ALACHUA, Fla.–(BUSINESS WIRE)–RTI Surgical, Inc. (Nasdaq: RTIX), a global surgical implant company, today signed an agreement to acquire Zyga Technology, Inc., with closing subject to filing with the state of Delaware. Zyga Technology is a leading spine-focused medical device company that develops and produces innovative minimally invasive devices to treat underserved conditions of the lumbar […]
NEWS
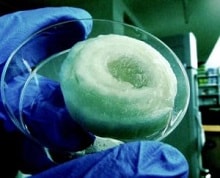
Scientists create spinal disc from mulberry silk
(deccanherald.com)–An Indo-US team of scientists has created an artificial inter-vertebral disc from mulberry silk. Because of its abilities to accurately mimic the human disc that acts as a soft cushion between two vertebra, the artificial biodisc has the potential to replace the metallic or ceramic or collagen-based discs that neuro-surgeons use at the moment to […]
Centinel Spine Completes Acquisition of prodisc Assets
NEW YORK, Dec. 22, 2017 /PRNewswire/ — Centinel Spine, LLC (CS) is pleased to announce the acquisition of the worldwide assets of the prodisc® Total Disc Replacement portfolio from DePuy Synthes Products, Inc. The prodisc line of products are total disc replacement (TDR) products with the longest history of use globally. The prodisc® portfolio includes prodisc® C and […]
Why an FDA Panel Shot Down Intrinsic’s Spinal Implant?
(MDDIONLINE.COM) –Although FDA doesn’t have to follow the recommendation of its advisory committees, a negative panel vote is never a good sign for a company hoping to bring a new device to the market. Intrinsic Therapeutic learned the hard way Tuesday that it’s usually best if clinical trials follow the protocol that was initially communicated to […]
The MIS Pedicle Screw Market is Growing. Competition is Fierce. Learn about the 75 most Relevant Systems.
According to iData Research, the U.S. minimally invasive spinal implants market was valued at $1.62 billion in 2016. This is expected to increase over the forecast period at a CAGR of 3.6% to reach $2.08 billion (iData Research). Traditionally, pedicle screw systems have been implanted through open surgery from posterior approach. However, a growing number […]
NuVasive To Acquire SafePassage
SAN DIEGO, Dec. 12, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), the leader in spine technology innovation, focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, and SafePassage™, a privately-held provider of intraoperative neurophysiological monitoring (IONM) services, announced today that they have entered into a definitive agreement under which NuVasive will acquire SafePassage. Terms […]
Cervical Artificial Disc Segment Expected to Comprise Nearly €57 million of the Overall Europe Motion Preservation Market by 2023. Learn about the 44 main devices
According to Europe Market Report for Spinal Motion Preservation Devices 2017 – MedCore by iData Research, Inc., procedural growth, favorable reimbursement policies, and its use as an alternative to spinal fusion are driving the European cervical artificial disc market. Growth rates will be maintained due to increased adoption rates and patient education. The total artificial […]
Why are Titanium Cages back after 20 years? Learn about 28 Titanium TLIF implants
Surgeons have historically preferred interbody spacers made of titanium because of their strength and long clinical history. But 20 years ago, everything changed with a new material called PEEK (polyetheretherketone) that appeared in the spinal market offering many advantages as elastic modulus close to bone, high biocompatibility and biostability, excellent resistance to compressive loading, and radiolucency. For almost […]
Congratulations to The SPINE GROUP on reaching 12,000 LinkedIn members
Congratulations, The SPINE Group, on topping 12000 members in this awesome group. Since 2008, The SPINE Group has been supporting the Spine Industry providing relationships, discussions, events and information .Thank you for your partnership and we encourage others to keep joining! About The SPINE Group This LinkedIn group is for all the people involved in the SPINAL business willing to receive fresh information […]
Sponsorship 2018 | When you’re competing in an extremely crowded marketplace as Spine, how can you stand out?
Differentiation is the KEY! When you’re competing in an extremely crowded marketplace as Spine, the thing you must avoid at all costs is becoming a faceless provider. Sponsorship offers the possibility to increase your Company Visibility and Differentiate! WHO Can Benefit from our Sponsorship? 1.- Spine Companies: You already Compete against at least 400 companies worldwide! […]
Medicrea Files a Lawsuit against K2M Spine, Inc.
LYON, France & NEW YORK–(BUSINESS WIRE)–The Medicrea Group (Euronext Growth Paris: FR0004178572 – ALMED), pioneering the convergence of healthcare IT and next-generation, outcome-centered device design and manufacturing with UNiD™ Adaptive Spine Intelligence (“ASI”) technology, announces that on Wednesday, November 8th, 2017, Medicrea USA, Inc., filed a lawsuit in the United States District Court for the […]
Is the Lumbar Total Disc Replacement (TDR) Back? Learn about the 10 most relevant Lumbar Artificial Discs
The lumbar artificial disc (LAD) market has experienced significantly lower procedural growth than other market segments as a result of a lack in clinical efficacy data and poor reimbursement. In 2014, the reimbursement environment severely limited the number of LAD procedures performed. However, reimbursement policies are expected to change because Medicare and other organizations are considering […]
Medical Device Sales Reps in the Operating Room: An Asset or a Liability?
November 1, 2017 (mddionline.com)–In this age of rapidly developing technologies, medical device sales representatives fill a unique role. In the business of surgery, sales reps not only deliver medical devices to hospitals as needed, but they ensure that surgical teams know precisely when and how a device and its components are to be used. Often knowing […]
New Studies Look At Cost And Benefits Of Robotic Surgery
As robotics in medicine becomes more widely adopted, two new studies look at the cost and advantages and disadvantages of robotic surgery versus freehand surgery. University of Stanford researchers conducted a multiyear analysis and study with 24,000 patients with kidney cancer who needed laparoscopic surgery to remove a patient’s kidney indicated that the two approaches had comparable patient outcomes and […]
Surgeon sues J&J’s DePuy in royalties dispute
OCTOBER 31, 2017 (massdevice.com)–A surgeon who helped Johnson & Johnson (NYSE:JNJ) subsidiary DePuy Synthes Spine develop instruments and implants for spine procedures wants a Massachusetts federal court to overturn an arbitrator’s ruling that the company doesn’t owe royalties on the products, according to a lawsuit filed last week in the Bay State. Dr. Carl Lauryssen and his company, Jordan […]
Exactech to be acquired by private equity firm
(Gainesville.com)–After 21 years as a public company, Exactech, a Gainesville-based medical device company, announced Monday it agreed to be acquired by TPG Capital, a global private equity platform of alternative asset firm TPG — making it private. Exactech’s board of directors approved the agreement, which says TPG Capital will acquire all common stock shares of […]
VEXIM to be Acquired by Stryker Corporation
BALMA, France–(BUSINESS WIRE)–Regulatory News:Stryker Corporation, a global leading medical technology company, acquired on October 24th, 2017 a majority of the share capital and voting rights of VEXIM, a medical device company specializing in the minimally invasive treatment of vertebral fractures listed on Euronext Growth (FR0011072602 – ALVXM). Stryker Corporation (NYSE:SYK) has indirectly acquired 4,115,151 VEXIM […]
In’Tech Medical Acquires Pyxidis, Manufacturer of Surgical Cases and Trays for the Medical Device Industry
MEMPHIS, TENN. (PRWEB) OCTOBER 24, 2017–In’Tech Medical SAS (http://www.intech-medical.com), the leading Contract Manufacturer of surgical instruments in Orthopedics, announced that it has acquired Pyxidis SA., a reputable French-based manufacturer of customized sterilization cases and trays for surgical Instruments. With over 40 years of experience and presence worldwide, Pyxidis is renowned for its total commitment to building medical […]
Will the Latest Smith & Nephew Rumor Actually Pan Out?
It must be that time of year.Wall Street is once again abuzz with speculation of a potential Smith & Nephew takeout. Deal rumors have swirled around the London-based medical device firm for years, this time fueled by CEO Olivier Bohoun’s retirement plans, coupled with unconfirmed reports that activist investor Elliott Management Corp. has taken a stake […]
Alphatec Denounces “Baseless” NuVasive Complaint
CARLSBAD, Calif., Oct. 11, 2017 (GLOBE NEWSWIRE) — Alphatec Holdings, Inc. (Nasdaq:ATEC), a provider of innovative spine surgery solutions with a mission to improve patient lives through the relentless pursuit of superior outcomes, released a statement in response to a recent complaint filed by NuVasive, Inc. (Nasdaq:NUVA) against Patrick S. Miles, Alphatec’s recently appointed Executive […]
Nuvasive Files Lawsuit Against Patrick S. Miles To Protect Corporate Assets And Stakeholders’ Interests
SAN DIEGO, Oct. 10, 2017 /PRNewswire/ — NuVasive, Inc. (NASDAQ: NUVA), a leading medical device company focused on transforming spine surgery with minimally disruptive, procedurally-integrated solutions, today announced that it has filed a lawsuit in the Delaware Chancery Court against Patrick S. Miles, former vice chairman of the Company and a member of NuVasive’s Board of Directors. The lawsuit asserts […]
Globus Medical Announces First Spine Surgeries Using ExcelsiusGPS™
AUDUBON, Pa., Oct. 10, 2017 (GLOBE NEWSWIRE) — Globus Medical, Inc. (NYSE:GMED), a leading musculoskeletal solutions company, today announced the first spine surgeries using ExcelsiusGPS™, a revolutionary robotic guidance and navigation system. Surgeries using ExcelsiusGPS™ were performed last week at The Johns Hopkins Hospital in Baltimore, Maryland and St. Mark’s Hospital in Salt Lake City, […]
Expandable interbody cages: Are they here to stay? Learn about the 27 most relevant devices
Will Expandable interbody cages succeed in the spinal market? According to many surgeons and many of the Spinal companies, these new devices may help surgeons improve efficiency in the operating room and increase patient safety during TLIF, PLIF and even Lateral cages procedures. These advantages are the following: Expandable interbody cages allow to the surgeon to […]
Spineguard Reports €6.0m Revenue and 13% Growth for the 9 Months of 2017, US Third-Quarter Growth of 23% cc
PARIS & SAN FRANCISCO–(BUSINESS WIRE)–Regulatory News: SpineGuard (Paris:ALSGD) (FR0011464452 – ALSGD), an innovative company that designs, develops, and markets disposable medical devices intended to make spine surgery safer, announced today that its 9 months revenue grew to €6.0 million, a 13% increase compared with the same period in 2016. Stéphane Bette, CEO and co-founder of […]
K2M Group Holdings, Inc. President & Chief Executive Officer Eric Major Elected Chairman of the Board of Directors
LEESBURG, Va., Oct. 04, 2017 (GLOBE NEWSWIRE) — K2M Group Holdings, Inc. (NASDAQ:KTWO) (the “Company” or “K2M”), a global leader of complex spine and minimally invasive solutions focused on achieving three-dimensional Total Body Balance™, today announced that President and Chief Executive Officer Eric Major has been elected Chairman of the Company’s Board of Directors, effective immediately. Major […]
ChoiceSpine™ Granted FDA Clearance for 3D Printed Vertebral Body Replacement Device
KNOXVILLE, TN (PRWEB) OCTOBER 04, 2017-ChoiceSpine LP, a privately-held spinal device manufacturer based in Knoxville, TN, announced that it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) to market HAWKEYE Ti, a 3D Printed Titanium Vertebral Body Replacement (VBR) device. “Additive manufacturing techniques create intricate three-dimensional implants, layer by layer, allowing a […]
4WEB Medical Announces Launch of Next Generation Anterior Spine Truss System
DALLAS, Oct. 4, 2017 /PRNewswire/ — 4WEB Medical, the industry leader in 3D printed implant technology, has announced the launch of its next generation interbody fusion product line for anterior lumbar spine procedures. The new release of the Anterior Spine Truss System recently received FDA clearance for several impactful line extensions along with some key new indications for […]