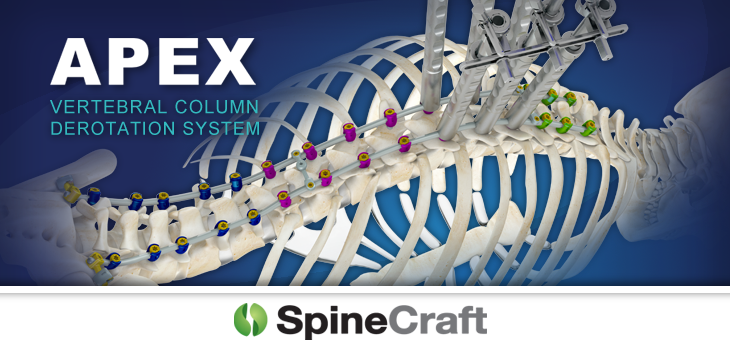
SpineCraft announced today that it has received clearance from the China Food and Drug Administration (CFDA) to market its APEX Spine System® in China. The APEX Spine System is designed for complex deformity correction as well as tumor, trauma and degenerative procedures and has proven clinical track record with over 15,000 surgeries performed since its launch in the US and internationally.
“This CFDA clearance is a significant milestone achievement for SpineCraft and further advances our strategy to establish SpineCraft as a true global spine company.” said SpineCraft CEO Wagdy Asaad, MD.
The China spine market has become the second most important after the USA and is projected to be worth over $1 billion by 2019. This is driven by China’s large population, rising standard of living, improving treatment options and growing ageing population; all signal significant room for growth.
About SpineCraft
SpineCraft is a privately-held US medical device company offering a wide range of products for addressing spine deformities &complex spine problems, minimally invasive spine surgery, cervical &thoracolumbar degenerative conditions, non-fusion applications and orthobiologics &bone substitutes.Surgeon input remains central to the way the company approaches working on new innovations and improving existing products. We hear and see, first hand, the concerns and obstacles that surgeons encounter which allow us to create intelligent implant&instrument solutions. This surgeon-centric approach results in more practical devices that provide intraoperative efficiency for surgeons, cost-effectiveness for hospitals &healthcare systems, and superior outcomes for patients.www.spinecraft.com