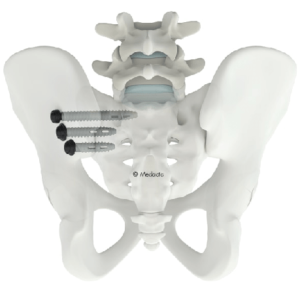
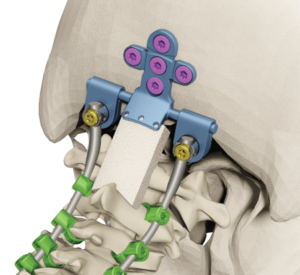
CASTEL SAN PIETRO, Switzerland–(BUSINESS WIRE)–Family-owned orthopedics leader Medacta International today announced the first surgery utilizing its Medacta Unconstrained Screw Technology Sacro-Iliac Joint Screw System (M.U.S.T. SI) for sacroiliac joint fusion surgery, following clearance by the U.S. Food and Drug Administration (FDA). The M.U.S.T. Sacro-Iliac Joint Screw System is designed for sacroiliac joint fusion for patients […]
NEWS
NuVasive Announces Key Finance Appointments
SAN DIEGO, June 11, 2018 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced the appointments of Lisa Pecora as vice president, Global Commercial Finance and Tristan Ribar as vice president, Corporate Finance and Treasury. In addition, Marc Rosenbaum is promoted to corporate controller. As the head of Global […]
Posterior Cervical Fusion Market: 40 systems to Know | Encore 10 more systems!
Last weeks we have been publishing about the attractiveness of the Posterior Cervcial market and about the different systems. Today we have included 10 more Posterior cervical products added to the 30 already included. To know about 40 Posterior Cervical Systems, please visit: https://thespinemarketgroup.com/category/posterior-cervical/. Medtronic: Vertex Reconstruction DepuySynthes:Synapse Nuvasive:Vuepoint Globus Medical: Quartex Stryker: Oasys Zimmer Biomed:Lineum […]
SPINE Companies Catalogue From A to Z: 23 Spine Companies to know with “B”
We started last week to work on the new SPINE Catalogue that will include most of the Spinal companies worldwide. We want to promote and build awareness of most of the market player even the smallest ones. Today we present the SECOND chapter (B) that include all the Spinal companies which name start with B […]
Centinel Spine Announces 510(k) Clearance of FLX™ Platform of 3D Printed All-Titanium Interbodies
NEW YORK, June 7, 2018 /PRNewswire/ — Centinel Spine, LLC is pleased to announce that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its FLX™ Platform of Integrated Interbody™ and non-integrated interbody fusion devices. Centinel Spine is the largest privately-held spine company, focused on anterior column reconstruction. FLX devices are 3D-printed, […]
K2M Surpasses 100th Product Milestone with FDA Clearance and Launch of OZARK™ Cervical Plate Systems
LEESBURG, Va., June 07, 2018 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today surpassed its 100th product milestone with the announcement of U.S. Food and Drug Administration (FDA) 510(k) clearance and commercial launch […]
MedTech Momentum Inc. Expands Global Footprint with the Creation of MedTech Momentum Europe SAS
ALTAMONTE SPRINGS, FLA. JUNE 05, 2018–MedTech Momentum, Inc. (http://www.MedTechMomentum.com) a full-service Medical Marketing agency with decades of experience in the medical device arena, announced today the creation of MedTech Momentum Europe SAS, in France. Claire Daculsi will assume the title of President. She comes to the MedTech Momentum Group with a distinguished track record in medical […]
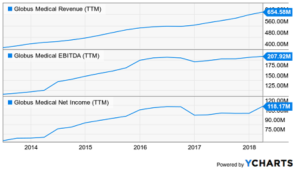
Globus Medical Is Gaining Share In Its Mainstay Market
(SEEKINGALPHA.COM )–Globus Medical (NYSE:GMED) is a medical device company that focuses on the design, development, and commercialization of musculoskeletal implants that promote healing in patients with spine disorders and products for patients suffering from orthopedic traumas; all in all some 180 products. GMED is doing well because it is growing considerably faster than the market; […]
CoreLink Acquires Expanding Orthopedics, Inc.
ST. LOUIS–(BUSINESS WIRE)–CoreLink, LLC, a fast growing, vertically integrated designer and manufacturer of spinal implant systems, announced today the acquisition of the privately-held Israeli medical device company Expanding Orthopedics, Inc (EOI). Along with the FDA cleared expanding and articulating FLXfit™ and FLXfit 15™ titanium TLIF interbody systems, CoreLink has also acquired a broad array of […]
Vertiflex Announces Appointments to Board of Directors, Ray Baker, MD, and Richard Mott
CARLSBAD, Calif.–(BUSINESS WIRE)–Vertiflex, Inc., a leading innovator of advanced, minimally invasive interventions for spinal stenosis, today announced two new members of its Board of Directors, adding strong clinical and industry expertise to the company’s leadership team. Ray Baker, MD, brings extensive clinical experience to Vertiflex. He is currently chief medical officer at Relievant Medsystems, Inc. […]
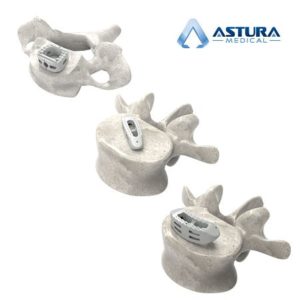
Astura Medical Receives FDA 510(k) Clearance For Cervical and Lumbar HA PEEK Interbody Systems
CARLSBAD, CA – June 4, 2018 – Astura Medical, a high growth, innovative spine technology company, today announced that it received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for its ALTA ACDF Interbody Spacers and HALF DOME Lumbar Interbody Spacers in PEEK-OPTIMAä HA Enhanced from Invibio Biomaterial Solutions. Combining the clinically proven […]
Aesculap Implant Systems Announces U.S. Military’s TRICARE Now Covers Lumbar Total Disc Replacement
CENTER VALLEY, Pa., June 4, 2018 /PRNewswire/ — Aesculap Implant Systems, LLC announced today that TRICARE now covers the company’s activL® Artificial Disc for active duty and retired military members and their families in need of Lumbar Total Disc Replacement (CPT® procedure code 22857). Aesculap is a privately-held spine and orthopaedic implant company and a […]
IMPLANET Announces Opening of a United Kingdom Sales Branch
BORDEAUX, France & BOSTON–(BUSINESS WIRE)–Regulatory News:IMPLANET (Paris:ALIMP) (OTCQX:IMPZY) (Euronext Growth: ALIMP, FR0010458729, eligible for PEA-PME equity savings plans; OTCQX: IMPZY), a medical technology company specializing in vertebral and knee-surgery implants, announces the opening of a branch in the United Kingdom, accelerating expansion and direct growth in the growing adult degenerative market. The Implanet UK branch […]
Spineology Rampart One™ Standard ALIF Interbody Fusion System Gains FDA Clearance for Stand-Alone Use
ST. PAUL, Minn.–(BUSINESS WIRE)–Spineology Inc., announces FDA clearance of its Rampart One™ Standard ALIF device allowing it to be used with or without supplemental fixation. “Following the success of our beta launch, this stand-alone clearance will provide additional momentum as we prepare for the full market release of the Rampart One ALIF Interbody Fusion System,” […]
Surgeons Preferred HD LifeSciences™ Hive™ Based on Radiographic Visualization
(Ilsa Webeck)–In the last few years, companies have been launching new and improved interbody fusion devices produced using advanced manufacturing technologies, including 3D printing. Commercially available devices are marketed “to actively participate in the fusion process”, to “create a favorable environment for cell attachment and proliferation,” and be an “active participant in the fusion process.” […]
Camber Spine Announces 510(k) Approval Of ENZA™-A Titanium ALIF
WAYNE, Pa., June 4, 2018 /PRNewswire/ — Camber Spine today announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market its ENZA™-A Titanium Anterior Lumbar Interbody Fusion (ALIF) system, a unique, minimally invasive interbody fusion device providing integrated fixation. Daniel Pontecorvo, CEO, said, “ENZA™-A leverages two of Camber […]
SPINE Companies Catalogue From A to Z: 25 Spine Companies to know with “A”
Some years ago, we started to publish what we called SPINE Catalogue because it included most of the Spinal companies worldwide. The objective was to promote and build awareness of most of the market player even the smallest ones. Last year we accounted for almost 400 companies from many different countries. Now, it is time […]
Did an Orthopedic Surgeon accidentally shoot a Sales Rep?
(ryortho.com)–In Cayce, South Carolina a 46-year old orthopedic surgeon has been accused of fatally shooting William Holland, a sales rep employed by CrossLink Orthopaedics, one of Stryker’s largest distributors.According to the police initially, this was a tragic, horrible gun accident.In their first recounting of events, the police said that the surgeon, Adam Lazzarini, M.D., had […]
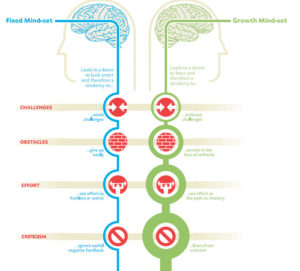
Here’s why your attitude is more important than your intelligence
When it comes to success, it’s easy to think that people blessed with brains are inevitably going to leave the rest of us in the dust. But new research from Stanford University will change your mind (and your attitude). Psychologist Carol Dweck has spent her entire career studying attitude and performance, and her latest study […]
Is the Posterior Cervical Fusion Market Attractive? (Final Part) 30 Systems to Know!
Posterior Cervical fusion technique involves the fusion of more than two vertebrae to prevent unwanted motion of the discs or the compression of posterior spaces between the cervical vertebrae. Posterior Cervical Fusion is a procedure intended for the stabilization of the cervical spine through a posterior approach. The procedure is commonly performed using hooks, plates, […]
iOrthopedics Inc. (iOI), Las Vegas, Nevada, is pleased to announce patent awards and allowances for its Universally Expanding Cage (UEC)
iOrthopedics Inc. (iOI), Las Vegas, Nevada, is pleased to announce patent awards and allowances for its Universally Expanding Cage (UEC) giving exclusive rights in the spine expandable interbody arena. Validation is granted by the U.S. Patent Office (US9,861,494 and US9,999,515) with confirmed claims covering a device and method for independently altering the distance and angle […]
Safe Orthopaedics acquires a direct sales force in the United Kingdom
Eragny-sur-Oise, France, June 1st, 2018 at 7:30am CEST – SAFE ORTHOPAEDICS (FR0012452746 – SAFOR), a company specialising in the design and marketing of single-use implants and instruments facilitating the minimally invasive treatment of spinal fracture conditions, is today announcing a deal to acquire the sales force of UK company Qspine, which specialises in distributing medical […]
Posterior Cervical Fusion Market (Part2) 10 More Systems to Know!
The posterior cervical fusion is a type of cervical fusion surgery, that accounts the 15% to the cervical devices market share. Although it is still an small segment of the overall Spinal market, it is growing due to the increasing prevalence of osteoarthritis and rheumatoid and the rising aging population. The estimated market value of the posterior […]
In’Tech Medical enters into agreement to acquire Bradshaw Medical, industry leader in silicone overmolded solutions, ratchet and torque devices
Memphis, Tenn. May 23, 2018–In’Tech Medical SAS (http://www.intech-medical.com), the leading Contract Manufacturer of medical devices in Orthopedics, announced today that it has entered into a binding agreement to acquire world renowned instruments and silicone overmold company, Bradshaw Medical Inc. The transaction, supported by Eurazeo PME, has been approved by the Board of Directors of both organizations. Based in […]
Is the Posterior Cervical Fusion Market Attractive? 7 Systems to Know
Posterior Cervical fusion technique involves the fusion of more than two vertebrae to prevent unwanted motion of the discs or the compression of posterior spaces between the cervical vertebrae. Posterior Cervical Fusion is a procedure intended for the stabilization of the cervical spine through a posterior approach. The procedure is commonly performed using hooks, plates, […]
Spine Industry: Growth Through Acquisitions! 7 Relevant Acquisitions in the last 12 months
During the last year, the spinal market has seen more than 7 acquisitions for many different reasons. Buy Sales or Market Share: Although the global spinal implants market is still growing at an expected rate of 6.6% CAGR 2022, acquisitions are an importante alternative way for Spinal Companies to achieve more than single digit organic spine […]
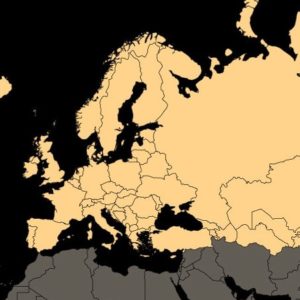
Europe spinal implants market: (Part IV) The Top 20 European Manufacturers Of Spinal Implants
Spine surgery is a relatively mature market — all segments of the spine can be treated, significant share has been garnered by a few major companies, and there are no radically new technologies disrupting current market positions. Yet, trends in the patient population, the active innovation by all companies (see further, below, on market shares […]