BIOMECH Paonan will feature their new NEST 3D printing Titanium Interbody System, the Trend II innovative Spinal Fixation System, and the more successful stories with Rocker, the Interspinous Process Distractor System at the Global Spine Congress 2019 in Toronto, Canada. Exhibit Period: May 16-18, 2019 Venue: Metro Toronto Convention Centre – MTCC Time: 16th May, 8.00 – 19.30 […]
2019
Orthofix Reports First Quarter 2019 Results
LEWISVILLE, Texas–(BUSINESS WIRE)–May 6, 2019– Orthofix Medical Inc. (NASDAQ:OFIX) today reported its financial results for the first quarter ended March 31, 2019. Net sales were $109.1 million, earnings per share (“EPS”) was $0.05 and adjusted EPS was $0.27. “For the first quarter, our top and bottom line results were slightly better than anticipated and we remain confident about our expected sales acceleration […]
Alphatec Announces Commercial Launch Of PLIF IdentiTi™ Posterior Straight Porous Titanium Interbody System Implant And AlphaGRAFT® DBM Fiber
CARLSBAD, Calif., May 06, 2019 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (“ATEC” or the “Company”) (Nasdaq: ATEC), a medical device company dedicated to revolutionizing the approach to spine surgery, announced today the commercial releases of its PLIF IdentiTi-PS Porous Titanium Interbody Implant System for Posterior Lumbar Interbody Fusion Procedures (PLIFs) and its AlphaGRAFT Demineralized Bone […]
U.S.Market: Spine Companies to Follow: Part I
According to iData Research, the global spine market is valued at over $14.4 billion USD and is projected to approach $18 billion by 2023. The largest among the regions is undoubtedly the U.S. market, valued at $7.7 billion in 2017. The US TOP PLAYERS The 10 top spine players in the US market are the […]
SAGICO’s Growth Expands Post the Annual AANS Scientific Meeting
TAMPA, FLA. (PRWEB) MAY 01, 2019–Spinal Analytics & Geometrical Implant Co. (SAGICO), an innovative spinal implant company, announced today that SAGICO has expanded its active surgeon base to include seven states with the eighth state currently on the surgical rotation schedule for May. The growth of SAGICO continues with a level mixture of orthopedic surgeons […]
DiFusion Announces Data for ZFUZE Immuno-Stealth Surgical Polymer
AUSTIN, Texas–(BUSINESS WIRE)–DiFusion Technologies, Inc. today announced promising data for the company’s ZFUZE Immuno-Stealth™ Surgical Polymer. The data and the importance of the immune system response in new technologies such as ZFUZE were debated at the Castellvi Spine Symposium in Key West in April. Stephen Badylak, DVM, MD, PhD, Deputy Director of the McGowan Institute […]
Globus Medical Reports First Quarter 2019 Results
AUDUBON, Pa., May 02, 2019 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced its financial results for the first quarter ended March 31, 2019. Worldwide sales were $182.9 million, an increase of 4.9% as reported First quarter net income was $33.2 million, a decrease of 16.0% Diluted earnings per share (EPS) was $0.33 and non-GAAP diluted EPS was $0.36 […]
New Study Strongly Supports the Safety of Centinel Spine’s prodisc® C Cervical Total Disc Replacement System
NEW YORK, May 2, 2019 /PRNewswire/ — Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced findings of a new study first presented at the annual meeting of the International Society for the Advancement of Spinal Surgery (3-5 April 2019, Anaheim, CA). Jack E. Zigler, MD (Texas Health Center […]
NuVasive Straightens Up to Start 2019
Dan Caplinger, The Motley Fool May 2, 2019–NuVasive (NASDAQ: NUVA) has been at the forefront of medical advancements in surgical procedures. With its focus on surgery to treat spinal conditions, NuVasive’s minimally disruptive surgical platform and accessories have played a key role in driving more effective treatment options for patients suffering from these health conditions. […]
RTI Surgical® Announces First Quarter 2019 Results
Deerfield, Ill., May 2, 2019 – RTI Surgical Holdings, Inc. (Nasdaq: RTIX), a global surgical implant company, reported operating results for the first quarter of 2019. First Quarter 2019 Highlights: Revenue of $69.7 million, inclusive of $2.0 million from acquisition of Paradigm Spine Net loss of $9.1 million, inclusive of $9.0 million of net non-recurring […]
SeaSpine Reports First Quarter 2019 Financial Results
CARLSBAD, Calif., May 01, 2019 (GLOBE NEWSWIRE) — SeaSpine Holdings Corporation (NASDAQ: SPNE), a global medical technology company focused on surgical solutions for the treatment of spinal disorders, announced today financial results for the quarter ended March 31, 2019 and updated its financial outlook for 2019. Summary First Quarter 2019 Financial Results Revenue of $36.2 million, an increase of 9% year-over-year […]
NuVasive Announces First Quarter 2019 Financial Results
SAN DIEGO – May 1, 2019 – NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced financial results for the quarter ended March 31, 2019. First Quarter 2019 Highlights Revenue increased 5.5% to $274.8 million, or 6.4% on a constant currency basis; GAAP operating […]
AIS-C Stand-Alone System Honored as a Finalist in the 2019 Medical Design Excellence Awards
Genesys Spine is pleased to announce that the AIS-C Stand Alone System has been selected as a finalist in the Implant & Tissue Replacement Products category of the 21st Annual Medical Design Excellence Awards competition. The 2019 winners will be announced at the MDEA Ceremony on Tuesday, June 11, 2019 in conjunction with the MD&M East […]
Life Spine Announces Continued Revenue Growth of SIMPACT® Sacroiliac Fixation System
HUNTLEY, Ill.–(BUSINESS WIRE)– Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced today that they exited Q1 of 2019 at a month-over-month sales growth rate of 73% in 2019 for the SIMPACT Sacroiliac Joint Fixation system. “Sacroiliac Joint dysfunction is a debilitating condition […]
Did you Know that there are at least 60 Lateral Cages in the market?
The Lumbar Lateral Cage surgery for many years has been a growing trend. According to Allied Market Research, the global interbody fusion cage market was valued at $1,818 million in 2016, and is expected to reach $2,309 million by 2023 at a CAGR of 3.4% during the forecast period. The lateral approach LLIF (Lateral Lumbar […]
Safe Orthopaedics announces its 2018 results and its revenues for the first quarter of 2019
Eragny-sur-Oise, France, April 26, 2019 – SAFE ORTHOPAEDICS (Paris:SAFOR) (FR0012452746 – SAFOR), a company specializing in the design and marketing of single-use implants and instruments for the minimally invasive treatment of spine fracture pathologies, today announced its 2018 annual results and its sales for the first quarter of 2019. The 2018 Registration Document including Safe Orthopaedics’ […]
Centinel Spine prodisc Total Disc Replacement Patients Team Together at Zurich Classic
New York, NY, April 25, 2019 –Centinel Spine®, LLC, the largest privately-held spine company focused on anterior column reconstruction, today announced that six-time PGA Tour Winner, Rory Sabbatini, and four-time PGA Tour Winner, Brian Gay have teamed up to play together during the Zurich Classic two-man PGA team event at TPC Louisiana in Avondale, La […]
Nexxt Spine continues investment in additive to drive medical device innovation
Indiana-based manufacturer installs fifth Concept Laser Mlab machine as it scales to tap further into the growing global spinal implant market “Additive is booming,” says Alaedeen Abu-Mulaweh, director of engineering at Nexxt Spine a medical device company focused on designing, manufacturing and distributing innovative spinal implant solutions, who is about to take delivery of his fourth and […]
Implanet Announces 1st Quarter 2019 Revenue
BORDEAUX, France & BOSTON–IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee surgery implants today announced its revenue for the first quarter of 2019. € ‘000s – IFRS* Q1 2019 Q1 2018 Change Spine (JAZZ) 1,129 1,050 8% Knee 775 817 -5% Total revenue […]
Confidentiality Agreements and Your Medical Device Idea (Free Template)
by Lawrence Binder– Chairman at Binder Biomedical, Inc Do I need to establish Confidentiality? I regularly advise clients on the importance of protecting your new medical device ideas and how that relates to your investment in developing them. My general recommendation is protecting your idea as a 2-tiered approach. First, establishing Confidentiality with anyone you share […]
Stryker reports first quarter 2019 operating results
Kalamazoo, Michigan, April 23, 2019 (GLOBE NEWSWIRE) — Stryker (NYSE:SYK) reported operating results for the first quarter of 2019: First Quarter Highlights Reported net sales increased 8.5% to $3.5 billion Organic net sales increased 7.3% Reported operating income margin of 15.0% Adjusted operating income margin(1) expanded 10 bps to 25.1% Reported EPS decreased 6.0% to $1.09 Adjusted […]
Orthofix Announces First US Patient Implants Following FDA Approval of the M6-C Artificial Cervical Disc Designed to Treat Cervical Disc Degeneration
LEWISVILLE, Texas–(BUSINESS WIRE)–Apr. 23, 2019– Orthofix Medical Inc. (NASDAQ:OFIX), a global medical device company focused on musculoskeletal products and therapies, today announced the first commercial implants of patients with the M6-C™ artificial cervical disc. The Center for Disc Replacement at Texas Back Institute (TBI) in Dallas, Texas recently implanted four patients suffering from single level […]
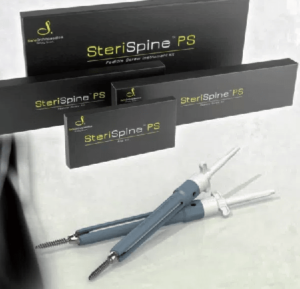
Safe Orthopaedics Announces the Approval of the SteriSpineTM PS in Japan
ERAGNY-SUR-OISE, France–Safe Orthopaedics (Paris:SAFOR) (FR0012452746 – SAFOR), a company specialized in the design and marketing of single-use implants and instruments improving the minimally invasive treatment of spinal fracture conditions, is today announcing the approval of the SteriSpineTM PS in Japan. It was received by KiSCO Co., Ltd. (KiSCO) in March 2019, commencing an exclusive commercialization […]
Clinical Study Demonstrates NuVasive Cohere® Porous PEEK™ Spinal Interbody Promotes Improved Early Outcomes Over Structural Allograft And Smooth PEEK
SAN DIEGO – April 22, 2019 – NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced the results of the study “Early Clinical Outcomes Comparing Porous PEEK, Smooth PEEK, and Structural Allograft Interbody Devices for Anterior Cervical Discectomy and Fusion,” published […]
Stand alone and 3D printed cages have revolutionized the ALIF segment.Learn about 61 Differentiated Lumbar Anterior Cages
According to Allied Market Research, the global interbody fusion cage market is projected to reach $2,309 million by 2023.The lumbar segment capture the highest share of about 30% and is expected to remain dominant throughout 2023. This is attributed to the growing popularity of lumbar spine fusion techniques due to their ability to provide long-term […]
SpineCraft Announces the introduction of the ASTRA SPINE SYSTEM POWER Instrumentation module
The ASTRA SPINE SYSTEM was introduced in early 2017 and includes a comprehensive implant and instrument offering for complex spine, deformity, degenerative and MIS Spine procedures. The new ASTRA POWER INSTRUMENTATION includes taps and drivers that are uniquely designed to improve surgical ergonomics, reduce operating fatigue and provide better precision when implanting spinal implants. Dr. […]
Implanet reports full-year 2018 results
Bordeaux, Boston, April18, 2019 – 5:45 CEST – Implanet (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral and knee-surgery implants, today announces its results for the financial year ended on December 31, 2018, and approved by the Board of Directors on April 18, 2019. “In 2018, […]