Posterior spine fusion with screw fixation is a common and reliable method of instrumentation of the spine for a variety of spine pathologies when the ultimate aim of surgery is the fixation of the spine to allow bony fusion. The mainstay of this kind of surgery has been the use of pedicle screw fixation. The trajectory of pedicle screws aims to transverse the pedicle down its anatomical axis, with a screw specifically designed for fixation in cancellous bone. More recently, Santoni et al., in 2009 proposed using an alternative trajectory, a cortical bone based fixation technique, which utilizes a different path down the pedicle with the aim of maximizing the contact between screw and cortical bone.
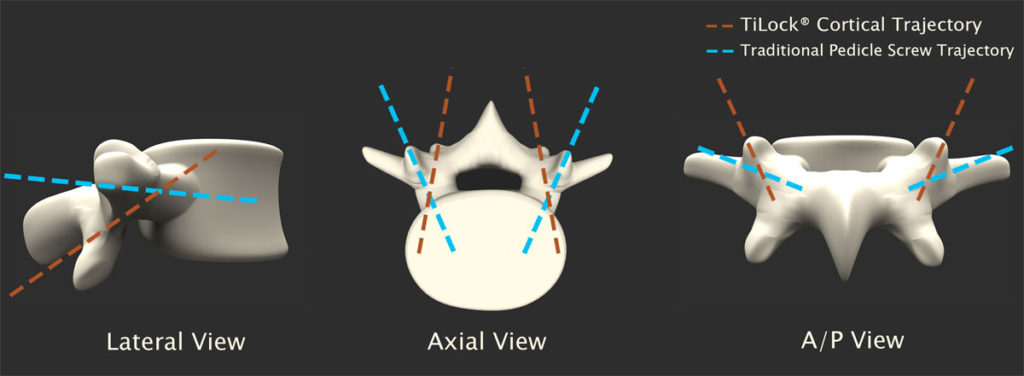
Posterior lumbar spine fusion using the cortical based trajectory has advantages over traditional pedicle screw techniques including reduced dissection and tissue trauma, increased biomechanical stability in both high and low quality bone and is thereby a valid alternative to traditional techniques.¹
The Genesys Spine TiLock® Cortical Spinal System is designed to provide surgeons and their patients with a robust, anatomy-conserving alternative to traditional pedicular fixation.

Features:
• Low-profile tulip head
• Over-the-wire insertion for a simple, less-invasive approach
• Reduced tulip head that allows for efficient screw placement with nominal bony removal
• Corticocancellous thread pattern for maximum fixation in bone
• Tapered cortical self-tapping thread for initial placement
About Genesys Spine
Founded in December of 2009, Genesys Spine’s mission was to bring a suite of medical implants and instruments with novel characteristics into a mature spinal fusion market. Since that time, Genesys has released four new FDA cleared product lines with several other products in various stages of the design and development cycle. Genesys has also developed several proprietary instruments to aid in the use of its products. All of Genesys’ inaugural products including our system specific instruments, have garnered very positive feedback from the marketplace. Our implants, with their proprietary features, all remain within existing parameters for today’s current reimbursement codes.
For more information please visit: http://www.genesysspine.com
