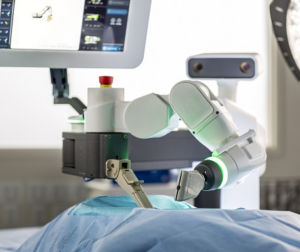
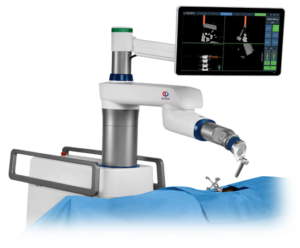
(INVESTORS.COM)–With artificial intelligence now firmly entrenched in many hospital operating rooms, the field of robotic surgery is starting to get competitive.Giant companies like Alphabet (GOOGL), Johnson & Johnson (JNJ) and Medtronic(MDT) are training their sights on Intuitive Surgical (ISRG), the king of robotic surgery companies. But analysts say the booming medical technology segment has lots of room to run. Robotic surgery is […]
2018
Medtronic – Adding Some Robotics To The Mix
(SEEKINGALPHA.COM)– (MDT) has announced a rather interesting bolt-on deal, although the timing of the deal can be questioned. The company is acquiring robotic guidance system provider Mazor Robotics (MZOR) in a $1.64 billion deal, as the cash component comes in $300 million less thanks to net cash holdings of Mazor, and the fact that Medtronic already holds a […]
10 MORE Spine Companies to Know with “S”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems.But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all the […]
Medtronic to Acquire Mazor Robotics
DUBLIN and CAESAREA, Israel – September 20, 2018 – Medtronic plc (NYSE:MDT), a global leader in medical technology, and Mazor Robotics (NASDAQ:MZOR, TASE:MZOR.TZ), a pioneer in the field of robotic guidance systems, today announced the companies have entered into a definitive merger agreement under which Medtronic will acquire all outstanding ordinary shares of Mazor for […]
Zavation Medical Products, LLC, a LongueVue Capital Portfolio Company, Completes Investment in Pan Medical U.S. Corp.
JACKSON, Miss., Sept. 18, 2018 /PRNewswire/ — Zavation Medical Products (“Zavation”), a LongueVue Capital (“LVC”) portfolio company, is pleased to announce it has partnered with the management team of Pan Medical U.S. Corp (“PanMed” or the “Company”) to acquire the Company. This partnership broadens Zavation’s service offering by adding a full suite of minimally invasive products, including the […]
Globus Medical Acquires Surgimap®
AUDUBON, Pa., Sept. 13, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the acquisition of Nemaris Inc., a privately held company that markets and develops Surgimap®, a leading surgical planning software platform. Surgimap® allows healthcare professionals to simulate potential surgical outcomes and share medical imaging globally to improve […]
K2M Group Holdings, Inc. Announces Definitive Agreement To Be Acquired by Stryker Corporation
LEESBURG, Va., Aug. 30, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (Nasdaq: KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced a definitive merger agreement with Stryker Corporation (“Stryker”, NYSE: SYK) pursuant to which Stryker has agreed to acquire […]
SPINE Companies Catalogue From A to Z: 10 Spine Companies to know with “R”
Over the recent years, global spinal fusion market has been witnessing growth, on account of several driving factors including rising healthcare awareness amongst consumers, surging prevalence of spinal deformities due to accidents along with increasing health expenditure in developing countries. Moreover, ongoing demographic shift towards geriatric population with significant population suffering from inveterate spinal ailments, […]
Caldwell Cassady & Curry Helps Acantha Win $8.2 Million Patent Infringement Verdict Against Medical Device Giant DePuy Synthes
GREEN BAY, Wisc., Aug. 23, 2018 /PRNewswire/ — The Dallas-based intellectual property and business litigation law firm Caldwell Cassady & Curry has won an $8.2 million patent infringement verdict for Acantha LLC against medical device manufacturer DePuy Synthes, a subsidiary of health care giant Johnson & Johnson (NYSE : JNJ ). A jury of five […]
China: Innovative Hybrid PEEK-titanium Expandable Cage for DLIF and OLIF Procedures Demonstrated
THORNTON CLEVELEYS, ENGLAND (PRWEB) AUGUST 14, 2018//The growing interest in PEEK-OPTIMA™ spinal implants in China received further impetus at the 11th Congress of the Chinese Association of Orthopaedic Surgeons (CAOS) held in partnership with the North American Spine Society (NASS). At this event, Fule Science & Technology Development, Beijing, and Invibio Biomaterial Solutions partnered to […]
10 MORE Corpectomy Devices to Know!
Today we have included in our Corpectomy Section (CP) 10 more devices to know. We have included expandable devices as well as other differenciated corpectomy cages (Take into account that some of them may be already out of the market). As we said, there are more than 150,000 spinal fractures in North America* with an […]
OrthoPediatrics, Osseus, and many others…! SPINE Catalogue From A to Z: 18 Spine Companies to know with “O”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
Nuvasive, Neurostructures, and many others…! SPINE Catalogue From A to Z: 13 Spine Companies to know with “N”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
Mahe Medical USA launches Spine Emergency Screw Removal system and makes it available via Amazon
Mahe Medical USA announced the introduction and availability of its universal spine emergency screw removal system. “Anything that can be done in the OR to help the patient through a procedure in a shorter period of time without impacting the outcome should be celebrated and that’s exactly what the Mahe spine emergency screw removal system […]
Medtronic, Medacta, Medicon, Medicrea and many others…! SPINE Catalogue From A to Z: 20 Spine Companies to know with “M”
The Spine market is dominated by the following 10 global companies:Medtronic, DePuy Synthes, NuVasive Inc, Globus Medical, Stryker Corporation, Zimmer Biomet, K2M Group Holdings Inc, Alphatec Spine and Aesculap Implant Systems But in addition to these companies, there are more than 300 manufacturers worldwide. Many weeks ago, we started to display the updated list of all […]
SPINE Companies Catalogue From A to Z: 7 Spine Companies to know with “L”
We are already building (week by week) the new SPINE Catalogue that will include most of the Spinal companies worldwide. We want to promote and build awareness of most of the market players, even the smallest ones. Today we present all the Spinal companies which name start with L including LDR, Life Spine and many […]
Global spine market growth 2018-2023. Updated: 150 Companies from “A to K”
Over the recent years, global spinal fusion market has been witnessing growth, on account of several driving factors including rising healthcare awareness amongst consumers, surging prevalence of spinal deformities due to accidents along with increasing health expenditure in developing countries. Moreover, ongoing demographic shift towards geriatric population with significant population suffering from inveterate spinal ailments, […]
SPINE Companies Catalogue From A to Z: 6 Spine Companies to know with “J”
This week, we present the TENTH chapter (J) that includes all the Spinal companies which name start with J including Joimax among others.In that list you can find 6 Companies. Please visit the following page: https://thespinemarketgroup.com/category/spine-companies/page/12/ For any question please contact us: [email protected] or [email protected]
SPINE Companies Catalogue From A to Z: 17 Spine Companies to know with “I”
This week, we present the NINTH chapter (I) that includes all the Spinal companies which name start with I including Innovasis or Implanet among others.In that list you can find 17 Companies. Please visit the following pages: https://thespinemarketgroup.com/category/spine-companies/page/10/ https://thespinemarketgroup.com/category/spine-companies/page/11/ For any question please contact us: [email protected] or [email protected]
We are proud and happy to announce that Rudischhauser Surgical Instruments and Implants Manufacturing is Sponsor of SPINEMarketGroup in 2018!
Thank you Rudischhauser Surgical Instruments and Implants Manufacturing! On behalf of TheSPINEMarketGroup team, we would like to thank you for your PLATINUM Sponsorship and for your support this year 2018. About Rudischhauser Surgical Instruments and Implants Manufacturing Your partner and OE-Manufacturer of superior instruments, implants and complete set configurations for spinal, orthopaedic and trauma injuries.Rudischhauser […]
SPINE Companies Catalogue From A to Z: 6 Spine Companies to know with “H”
This week, we present the EIGHT chapter (H) that includes all the Spinal companies which name start with H including HD Lifesciences among others.In that list you can find 6 Companies. Please visit the following page: https://thespinemarketgroup.com/category/spine-companies/page/10/ For any question please contact us: [email protected] or [email protected]
SPINE Companies Catalogue From A to Z: 13 Spine Companies to know with “G”
This week, we present the SEVENTH chapter (G) that includes all the Spinal companies which name start with G including Globus Medical among others.In that list you can find 13 Companies. Please visit the following page: https://thespinemarketgroup.com/category/spine-companies/page/9/ For any question please contact us: [email protected] or [email protected]
Renovis Surgical Receives FDA Clearance for Tesera Trabecular Technology™ Hyperlordotic ALIF Interbody Spinal Fusion System
REDLANDS, Calif. and AUSTIN, Texas, June 28, 2018 /PRNewswire/ — Renovis Surgical Technologies, Inc. today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market the Tesera® SA Hyperlordotic ALIF Interbody Spinal Fusion System. Tesera SA is a porous titanium stand-alone anterior lumbar interbody fusion system featuring a […]
EOS imaging Announces First Private Practice Installation of An EOS® System in Germany
PARIS–EOS imaging (Paris:EOSI) (Euronext, FR0011191766 – EOSI – Eligible PEA – PME), the pioneer of 2D/3D imaging and data solutions for orthopedics, today announced the installation of an EOS® imaging system at ATOS Klinik Heidelberg, establishing it as first private practice in Germany to offer the low-dose 2D/3D imaging system. The system will be available at […]
GS Medical Announces New Product Launch and Successful Cadaver Workshop
IRVINE, Calif., June 27, 2018 (GLOBE NEWSWIRE) — GS Solutions Inc., DBA GS Medical USA (GSM), a leader and innovator of surgeon-driven spinal technology that advances patient care, today announced the launch of the AnyPlus® Extended Tab Minimally Invasive Pedicle Screw system. The AnyPlus® Extended Tab Minimally Invasive Pedicle Screw System was engineered to offer […]
Globus Medical Announces ExcelsiusGPS® Milestone: 3,000 screws implanted
AUDUBON, Pa., June 26, 2018 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, announced today a new clinical milestone with the successful implantation of over 3,000 screws using ExcelsiusGPS®, the company’s revolutionary robotic guidance and navigation system. Since the launch of ExcelsiusGPS® in late 2017, orthopedic and neurosurgeons have used the […]
Spineart announces the first European PERLA® surgeries
Spineart is proud to announce the first European PERLA® surgeries that simultaneously took place in Spain with Dr. ULLOA and Dr. DARDER, in Ireland with Dr. POYNTON and in Germany with Prof. RINGEL. PERLA® is a Posterior Cervical System offering a complete range of sterile-packed implants and intuitive instruments in a single compact set.The system […]