Why Medtronic wants to acquire MEDICREA®? We have asked ourselves that question several times since last Wednesday the press release with that news was published. MEDICREA® is a Small and Medium sized Enterprise (SME) with 200 employees worldwide, which includes 50 who are based in the U.S.Its sales in 2019 were 32 million with an […]
NEWS
Empirical Spine Completes Enrollment in Pivotal IDE Clinical Trial Studying LimiFlex in Degenerative Spondylolisthesis
SAN CARLOS, Calif., July 16, 2020 /PRNewswire/ — Empirical Spine, Inc., maker of the LimiFlex Paraspinous Tension Band, today announced completion of enrollment in its U.S. IDE trial studying the use of LimiFlex with a decompression for patients suffering from degenerative spondylolisthesis with lumbar spinal stenosis. The trial compares outcomes of patients who receive either LimiFlex or […]
SPINEART announces over 1,000 patients treated with SCARLET® AL-T in the USA.
Spineart USA announces over 1,000 patients treated with SCARLET® AL-T, its ALIF system launched in 2019. The implant was developed using the company’s flagship Ti-LIFE Technology, a titanium bone-like structure that supports bone cell adhesion and allows for bone ingrowth. A number of design features make SCARLET® AL-T unique: the 3D printed cages feature tunnels […]
Implanet extends its JAZZ Cap® intellectual property to Japan
Bordeaux, Boston, July 16, 2020 – 8.00 am CEST: IMPLANET (Euronext Growth: ALIMP, FR0013470168, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral andknee-surgery implants, today announces that its JAZZ Cap®technology has been patented in Japan. Ludovic Lastennet, CEO of IMPLANET, commented: “Japan represents the world’s third largest market in the treatment […]
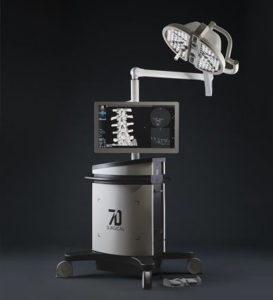
7D Surgical Achieves CE Mark Certification for Machine Vision Image Guided Spine Surgery
TORONTO, July 15, 2020 /PRNewswire/ — 7D Surgical announced today that it has achieved CE Mark certification for its innovative Machine-vision Image Guided Surgery (MvIGS) system for spine surgery. This achievement clears the way for 7D Surgical to commence commercialization efforts of its spinal platform across the European market and additional global regions. The 7D Surgical MvIGS System […]
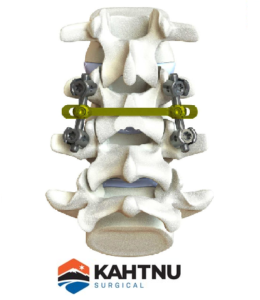
KAHTNU Surgical Receives $2M Funding, Partners with U.K. Ortho Consulting Group for Global Sales Expansion
SOLDOTNA, Ala., July 15, 2020 /PRNewswire/ — KAHTNU Surgical, a U.S. medical technology company based in Alaska, announced today it had completed a $2.15 million Series A round of financing led by Kinetic Medical Co., Ltd. The investment will help KAHTNU Surgical aggressively expand worldwide sales and marketing efforts as well as broaden and accelerate product development for spine and […]
Medtronic to Acquire Medicrea
DUBLIN and LYON, France, July 15, 2020 (GLOBE NEWSWIRE) — Medtronic plc (NYSE:MDT), a global leader in medical technology, and Medicrea (Euronext Growth Paris: FR0004178572 – ALMED Medicrea; OTCQX Best Market – MRNTF), a pioneer in the transformation of spinal surgery through artificial intelligence, predictive modeling and patient specific implants, today announced that they have entered into a tender offer […]
LimiFlex™ Paraspinous Tension Band IDE Study Results to be Presented at the 27th International Meeting on Advanced Spine Techniques (IMAST); Nominated for Best Clinical Research Paper Award
SAN CARLOS, Calif., July 14, 2020 /PRNewswire/ — Empirical Spine, Inc., a Silicon-Valley based medical technology company, announced today that data from the IDE study of the company’s LimiFlex Paraspinous Tension Band for patients suffering from degenerative spondylolisthesis with lumbar spinal stenosis, will be presented at the 27th International Meeting on Advanced Spine Techniques. IMAST, originally scheduled April […]
Nanovis Experiences Record High Sales in June 2020
CARMEL, Ind., July 14, 2020 /PRNewswire/ — Nanovis, a unique technology company focused on nanotechnology for improving bone growth and fighting infection, today reported a record sales month in June 2020. “I am inspired by the commitment and care demonstrated by our customers and our Nanovis Team in helping a record number of patients with Nanovis implants in June […]
From our opinion, What are the three best lateral systems on the market?
In today’s market, there are numerous lateral systems and most of them are of very high quality. It is extremely difficult to choose the best three, but for this we have taken into account the following criteria: Retractor: The implant is important, but a good retractor is absolutely key. Therefore, all those systems that do not have a […]
Spine Wave Announces the Commercial Launch of Tempest® Allograft Bone Matrix
SHELTON, Conn., July 14, 2020 (GLOBE NEWSWIRE) — Spine Wave is pleased to announce the successful completion of a limited market release of Tempest® Allograft Bone Matrix and its entry into the spinal biologics market. Tempest® Allograft Bone Matrix allows the company to strongly compete in the demineralized bone allograft segment of that market and […]
ulrich medical USA® Brings Strong Momentum® to U.S. Product Launch
ST. LOUIS, July 13, 2020 /PRNewswire/ — ulrich medical USA, Inc., a medical device company focused on developing and commercializing musculoskeletal implant technologies in the United States announced the nationwide commercial release of the Momentum Posterior Spinal Fixation System. “After completing hundreds of procedures during the alpha launch, Momentum has already made its mark with an enthusiastic reception and positive reviews from our alpha surgeon […]
Nexxt Spine Expands Sales Team with Hiring of Additional Regional Sales Managers
July 13, 2020- Noblesville, IN- Nexxt Spine, LLC recently announced the appointment of new management to the sales department. Augmentation of the Nexxt Spine team comes during a period of tremendous expansion for the company.This year Nexxt Spine has already received FDA 510(k) clearance on 4 new products and will double their Indianapolis based headquarters, […]
SPINEMarketGroup YouTube Channel Update: List of the Latest Videos added.
We want to offer you all the audiovisual information about the Spine industry available, so that you can have it at your disposal on a YouTube channel. Our website thespinemarketgroup.com offers you news about the Spine industry as well as about products and competitors. In many cases we have included Surgical techniques and brochures, but […]
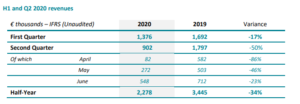
Spineway:2020 1st semester turnover and prospects
Ecully, July 10, 2020 – 12 pm Revenue for the first half of 2020 Gradual recovery Cash position of €3.2M secure through end of June The first half of this year was understandably affected by the health crisis caused by the worldwide Coronavirus pandemic, which led to the cancellation of all non-essential surgeries. During this unprecedented […]
Spineart reaches definitive agreement to acquire Meditech Spine LLC.
Spineart, a global spine player with headquarters in Geneva, Switzerland and US operations in Laguna Hills, California, has agreed to acquire 100% of Atlanta-based Meditech Spine LLC. Spineart has been a distributor of Meditech Spine’s products in the US since 2019. The companies anticipate significant synergies between the two organizations. As part of the transaction, […]
MEDICREA Reports First Half 2020 Sales
Lyon and New York, July 9, 2020 – The MEDICREA® Group (Euronext Growth Paris: FR0004178572 –ALMED ; OTCQX Best Market –MRNTF), pioneering the transformation of spinal surgery through ArtificialIntelligence, predictive modeling and patient specific implants with its UNiD™ ASI (Adaptive Spine Intelligence) proprietary software platform, services and technologies, publishes sales for the first half of […]
SpineGuard posts H1 2020 revenue
PARIS and BOULDER (CO), July 9, 2020 – 08:00am CEST – SpineGuard (FR0011464452 – ALSGD), an innovative company that deploys its DSG® (Dynamic Surgical Guidance) sensing technology to secure and streamline the placement of bone implants, announced today that its first half 2020 revenue was € 2.3 M. Pierre Jérôme, Co-founder, Chairman and CEO of […]
Which are the most relevant Spinal Fixation Bands?
Recent literature has shown clinical benefits of posteromedial translation using sublaminar bands, for treatment of AIS. Adolescent idiopathic scoliosis (AIS) is a tridimensional deformity that affects the trunk height changing both in coronal and sagittal profiles, with usually a loss of thoracic kyphosis and lumbar lordosis and a trend to cervical kyphosis. The aim of surgical […]
HAPPE Spine™ Announces Additional Technology Patent For Porous And Bioactive PEEK
HOLLAND, Mich., July 8, 2020 /PRNewswire/ — HAPPE Spine announces that it has received notice of allowance from the U.S. Patent and Trademark Office for patent claims providing broad protections for its unique material and manufacturing technology providing fully porous, bioactive polyetheretherketone (PEEK) technology (U.S. Pat. App. No. 14/078,614). HAPPE, a company focused on developing next generation orthopedic implants, is one of […]
Life Spine Announces Initiation of the PROLIFT® Lateral Expandable System Outcomes Study
Huntley, IL,July 7, 2020 –Life Spine, a medical device company that designs, develops, manufactures and markets products for the surgical treatment of spinal disorders, announced todaythe initiation of the PROLIFT Lateral Expandable System prospective study. “Research and Clinical studies are incredibly important to our organization and to the spine community,” said Michael Butler, President and […]
Aesculap Implant Systems Announces New Positive Third-Party Clinical Guidelines that May Introduce Access for More than 8.5 Million Commercially Insured Americans to the activL® Artificial Disc
CENTER VALLEY, Pa., July 7, 2020 /PRNewswire/ — Aesculap Implant Systems, LLC today announced that National Imaging Associates (NIA), a Magellan Health Company, has updated its “Clinical Guidelines for Medical Necessity for Musculoskeletal and Surgery” to include a medical necessity recommendation for Lumbar Total Disc Replacement (TDR), including the latest generation activL® Artificial Disc. Multiple U.S. commercial payers […]
Implanet announces H1 2020 revenue
Bordeaux, Boston, July 7, 2020 – 5.45 pm CEST: IMPLANET (Euronext Growth: ALIMP, FR0013470168, eligible for PEA-PME equity savings plans), a medical technology company specializing in vertebral andknee-surgery implants, today announces its revenue for the first half of 2020. Impact of the Covid-19 pandemic:To provide greater clarity regarding the impact of the Covid-19 pandemic, Implanet has […]
Orthofix Announces FDA Clearance and First Patient Implants of FIREBIRD SI Fusion System for Low Back Pain
LEWISVILLE, Texas–(BUSINESS WIRE)–Jun. 29, 2020– Orthofix Medical Inc. (NASDAQ:OFIX), a global medical device company focused on musculoskeletal healing products, announced the U.S. Food and Drug Administration (FDA) 510(k) clearance and the first patient implants of the FIREBIRD™ SI Fusion System. Designed to compress and stabilize the sacroiliac joint (also called the SI joint) during fusion, the FIREBIRD SI Fusion System is […]
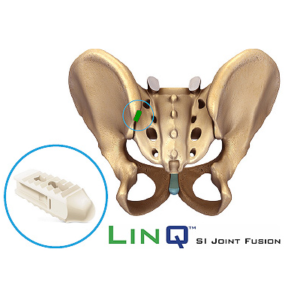
Michael Enxing joins PainTEQ Board of Directors
TAMPA, Fla., July 1, 2020 /PRNewswire/ — PainTEQ, a leading medical device innovator, announced the appointment of Michael Enxing, Chief Commercial Officer of Boston Scientific Corporation, Neuromodulation Division. PainTEQ is the creator of LinQ (pronounced “link”), a minimally invasive therapy for SI joint dysfunction. With nearly three decades of executive experience, Enxing provides commercial know-how and strategic insight for medical device businesses. Most recently, […]
Aurora Spine Announces New Chief Financial Officer
CARLSBAD, Calif., July 02, 2020 (GLOBE NEWSWIRE) — Aurora Spine Corporation (“Aurora Spine” or the “Company”) (TSX VENTURE:ASG) announced the promotion of Chad Clouse to the position of Chief Financial Officer (CFO), effective July 2, 2020. Mr. Clouse graduated from Pennsylvania State University and is a veteran of the United States Army, having served with […]
RTI Surgical Holdings, Inc.® to Hold Virtual-Only Annual Meeting of Stockholders
DEERFIELD, Ill., July 02, 2020 (GLOBE NEWSWIRE) — RTI Surgical Holdings, Inc. (Nasdaq: RTIX), a global surgical implant company, today announced that, due to the public health impact of the coronavirus outbreak (COVID-19), its Annual Meeting of Stockholders, to be held on Wednesday, July 15, 2020 at 9:00 a.m. Central Daylight Time (the “Annual Meeting”), […]