies and techniques, hosted more than 120 of the world’s leading spine surgeons for its annual Meeting of […]
Life Spine® to Host PULSE™ Lab and Attend AANS
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that it will be participating in the 84th AANS Annual Scientific Meeting in Chicago, April 30-May 4, 2016. The meeting is expected to attract over 3,000 neurosurgeons and other healthcare professionals […]
Full-year 2015 results and first-quarter 2016 revenue
SAFE ORTHOPAEDICS (Paris:SAFOR) (FR0012452746 – SAFOR), a company offering innovative ranges of sterile implants combined with their single-use instruments for back surgery, is announcing its full-year 2015 results and its first-quarter 2016 revenue. “In 2015, we continued to pursue our R&D efforts, launching trauma and fenestrated screws. A new cervical range is also under development […]
Q1 2016 revenues Strong increase in U.S. JAZZ sales: +106%
Bordeaux, Boston, April 28, 2016 – IMPLANET (Euronext: IMPL, FR0010458729, PEA-PME eligible; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, today announces its revenue for the 1st quarter of 2016. Ludovic Lastennet, CEO of Implanet, says: “The solid growth in the Group’s revenues is a result of the ongoing momentum and […]
Genesys Spine Wins International Award
AUSTIN, Texas, April 27, 2016 /PRNewswire-iReach/ — Genesys Spine is pleased to announce that the Binary® Anterior Cervical Plating System has been awarded the Iron A’ Design Award in the category of Scientific Instruments, Medical Devices and Research Equipment Design by the grand jury panel of the A’ Design Award & Competition, which consists of […]
NuVasive Rides Its Spinal Surgery Business Higher
April 26, 2016 (Motley Fool)– NuVasive’s first-quarter results were positive on all fronts. Revenue jumped almost 12% to $215.1 million, easily surpassing the consensus forecast among investors for sales of $206 million. NuVasive posted a GAAP net loss, but after adjusting for several one-time items, adjusted earnings of $0.31 per share were $0.04 greater than […]
Titan Spine Appoints Vice President of General Counsel
MEQUON, Wis.–(BUSINESS WIRE)–Titan Spine, a medical device surface technology company focused on developing innovative spinal interbody fusion implants, today announced the appointment of Ragan E. Cheney, JD, as Vice President, General Counsel. Ms. Cheney’s main responsibility will be to protect Titan Spine’s legal interests, and importantly, to ensure that the Company’s extensive intellectual property portfolio […]
Spinal Elements® Granted 50th U.S. Patent
CARLSBAD, CA (PRWEB) APRIL 26, 2016–Spinal Elements, a spine technology company, announced that they have been granted their 50th U.S. patent by the United States Patent and Trademark Office (USPTO). Spinal Elements’ U.S. patent portfolio encompasses a wide range of inventions in spine fusion and motion preservation technologies. It includes interbody devices, pedicle screw implants, […]
NuVasive Reports First Quarter 2016 Financial Results
SAN DIEGO, CA –(Marketwired – April 26, 2016) – NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, announced today financial results for the quarter ended March 31, 2016. First Quarter 2016 Highlights Revenue increased 11.8% to $215.1 million, or 12.3% on a constant currency basis; […]
MEDICREA Announces FDA Approval Of First Patient-Specific UNiD™ Cervical Rod For Spine Surgery (Video)
MEDICREA,® (Alternext Paris: FR0004178572 – ALMED) the only medical device company offering patient-specific implant solutions for the treatment of spinal conditions, has announced FDA approval of the first-ever patient-specific UNiD™Cervical rod for spine surgery, secured by their complementary PASS OCT® posterior cervical stabilization system. Immediately following the FDA clearance, MEDICREA successfully expanded their personalized UNiD™ […]
Pinnacle Spine Group Granted Chinese Patent
DALLAS, April 26, 2016 /PRNewswire/ — Pinnacle Spine Group, LLC, a developer of innovative spinal fusion solutions to address unmet surgical needs and improve outcomes, today announced that the Chinese Patent Office is in the process of issuing Chinese Patent Application No. 201180020517.1 titled Intervertebral Implants and Graft Delivery Systems and Methods as an enforceable […]
Woman drops lawsuit against Drobot hospital over “counterfeit” spinal screws
SANTA ANA, Calif., April, 2016 /PRNewswire/ — An Orange County woman has dismissed her lawsuit against HealthSmart Pacific, former owner of Pacific Hospital of Long Beach, after it was revealed that the spinal implants used in her surgery were not counterfeit, as she alleged in the wake of similar, highly publicized claims by a group of […]