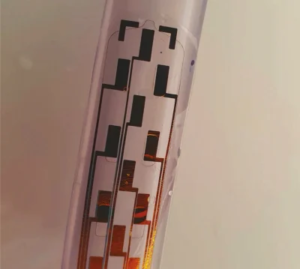SAN DIEGO, April 17, 2023 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced that proxy advisory firm, Glass Lewis & Co., LLC (“Glass Lewis”), recommends that NuVasive shareholders vote “FOR” the shareholder proposal to adopt the merger agreement with Globus Medical […]
2023
Aurora Spine Announces Initial Surgeries Conducted Using its SiLO TFX™ MIS Sacroiliac Joint Fixation System
Carlsbad, Calif., April 17, 2023 (GLOBE NEWSWIRE) — Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSXV:ASG) (OTCQB: ASAPF), a designer and manufacturer of innovative medical devices that improve spinal surgery outcomes, today announced that initial surgeries using its proprietary SiLO TFX™ MIS Sacroiliac Joint Fixation Device have been performed in recent weeks. Over a […]
Clinical Study Demonstrates Favorable Patient Outcomes with the dualPortal® Endoscopic TLIF with dualX®, the dualLIF® System
IRVINE, CALIF. (PRWEB) APRIL 14, 2023–Amplify Surgical, Inc., a medical device company focused on innovative minimally-invasive surgery for the lumbar spine, today announced positive data from a retrospective study on its dualLIF® System, the synthesis of dualPortal® Spinal Endoscopy and dualX® Dual-Expanding Interbody Technology. The study, “The Use of Dual Direction Expandable Titanium Cage With Biportal Endoscopic Transforaminal Lumbar Interbody […]
Leading Independent Proxy Advisory Firm ISS Recommends NuVasive Shareholders Vote “FOR” the Proposed Merger with Globus Medical
SAN DIEGO, April 14, 2023 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally integrated solutions, today announced that leading independent proxy advisory firm Institutional Shareholder Services (“ISS”) recommends that NuVasive shareholders vote “FOR” the shareholder proposal to adopt the merger agreement with Globus Medical (NYSE: GMED) […]
(UPDATED 2023): 16 Turkish Spine Companies to Know…!
Spine surgery has been performed in Turkey since 1897. Today, all spinal procedures including instrumentation, endoscopic spine surgery, spinal arthroplasty, vertebroplasty and kyphoplasty are performed in Turkey. The number of complex spinal operations have increased during the last decades. Statistics show that approximately 70−80% of spine surgery is performed by neurosurgeons and 20−30% by ortho−spine […]
Safe group announces revenues of €1.5 million in Q1 2023
Éragny-sur-Oise, Fleurieux-sur-l’Arbresle, April 13th, 2023 at 5:45 p.m. CET – Safe (ALSAF), a company specializing in the design, manufacture and marketing of ready-to-use technologies for back surgery, particularly safe for spinal fractures treated in emergency (the “Company”) announces its first quarter 2023 revenues and cash position. In the first quarter of 2023, the Safe Group’s revenue […]
SpineGuard announces its full-year 2022 financial results and its first quarter 2023 sales
PARIS and BOULDER (CO), April 13, 2023, at 06:00 pm CEST – SpineGuard (FR0011464452 – ALSGD), an innovative company that deploys its DSG (Dynamic Surgical Guidance) sensing technology to secure and streamline the placement of bone implants, announced today its full-year 2022 financial results, as approved by the Board of Directors on April 13, 2023, and its first quarter 2023 […]
Spineway 2023 : high increase in turnover
Press release Ecully, April 13, 2023 – Spineway has achieved a total revenue of €2.4 million as of Q1 2023, representing growth of 79% compared with Q1 2022, mainly driven by sales of Spine Innovations’ disc prostheses. Adjusted for this contribution, quarterly revenue remains robust and represents an all-time high of €1.4 million despite a temporary delay in Latin America due to regulatory procedures for the approval of […]
Premia Spine Completes FDA Site Inspections With No FDA Observations
NORWALK, Conn.–(BUSINESS WIRE)–Premia Spine, a medical technology company changing the way debilitating chronic leg and back pain is treated, today announced the U.S. Food and Drug Administration (FDA) has completed four Bioresearch Monitoring (BIMO) inspections as part of the PMA approval process for the TOPS facet joint replacement system. The inspections at two company facilities […]
ZimVie Announces the Recent Launches of RegenerOss® CC Allograft Particulate and RegenerOss® Bone Graft Plug
WESTMINSTER, Colo., April 13, 2023 (GLOBE NEWSWIRE) — ZimVie Inc. (Nasdaq: ZIMV), a global life sciences leader in the dental and spine markets, today announced the recent launches of two additions to its biomaterials portfolio, the RegenerOss® CC Allograft Particulate, a natural blend of cortical and cancellous bone particles that can be used to fill bony […]
Aurora Spine Announces Presentation on DEXA Technology at the American Association of Neurological Surgeons Annual Meeting
Carlsbad, California, April 12, 2023 (GLOBE NEWSWIRE) –Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSXV:ASG) (OTCQB: ASAPF), a designer and manufacturer of innovative medical devices that improve spinal surgery outcomes, today announced that its DEXA Technology will be highlighted at a presentation delivered by Dr. Sebastian Koga, neurosurgeon at Forrest Health Institute of Neuroscience, […]
Centinel Spine® Achieves Record Fourth Quarter 2022, Driven by Launch of prodisc® Cervical Total Disc Replacement Devices
WEST CHESTER, Pa., April 12, 2023 /PRNewswire/ — Centinel Spine®, LLC, (“the Company”) a leading global medical device company addressing cervical and lumbar spinal disease by providing the most robust and clinically-proven total disc replacement technology platform in the world (prodisc®), today announced record fourth quarter 2022 revenue achievement driven by growth in the Company’s Motion Preservation […]
SMAIO Reports 2022 Annual Results and Confirms 2023 Outlook
Lyon (France), April 11, 2023 – 6 pm CEST – SMAIO (Software, Machines and Adaptative Implants in Orthopaedics – Euronext Growth Paris ISIN: FR0014005I80 / Ticker: ALSMA), a French player specialized in complex spine surgery with a global offer comprising software, today published its 2022 annual results1 approved by the Board of Directors on April […]
IMPLANET receives FDA clearance for its SqualeTM anterior cervical cage range
Bordeaux, Boston, April 11, 2023 – 7:30 a.m. CEST: IMPLANET (Euronext Growth: ALIMP, FR0013470168, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral implants and the distribution of technological medical equipment, today announced the Food and Drug Administration (FDA) 510(k) clearance of its SqualeTM anterior cervical cage. The SqualeTM anterior cervical cage, from the […]
ZSFab Announces Second Clinical Use of the ZSFab Cervical Interbody System for ACDF
WALTHAM, Mass., April 10, 2023 (GLOBE NEWSWIRE) — ZSFab, Inc., a 3D-printing medical device company, is pleased to announce that their second case in the country last week was completed by Dr. Jeffrey Moore, Orthopaedic Surgeon in Oklahoma City, Oklahoma. Dr. Moore performed a 3-level ACDF procedure with theZSFab Cervical Interbody System at The Surgical Hospital […]
(UPDATED 2023): 10 Brazilian Spine Companies to know..!
The Brazil spinal surgery devices market is expected to register a CAGR of 4.97% over the 2022-27 period. The market leaders are global companies as Medtronic, Stryker, DePuy, Zimmer and Globus Medical.The nature of the public health system in Brazil encourages the participation of local companies that can provide devices at a lower price, as […]
ZSFab Announces First US Clinical Use of ZSFab Cervical Interbody System for ACDF
WALTHAM, Mass., April 05, 2023 (GLOBE NEWSWIRE) — ZSFab, Inc., a 3D-printing medical device company, is proud to announce that the ZSFab Cervical Interbody System has been successfully used in its first clinical case in the United States with Dr. Fernando Villamil, Orthopaedic Surgeon at the Center for Orthopedic Reconstruction and Excellence (CORE) Hospital in […]
Spinal Resources Inc. Unveils Groundbreaking Spine Platform for Improved Patient Care
FORT LAUDERDALE, FLA. (PRWEB) APRIL 04, 2023 Spinal Resources Inc. (SRI), a leading innovator in spinal technology, has announced the FDA-approval of its state-of-the-art, Solutions Based Spine Platform (SBSP). The unique platform is designed to provide surgeons with an unparalleled solution to address and assist in the reduction of critical spinal conditions like Proximal Junctional Kyphosis […]
Aurora Spine Receives IRB Multi-Site Selection Approval for its DEXA-C™ Cervical Interbody System
Carlsbad, Calif., April 04, 2023 (GLOBE NEWSWIRE) — Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSXV:ASG) (OTCQB:ASAPF), a designer and manufacturer of innovative medical devices that improve spinal surgery outcomes, today announced it has received Institutional Review Board (IRB) multi-site selection approval for its new multicenter study of DEXA-C™ Cervical Interbody System. The study […]
(UPDATED 2023): +12 Single-Use Spinal Systems Companies to Know! Do they really have Advantages?
In recent years, a new segment focused on single-use systems has emerged in the spine market. The pioneering companies were the French Safe Orthopedics and the American Xenco Medical. Both were visionaries and launched into the creation and consolidation of this new market. Its success has attracted new competitors such as the Swiss company Neo Medical, which has entered […]
ZimVie Announces Expanded Reimbursement and Exemplary Clinical Rating for Mobi-C®
WESTMINSTER, Colo., April 03, 2023 (GLOBE NEWSWIRE) — ZimVie Inc. (Nasdaq: ZIMV), a global life sciences leader in the dental and spine markets, today announced that the French Republic has published its approval for the reimbursement of the Mobi-C® Cervical Disc in both the public and private sectors in France. In addition, the clinical data for […]
Inflatable, Shape-Changing Spinal Implants Could Help Treat Severe Pain
A team of engineers and clinicians has developed an ultra-thin, inflatable device that can be used to treat the most severe forms of pain without the need for invasive surgery. The device, developed by researchers at the University of Cambridge, uses a combination of soft robotic fabrication techniques, ultra-thin electronics, and microfluidics. The device is […]
Camber Spine receives FDA 510k Clearance of two products!
Camber is excited to announce the 510(k) clearance of ENZA®-O Interbody System and the cervical fixation anchors designed for use with SPIRA®-C Integrated interbodies. ENZA®-O Interbody System Building on the innovative ENZA® Technology platform, ENZA®-O is a minimally invasive solution for the OLIF approach for surgeons who prefer an integrated fixation option. Designed for minimal […]
Surgalign Holdings, Inc. Announces Fourth Quarter and Full Year 2022 Results
DEERFIELD, Ill., March 30, 2023 (GLOBE NEWSWIRE) — Surgalign Holdings, Inc., (NASDAQ: SRGA) a global medical technology company focused on elevating the standard of care by driving the evolution of digital surgery, today reported operating results for its fourth quarter and full year 2022 ending December 31, 2022. 2022 Fourth Quarter and Subsequent 2023 Corporate Highlights: “I’m […]
3Spine Announces 200 of 300 Patients Enrolled in Ongoing Studies
CHATTANOOGA, Tenn., March 30, 2023 /PRNewswire/ — 3Spine, Inc., today announced completion of the 50th MOTUS lumbar total joint replacement in the United States under FDA Investigational Device Exemption (IDE) and the 150th Real-World Evidence (RWE) lumbar fusion control case. This second milestone marks full enrollment of the RWE fusion study, allowing all eligible sites to recruit MOTUS patients upon conversion. “Our […]
CoreLink Announces FDA Clearance of Siber® Ti–3D Printed SI Joint Fusion System
ST. LOUIS, March 29, 2023 /PRNewswire/ — CoreLink, LLC , a leading designer and manufacturer of innovative spinal implant systems announced today the 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its Siber® Ti Sacroiliac Joint Fusion System. The Siber Ti System offers a comprehensive portfolio of fully porous, nano surfaced, 3D-printed implants. These new implants incorporate CoreLink’s […]
Spineart Peek Cages&ROMEO®2 Posterior Thoracolumbar Fixation System now MDR Approved!
Spineart is thrilled to share a key milestone for the company: the PEEK Posterior Lumbar JULIET® cages as well as our PEEK Cervical TRYPTIK® cages and our Open and Minimally Invasive Thoracolumbar Fixation System ROMEO®2 are now CE Marked under the MDR 2017/745 (Medical Device Regulation) by the notified body TUV Sud. This is a step […]