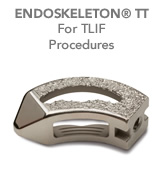
Titan Spine offers a full line of interbody fusion devices featuring its surface technology. The proprietary ENDOSKELETON® interbody devices are designed to promote bony ongrowth, optimize intervertebral stability, allow for increased bone graft material, and permit excellent visualization on postoperative CT and MRI scans.
Features:
- Smooth leading nose and edges allow for easy insertion
- Available in fourteen different sizes to accommodate various patient anatomies
- SURFACE TECHNOLOGY: Proprietary textured macro surface on the superior and inferior surfaces provide strong initial bone purchase. Micro and cellular textures on all surfaces support multi-axial new bone growth.
- UNIQUE SHAPES: Enable majority of the devices to be placed on the apophyseal ring, the strongest portion of the vertebral endplate, to preserve endplate integrity and reduce the risk of implant subsidence.
- LARGE WINDOWS: Devices utilize less material and feature extremely large windows for excellent postoperative radiographic visualization and fusion evaluation, as well as increased bone graft volume. Furthermore, side windows allow for additional bone growth in the axial plane, unlike traditional interbody devices that only allow for cephalad/caudal bone growth.
- SIMPLIFIED SURGICAL TECHNIQUE: The ENDOSKELETON® line of devices is supported by simple, intuitive instrumentation for easy and consistent implantation. The devices are available in a variety of sizes to accommodate various patient anatomies.
About TLIF
A transforaminal lumbar interbody fusion (TLIF) is performed to remove a portion of a disc that is the source of back or leg pain. Bone graft is used to fuse the spinal vertebrae after the disc is removed. TLIF provides fusion of the front and back of the lumbar spine. The front portion of the spine, called the anterior column, is stabilized by the interbody spacer and bone graft. The back portion, or posterior column, is locked in place with pedicle screws, rods and additional bone graft, alongside the backs of the vertebra.
Headquartered in Mequon, Wisconsin, Titan Spine is a medical device manufacturer established in 2006 specializes in the design, manufacture and marketing of titanium interbody cage devices that, for the first time, participate in the fusion process. Its ENDOSKELETON platform is distributed in the U.S. and Europe through the Company’s sales force and a network of independent distributors. http://titanspine.com