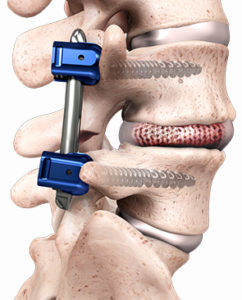
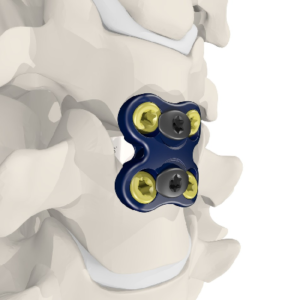
DOVER, Delaware, Oct. 3, 2018 /PRNewswire/ — CarboFix in Orthopedics LLC., has announced today that the U.S. Food and Drug Administration (FDA) has cleared its CarboClear® Carbon Fiber Transverse Connectors to be used in conjunction with its Carbon Fiber Pedicle Screw System, a novel device to surgically treat oncological patients, cleared by the FDA earlier […]
FDA
SCOUT:Spineology U.S. Clinical Trial for Interbody Fusion
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., an innovator in anatomy-conserving spine surgery, is excited to announce that John Chi, M.D., M.P.H., Associate Professor of Neurosurgery at Harvard Medical School and the Director of Neurosurgical Spinal Oncology at Brigham and Women’s Hospital in Boston, Massachusetts, presented 12-month outcomes data from Spineology’s SCOUT clinical trial at the recent […]
Stryker announces 510(k) clearance of SpineJack® Implantable Fracture Reduction System
KALAMAZOO, Michigan, USA, Sept. 26, 2018 /PRNewswire/ — Stryker announced today that it has received FDA 510(k) clearance to market the SpineJack Implantable Fracture Reduction System. The SpineJack system is indicated for use in the reduction of painful osteoporotic vertebral compression fractures. In the SAKOS clinical study, the SpineJack system demonstrated superiority to balloon kyphoplasty […]
Renovis Surgical Receives FDA Clearance for Tesera Trabecular Technology 3D-Printed Lateral Interbody Fusion System
REDLANDS, Calif. and AUSTIN, Texas, Sept. 26, 2018 /PRNewswire/ — Renovis Surgical Technologies, Inc. today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the Tesera® X Lateral Interbody Fusion System. Tesera X is the latest product to utilize Renovis’ proprietary 3D-printed porous architecture, which is made […]
Meditech Spine Receives FDA Clearance for its Cure™ Opel-C Plating System
ATLANTA, Sept. 18, 2018 /PRNewswire/ — Meditech Spine has received FDA 510(k) clearance to market the Cure™ Opel-C Plate System, an add-on technology to its already cleared CURE™ ACP System and Talos®-C (HA) Interbody Systems. With this approval, Meditech will now offer two additional CURE™ ACP plating options with its Talos® line of cervical interbodies made with the […]
SpineVision® Receives CE Mark for Next-Gen P.L.U.S.® Deformity-Correction Spinal System
ANTONY, France–(BUSINESS WIRE)–SpineVision announced today that it has received CE mark for its next-generation Pivot Link Universal System (P.L.U.S.) spine deformity-correction system. SpineVision will introduce the next-generation P.L.U.S. to spine surgeons at “EuroSpine 2018” (Booth #24C) in Barcelona, Spain (Sept. 19-21), and “NASS 2018” (Booth #1311) in Los Angeles (Sept. 26-29). Deformity of the spine includes any abnormality of the formation, alignment, or shape of the vertebral […]
CoreLink Surgical, LLC, Today Announced 510(k) Clearance to Market the M3™ Stand-Alone Anterior Lumbar (ALIF) System
ST. LOUIS–(BUSINESS WIRE)–CoreLink Surgical, LLC, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the M3™ Stand-Alone Anterior Lumbar (ALIF) System. Jay Bartling, CEO, said, “M3 is our first stand-alone interbody fusion device to feature 3D printing – I continue to be impressed by the ability of our design teams […]
FDA Grants Eden Spine 510(K) Clearance for Its Thoraco Lumbar Spine Locking Plate – SPHYNX™
GENEVA, SWITZERLAND (PRWEB) AUGUST 30, 2018–The SPHYNX™ is the ideal complement to Eden Spine proprietary, expandable titanium vertebral body replacement implant with rotatable endplates, the GIZA™ which provides multiple angulation options by simple endplates rotation. The GIZA™ is intended to replace and fuse a collapsed, damaged, or unstable vertebral body due to a tumor or […]
CoreLink, LLC Announces 510(k) Clearance for Foundation™ 3D Anterior Lumbar (ALIF)
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a vertically integrated manufacturer of spinal implant systems, today announced 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market The FoundationTM 3D Anterior Lumbar (ALIF) Interbody device. This marks the launch of the latest addition to CoreLink’s Foundation 3D Interbody Cage family. Jay Bartling, CEO, said, “The Foundation […]
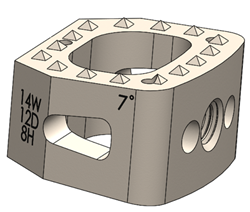
Mighty Oak Medical Receives FDA 510K Clearance for S2AI
July 25, 2018–Englewood, CO – Mighty Oak Medical announced today that its patient-specific, 3D printed FIREFLY® Pedicle Screw Navigation Guides have received an additional FDA 510K clearance for the expanded indications to include S2AI trajectory for sacral-iliac fixation in complex spinal reconstruction surgeries such as scoliosis. The precise mechanical guidance of patient-specific guides is ideal for […]
CarboFix’s CarboClear®, Carbon Fiber Pedicle Screw System Has Been Cleared to Be Used in the United States by the FDA
HERZELIYA, Israel, July 26, 2018 /PRNewswire/ — CarboFix Orthopedics LTD., is pleased to announce that the U.S. Food and Drug Administration (FDA) has given 510(k) clearance for the CarboClear® Carbon Fiber Pedicle Screw System, a novel device to surgically treat oncological patients in the United States. The CarboClear Pedicle Screw System is intended to restore […]
Zavation Announces 510(k) Clearance of the Normandy VBR System (Cervical and Thoracolumbar Expandable Corpectomy Cage)
FLOWOOD, Miss., July 17, 2018 /PRNewswire/ — Zavation, an employee-owned medical device company that designs, develops, manufactures and distributes medical device products, announced today that it has received 510(k) clearance from the FDA to market Normandy VBR System (Cervical and Thoracolumbar Expandable Corpectomy Cage). For More Information on Zavation’s Complete Product Portfolio, Visit: http://zavation.com/. The Normandy VBR System is an […]
FDA Clears First Nanotechnology Peek Devices for Spinal Intervertebral Fusion
NASHUA, N.H.–(BUSINESS WIRE)–Vallum Corporation, a medical device company, today announced that it has received clearance from the U.S. Food and Drug Administration (FDA) to market a polyetheretherketone (PEEK) spinal interbody fusion device with a PEEKplus® nanotextured1 surface created by Vallum’s proprietary and patented Accelerated Neutral Atom Beam (ANAB) technology. PEEKplus® is the first and only FDA-cleared […]
Acron Medical Announces FDA 510(k) Clearance of its Signature PEEK TLIF Interbody System, the ACRON Interbody
ORLANDO (PRWEB) JULY 16, 2018–Acron Medical, LLC (http://www.Acron-Medical.com), part of the spineMED Group, is a spine technology organization, dedicated to developing and commercializing globally innovative spinal implants. The company is proud to announce that it has received 510(k) clearance from the FDA for its signature new technology, the ACRON TLIF Interbody system. “The ACRON Interbody […]
Cutting Edge Spine Announces FDA 510(K) Clearance of its New HA Enhanced PEEK Interbody for the Cervical Spine, the EVOL ha-C
WAXHAW, N.C. (PRWEB) JULY 11, 2018//Cutting Edge Spine (http://www.CuttingEdgeSpine.com), a US leader in the development and commercialization of bio-active technologies for the spine, today announced the 510(k) clearance and commercial launch of it’s latest innovation, the EVOL ha-C cervical inter-body system. The EVOL ha-C system is made of PEEK-OPTIMA HA Enhanced material supplied by Invibio […]
Tenon Medical, Inc. Receives FDA Clearance For Sacroiliac Joint Fusion System
SAN RAMON, Calif., June 20, 2018 /PRNewswire/ — Tenon Medical, Inc., a manufacturer of minimally invasive instruments and implants for sacroiliac joint fusion surgery, today announced that it has received 510(k) clearance from the United States Food and Drug Administration (FDA) for the Catamaran™ Sacroiliac Joint Fixation System (CAT SIJ Fixation System) specifically indicated for sacroiliac joint fusion […]
Centinel Spine Announces 510(k) Clearance of FLX™ Platform of 3D Printed All-Titanium Interbodies
NEW YORK, June 7, 2018 /PRNewswire/ — Centinel Spine, LLC is pleased to announce that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its FLX™ Platform of Integrated Interbody™ and non-integrated interbody fusion devices. Centinel Spine is the largest privately-held spine company, focused on anterior column reconstruction. FLX devices are 3D-printed, […]
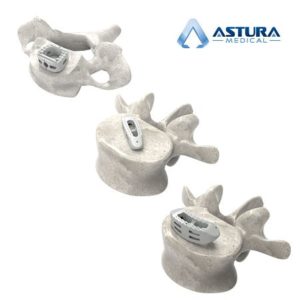
Astura Medical Receives FDA 510(k) Clearance For Cervical and Lumbar HA PEEK Interbody Systems
CARLSBAD, CA – June 4, 2018 – Astura Medical, a high growth, innovative spine technology company, today announced that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its ALTA ACDF Interbody Spacers and HALF DOME Lumbar Interbody Spacers in PEEK-OPTIMAä HA Enhanced from Invibio Biomaterial Solutions. Combining the clinically proven […]
Spineology Rampart One™ Standard ALIF Interbody Fusion System Gains FDA Clearance for Stand-Alone Use
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., announces FDA clearance of its Rampart One™ Standard ALIF device allowing it to be used with or without supplemental fixation. “Following the success of our beta launch, this stand-alone clearance will provide additional momentum as we prepare for the full market release of the Rampart One ALIF Interbody Fusion System,” […]
Camber Spine Announces 510(k) Approval Of ENZA™-A Titanium ALIF
WAYNE, Pa., June 4, 2018 /PRNewswire/ — Camber Spine today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its ENZA™-A Titanium Anterior Lumbar Interbody Fusion (ALIF) system, a unique, minimally invasive interbody fusion device providing integrated fixation. Daniel Pontecorvo, CEO, said, “ENZA™-A leverages two of Camber […]
SpineFrontier Receives FDA Approval for A-CIFT SoloFuse HA Standalone System
BOSTON (PRWEB) MAY 31, 2018–SpineFrontier has received FDA approval for A-CIFT SoloFuse HA, its next wave of fusion technology, designed and developed with intention by their team of engineers. As the first company to receive FDA clearance on HA PEEK in the U.S., SpineFrontier produced the A-CIFT SoloFuse HA standalone system to leverage the familiarity […]
Zimmer Biomet Announces 510(k) Clearance for Zyston® Strut Open Titanium Interbody Spacer System
WARSAW, Ind., May 22, 2018 /PRNewswire/ — Zimmer Biomet Holdings, Inc. (NYSE and SIX: ZBH), a global leader in musculoskeletal healthcare, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Zyston® Strut Open Titanium Interbody Spacer System. This marks Zimmer Biomet’s first titanium spinal implant manufactured via […]
Xtant Medical Receives FDA 510(K) Clearance for InTice™-C Porous Titanium Cervical Interbody System
BELGRADE, MT / ACCESSWIRE / May 21, 2018 / Xtant Medical Holdings, Inc. (NYSE American: XTNT), a leader in the development of regenerative medicine products and medical devices, announces U.S. Food and Drug Administration (FDA) 510(k) clearance for InTice™-C Porous Titanium Cervical Interbody System. InTice-C is designed using OsteoSync Ti, a best-in-class, cost-effective, highly porous titanium […]
Paradigm Spine Receives FDA Pre-Market Approval (PMA) For First Of Its Kind Disposable Instrument Kit
NEW YORK, May 2, 2018 /PRNewswire/ — Paradigm Spine, LLC, a leader in providing motion preservation solutions for the treatment of lumbar spinal stenosis, today announced that the U.S. Food and Drug Administration (FDA) has granted pre-market supplemental approval (PMA) for its coflex® Interlaminar Stabilization® disposable instrument kit. This marks the first approved disposable spinal instrument set for a […]
CTL Wins FDA Approval for Seurat Universal Pedicle Screw
DALLAS, TX / ACCESSWIRE / March 29, 2018 / CTL Medical Corporation, a Dallas-based medical device manufacturing and service company, has recently secured clearance and approval from the Food and Drug Administration to market its new SEURAT Universal Pedicle Screw System for the practice of spine fusion surgery. This is the second FDA clearance for the […]
Life Spine Announces FDA Clearance of PLATEAU®-LO Insert and Rotate Spacer System
HUNTLEY, Ill.–(BUSINESS WIRE)–Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that the U.S. Food and Drug Administration (FDA) has given 510(k) marketing clearance to the PLATEAU-LO Insert and Rotate Spacer System. “PLATEAU-LO is my go-to spacer for micro-invasive lumbar interbody fusions. […]
NeuroStructures, Inc. announces the 510(k) U.S. FDA clearance of the Cavetto®-SA Stand Alone Cervical Cage System and the Cortina™Lumbar Cage System
NeuroStructures, Inc. announces the 510(k) U.S. FDA clearance of the Cavetto®-SA Stand Alone Cervical Cage System and the Cortina™Lumbar Cage System . The Cavetto-SA Stand Alone Cervical Cage System comprises of Cavetto-SA cages and screws designed to optimize endplate contact and resist subsidence. The system uses a simple and unique One-Step locking mechanism to allow a […]