Bordeaux, Boston, September 15, 2020 – 5.45 pm CEST: IMPLANET (Euronext Growth: ALIMP, FR0013470168, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral and knee-surgery implants, announces its results for the first half of the year to June 30, 2020, as approved by the Board of Directors on September 15, 2020.
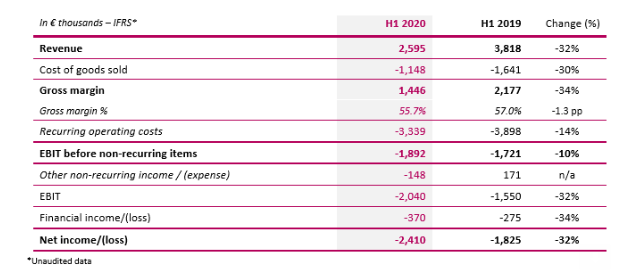
Implanet CEO Ludovic Lastennet said: “These first-half results illustrate the slowdown in activity due to the COVID-19 pandemic, embodied by the halting and postponement of scheduled surgical operations since March. Measures implemented in early spring have enabled us to navigate this period as best as possible. We reacted swiftly, and the decrease in activity was partly offset by adapting our cost structure and managing our operations in France and abroad. The latest trends confirm the gradual upturn in activity, we anticipate recovering a portion of the lost revenue we were unable to generate during this period during the second half of the year. Nevertheless, we are keeping a close eye on the continually changing public health situation and will activate every lever at our disposal in order to pursue our development by capitalizing on Implanet’s assets and product ranges”.
Upcoming financial events:– Q3 2020 revenue, October 13, 2020 after market close.
About Implanet
Founded in 2007, Implanet is a medical technology company that manufactures high-quality implants for orthopedic surgery. Its activity revolves around two product ranges, the latest generation JAZZ® implant, designed to improve the treatment of spinal pathologies requiring vertebral fusion surgery, and the MADISON implant designed for first-line prosthetic knee surgery. Implanet’s tried-and-tested orthopedic platform is based on product traceability. Protected by four families of international patents, JAZZ® and MADISON have obtained 510(k) regulatory clearance from the Food and Drug Administration (FDA) in the United States, the CE mark as well as the ANVISA authorization in Brazil. Implanet employs 36 staff and recorded 2019 sales of €7.4 million. For further information, please visit www.implanet.com. Based near Bordeaux in France, Implanet established a US subsidiary in Boston in 2013. Implanet is listed on Euronext™ Growth market in Paris.The Company would like to remind that the table for monitoring the equity line (OCA, OCAPI, BSA) and the number of shares outstanding, is available on its website: http://www.implanet-invest.com/suivi-des-actions-80.www.implanet.com
SOURCE:https://www.newcontact.eu/
